About ten years ago, I had a patient who came to see me for moderate carotid disease. While his carotid disease was asymptomatic, he also had metastatic colon cancer. With colectomy, cryoablation of liver mets, and chemotherapy, he was in remission. Every 6 to twelve months he had some kind of CT scan with contrast. His renal function was poor and this was blamed on his chemotherapy. While it had nothing to do with this patient, I thought to myself, “Having an aortic stent graft was a lot like having metastatic cancer in remission.” After a stent graft, the patient is forever tied to the health care system. Without surveillance, there may be an endoleak, sac expansion, rupture, and even death. Patients and vascular surgeons can make choices that lengthen life, improve its quality, and avoid the complications of disease. But what if a treatment becomes a condition and a burden on healthcare resources and the patient’s finances?
Fool me once…
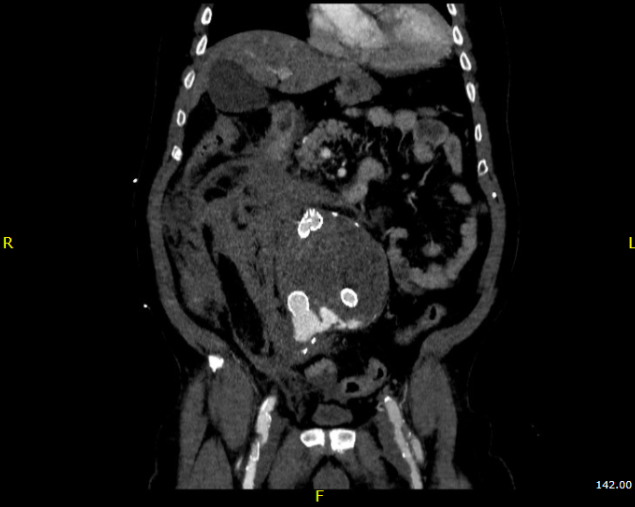
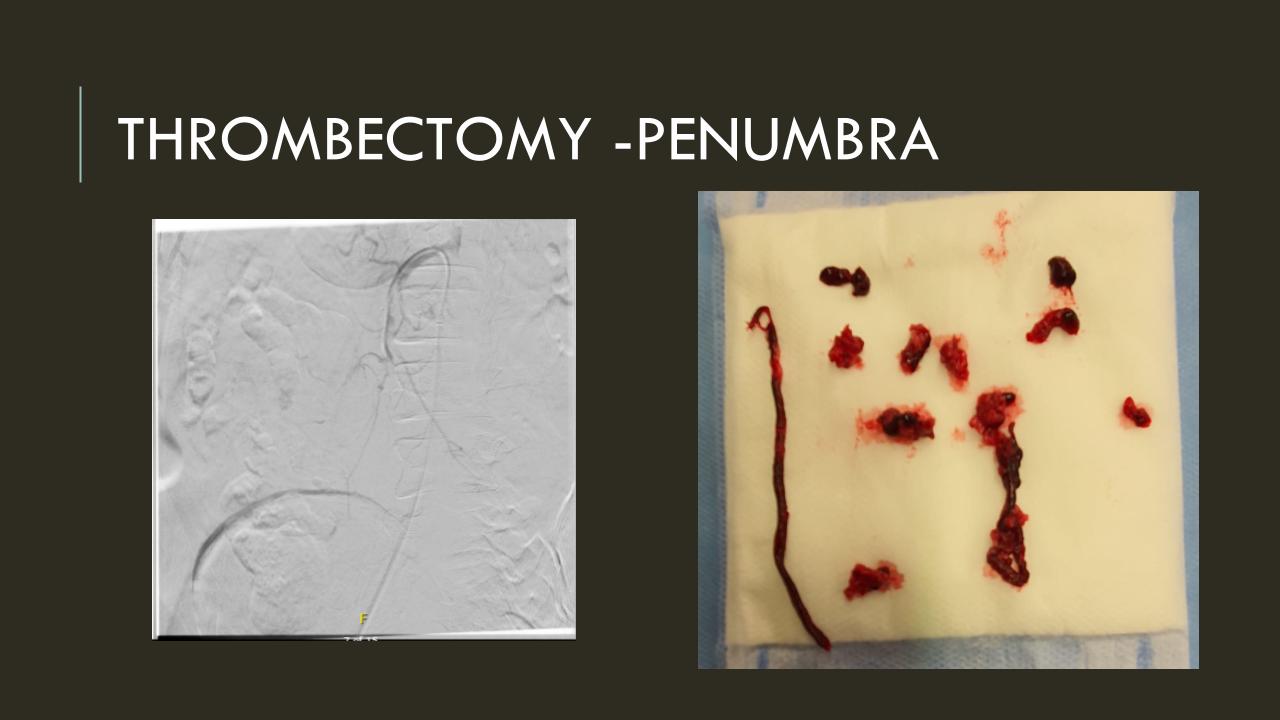
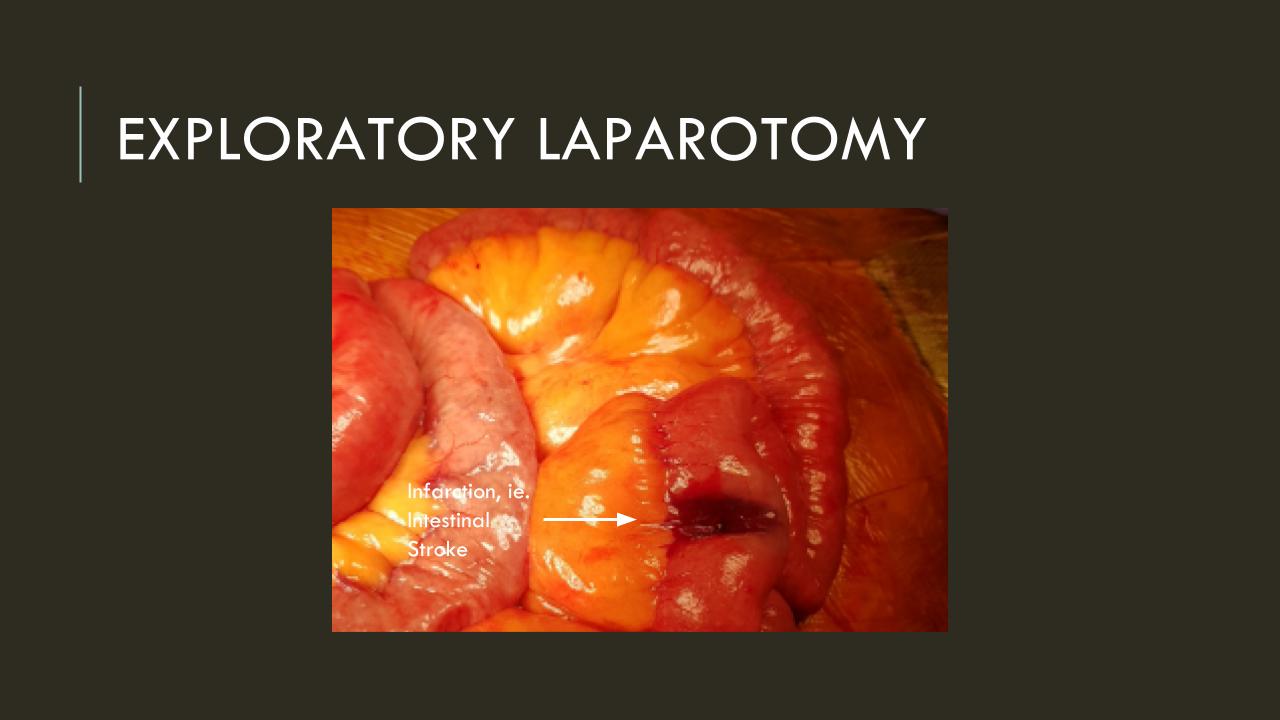
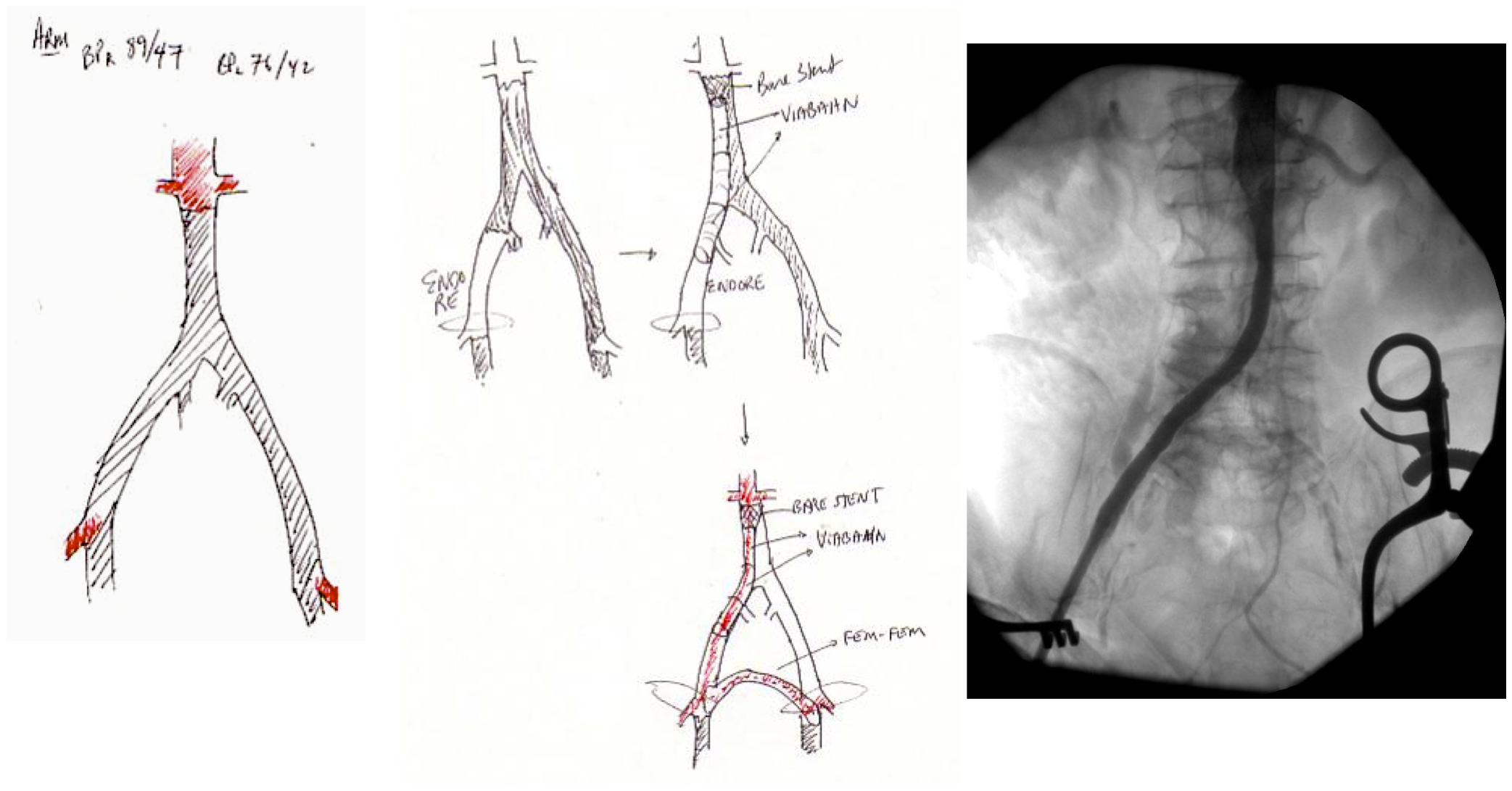
Take this patient who had previously ruptured his AAA and undergone EVAR. Several years out from his rupture, he ruptured again from a type Ib endoleak due to aneurysmal degeneration of his right common iliac artery. Per his family, he never followed up. Perhaps he assumed he was cured of his disease? Repairing this was tricky, primarily because I hopped up and down, thinking, “I could cure this!” An open revision with a bifurcated graft would eliminate the need for EVAR surveillance, avoid abdominal compartment syndrome, and the physiologic consequences of a large retroperitoneal hematoma. But who wants a laparotomy? Not this patient, who was hypovolemic shock, and whose family chose the minimally invasive option that everyone assumes is better.
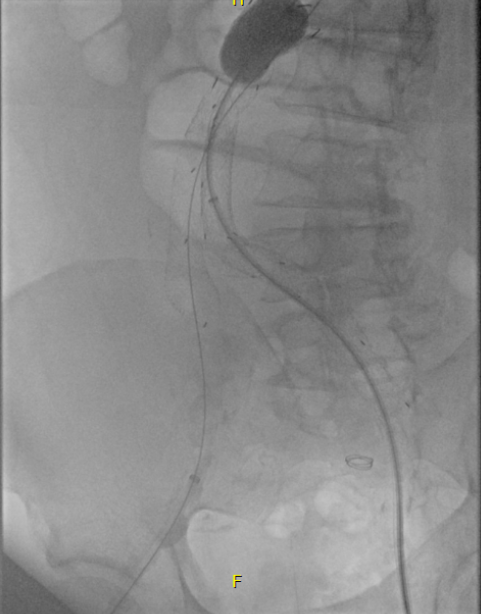
I took him to the hybrid operating room, balloon occluding to stabilize his blood pressure, embolizing the normal internal iliac artery and extending the stent graft into the external iliac artery.
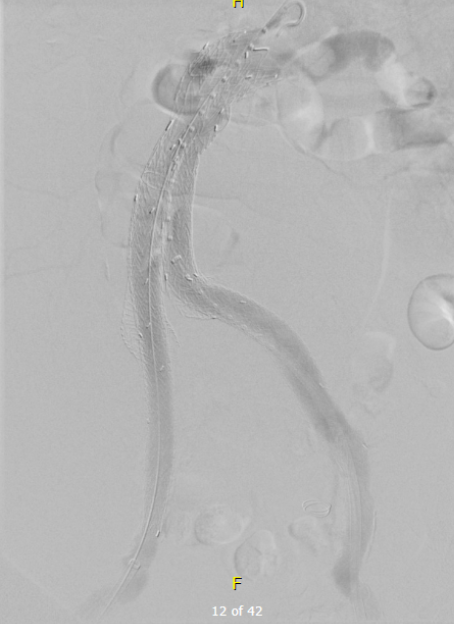
This patient stabilized and had abdominal tightness due to his large hematoma which did not need evacuation. After a stay lengthened by concern for abdominal compartment syndrome, moderate pain, fevers, and bilirubinemia (due to the hematoma), he was discharged and never showed up for followup. None of the phone numbers work. Without followup, EVAR is a menace. We will keep trying.
Regrets, I’ve Had a Few…
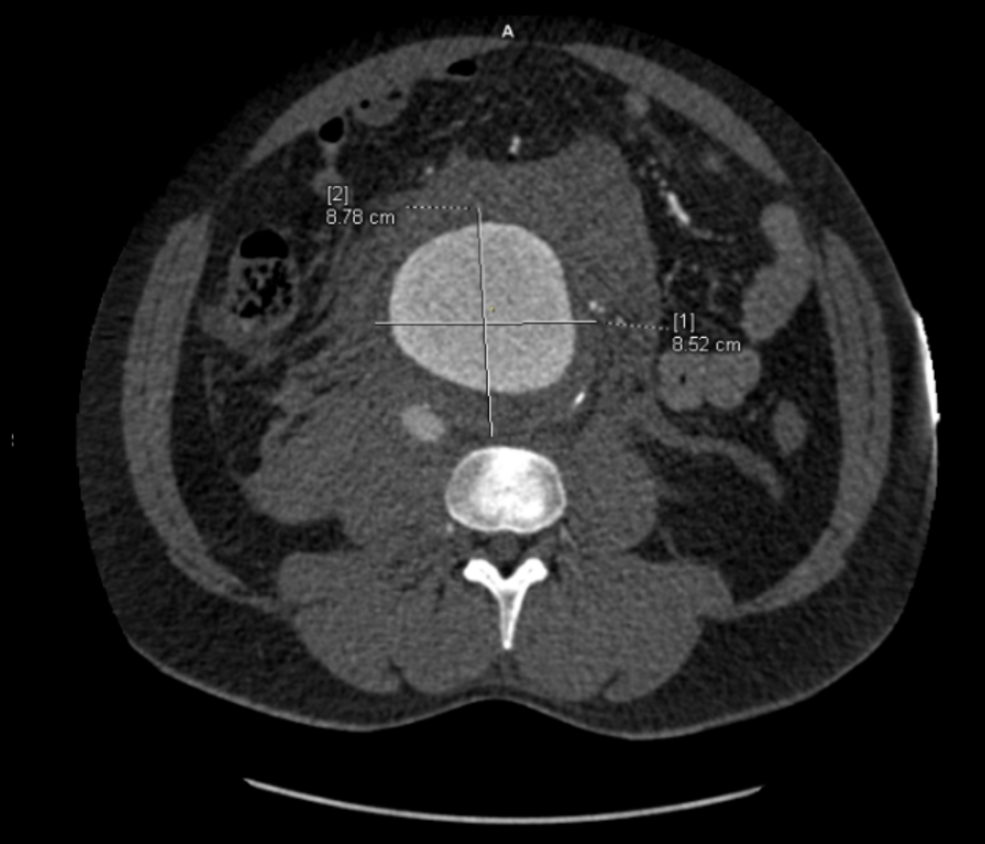
The great feature of EVAR is that the complications up-front at the time of surgery are wonderfully low. This patient pictured above here presented in middle age with a rupture into the retroperitoneum. He was unconscious and had hemorrhagic shock.
The decision to perform EVAR was made late in the transfer because I did not have the images from the transferring hospital (another subject for another blog post) so I set up for both open repair and EVAR. En route to the OR, I scanned, slowly, through the CT images sent via CD-ROM, and my internal discussion went something like this.
“He’s a 50-something smoker in shock with a contained rupture of a 8cm infrarenal AAA with a good neck. Let’s take care of this in 30 minutes with a percutaneous endovascular aneurysm repair (p-EVAR).”
“He’s a 50-something smoker in shock with a contained rupture with a good neck -let’s take care of this in 90 minutes with a tube graft, open aortic repair (OAR).”
“With p-EVAR, he’s going to have just two groin punctures and much lower complication rate, shorter length of stay, similar to lower mortality. Look -his blood pressure is 75mmHg systolic!“
“That hypotension is permissive to minimize bleeding. With OAR, he’ll avoid abdominal hypertension and complications of a giant hematoma. Because he’s young, he’ll avoid lifelong surveillance. If anyone can clamp this AAA, it’s me...”
“Pride cometh before the fall. Get this man off the table and figure out the logistics later. p-EVAR. You open him up, he’ll exsanguinate and expire before you get the clamp on.“
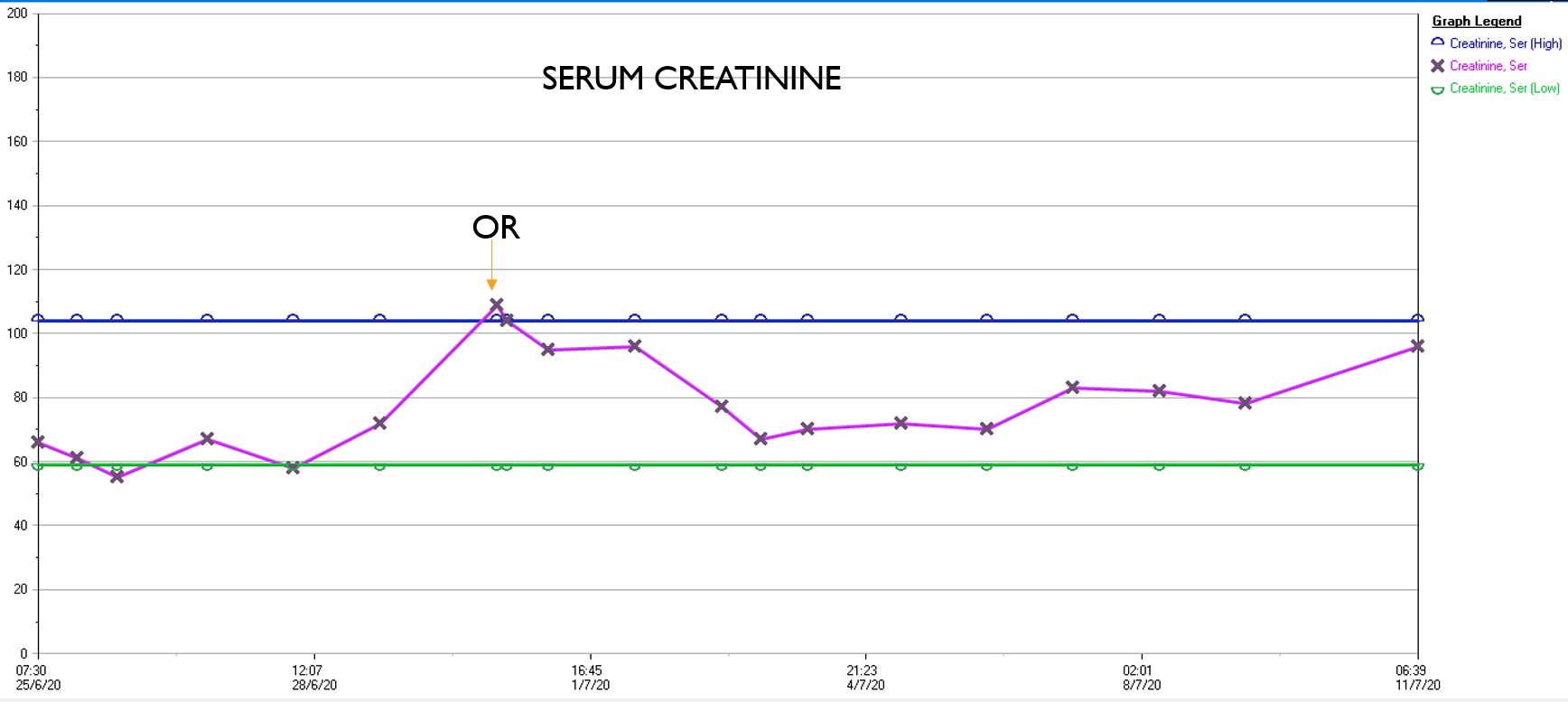
I sighed, looked up at the gathered team, and announced, “p-EVAR.” The percutaneous EVAR is something I’ve been doing since 2004, long before it was a big deal, and we were done under an hour. His blood pressure stabilized, but general surgery was consulted for his abdominal compartment syndrome. With sedation, fluids and time, his urine out put recovered but his belly remained distended and his bladder pressures which were never seriously elevated, trended down.
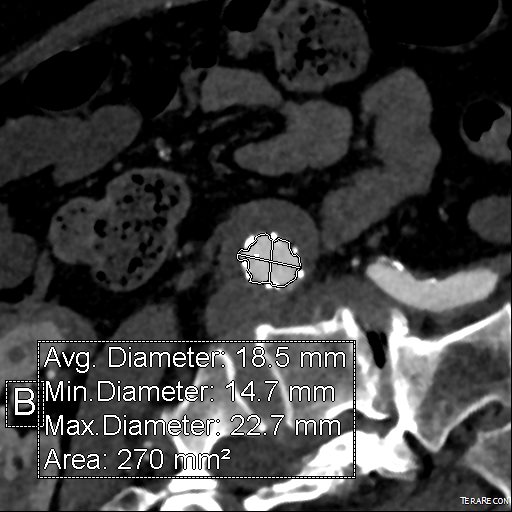
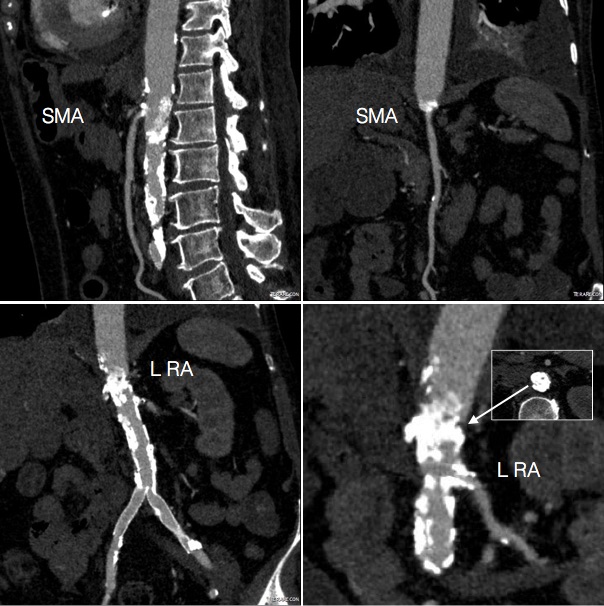
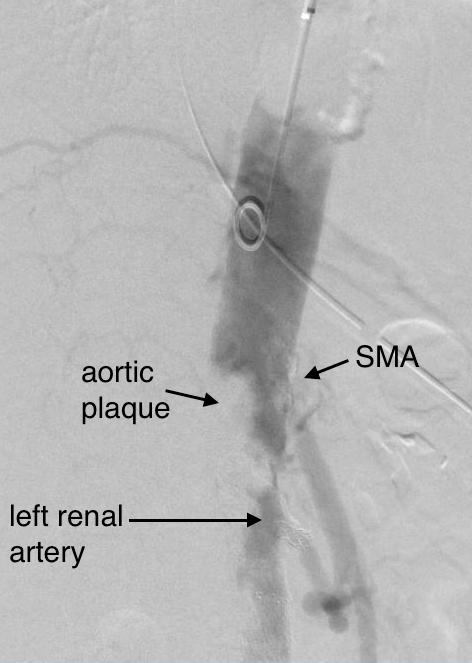
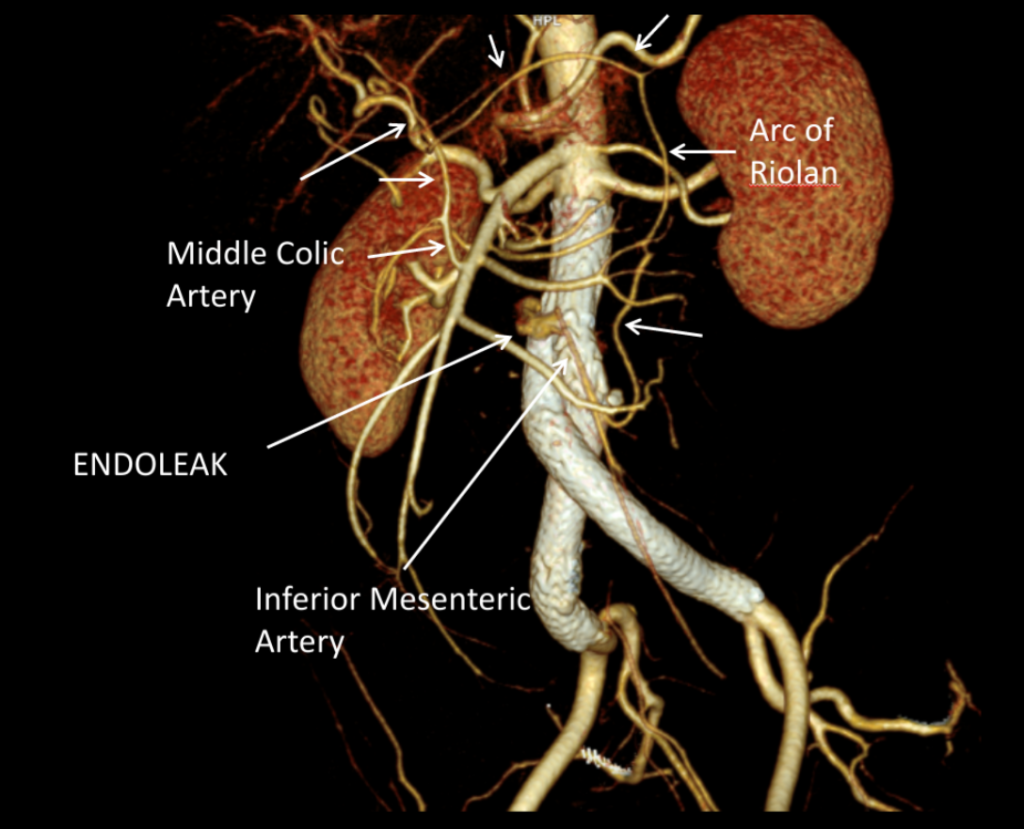
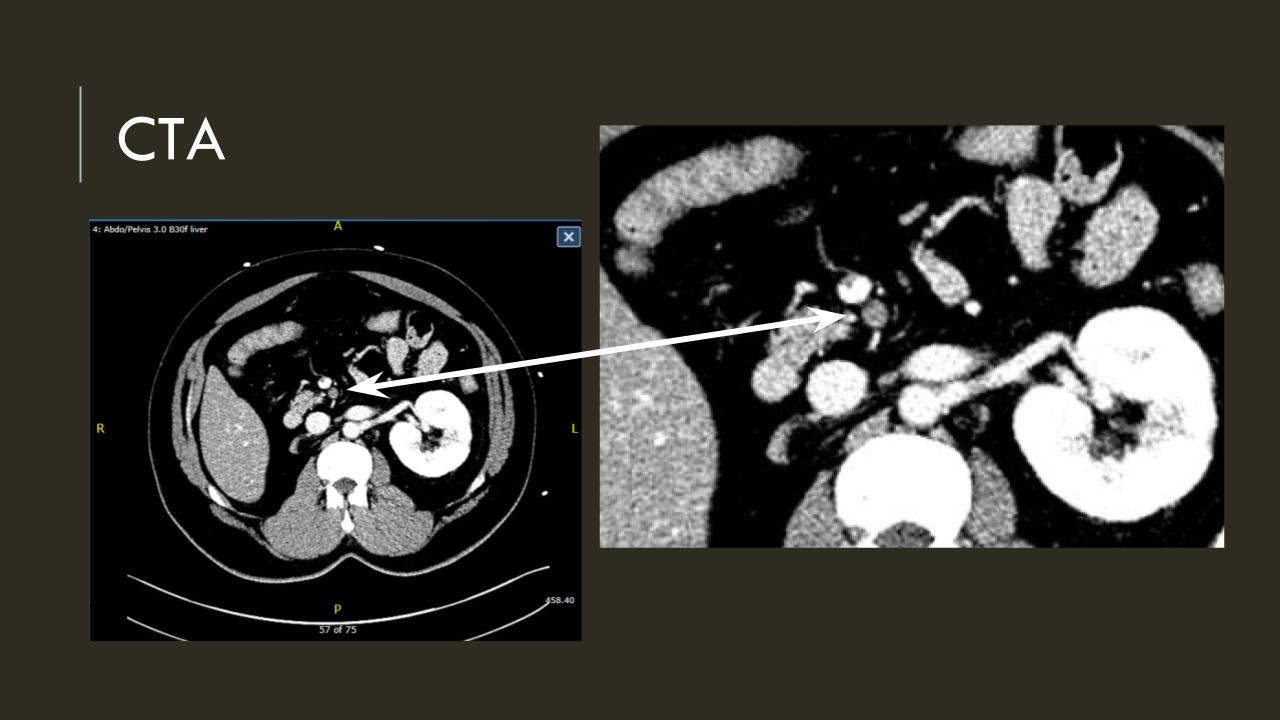
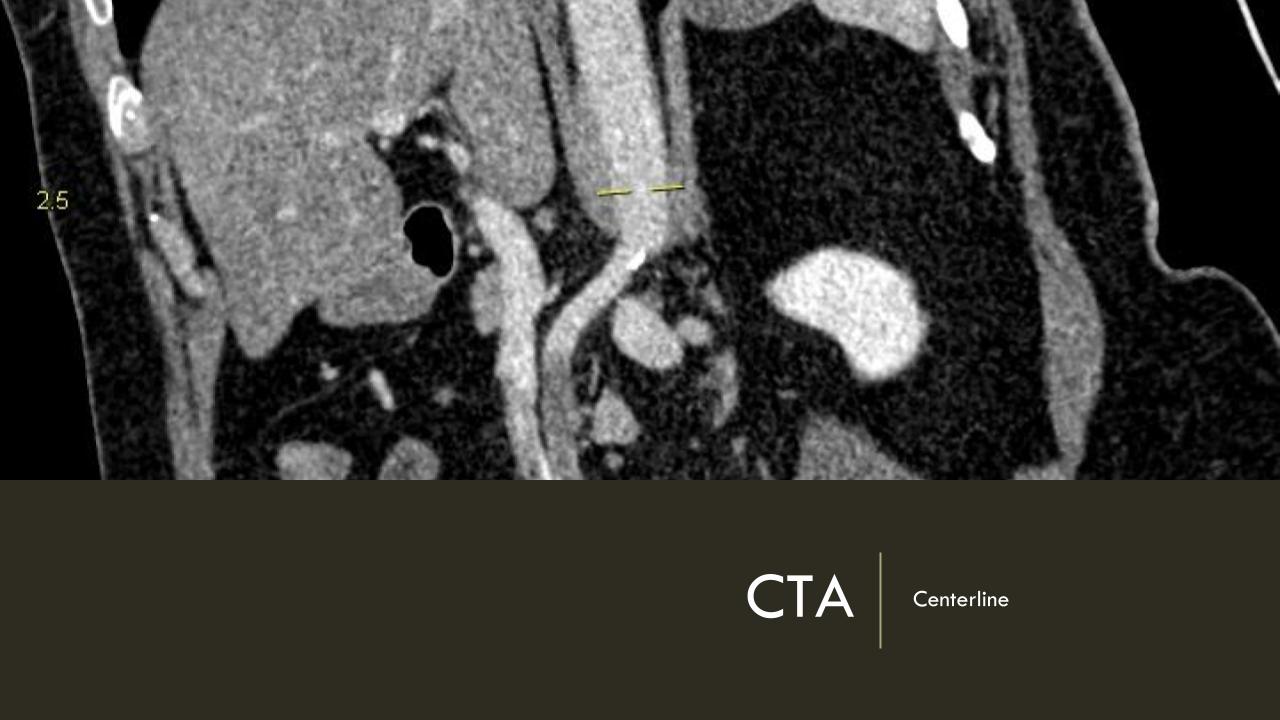
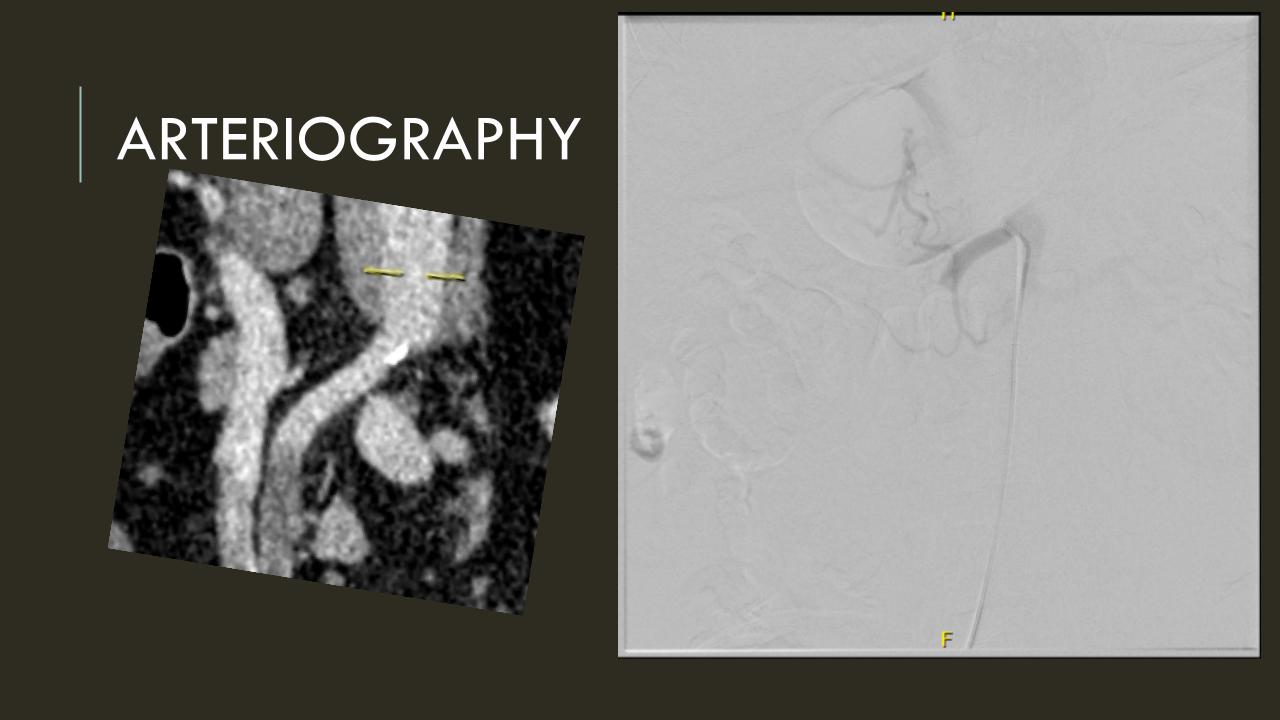
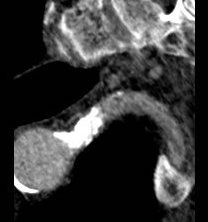
It was made known to me that the patient had very limited insurance making followup surveillance challenging. Due to his coverage, he had to have his imaging done at designated hospitals, so I wrote a detailed note -basically the timings of his followup CT scan, and asked that the reports should be sent to me. I ordered a CTA prior to discharge which showed a type II endoleak adjacent to the graft and connected to both lumbar and inferior mesenteric arteries (first image above). After some thinking, I took the patient for an aortogram, accessed the IMA via the SMA and coiled into his AAA sac and the IMA.
It was only a few weeks ago one of my colleagues across town contacted me that the patient had been admitted with abdominal pain, a CT showing a type II endoleak from his lumbars, but a smaller AAA sac than his pre-repair size of 8.5cm. The patient is seeking to repatriate, and I doubt he would be able to get adequate followup in his home country without paying in cash. Happy that the patient survived his rupture, I still have persistent regrets at not getting him through an open repair, which I am sure he would have done fine with… Or maybe not.
Je Ne Regret Rien…
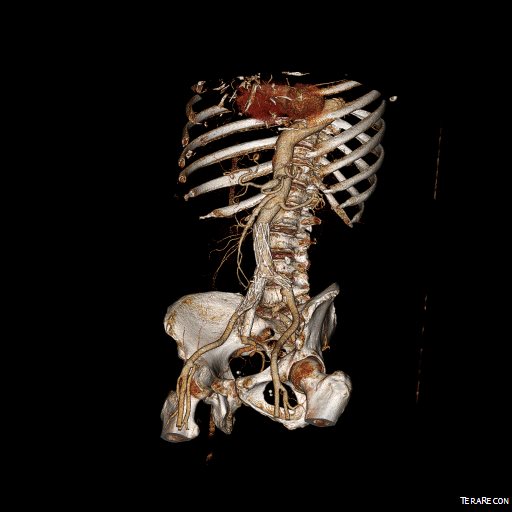
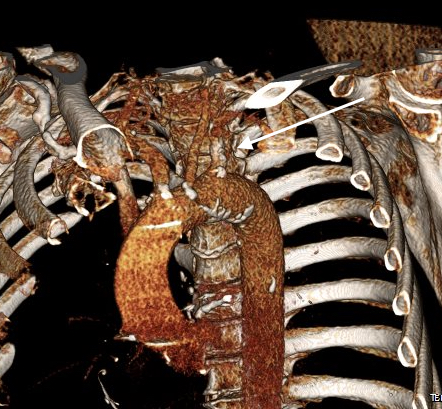
Recently I admitted a patient, in his 70’s, with a symptomatic 6.5cm infrarenal AAA with bilateral common iliac artery aneurysms, the right being 25mm, the left over 30mm. Because of the pandemic, he was stuck here, seeking to repatriate. Over ten years before, he had a segment of descending thoracic aorta repaired for a traumatic tear -probably one of the last before the wide adoption of thoracic stent grafts which work great by the way.
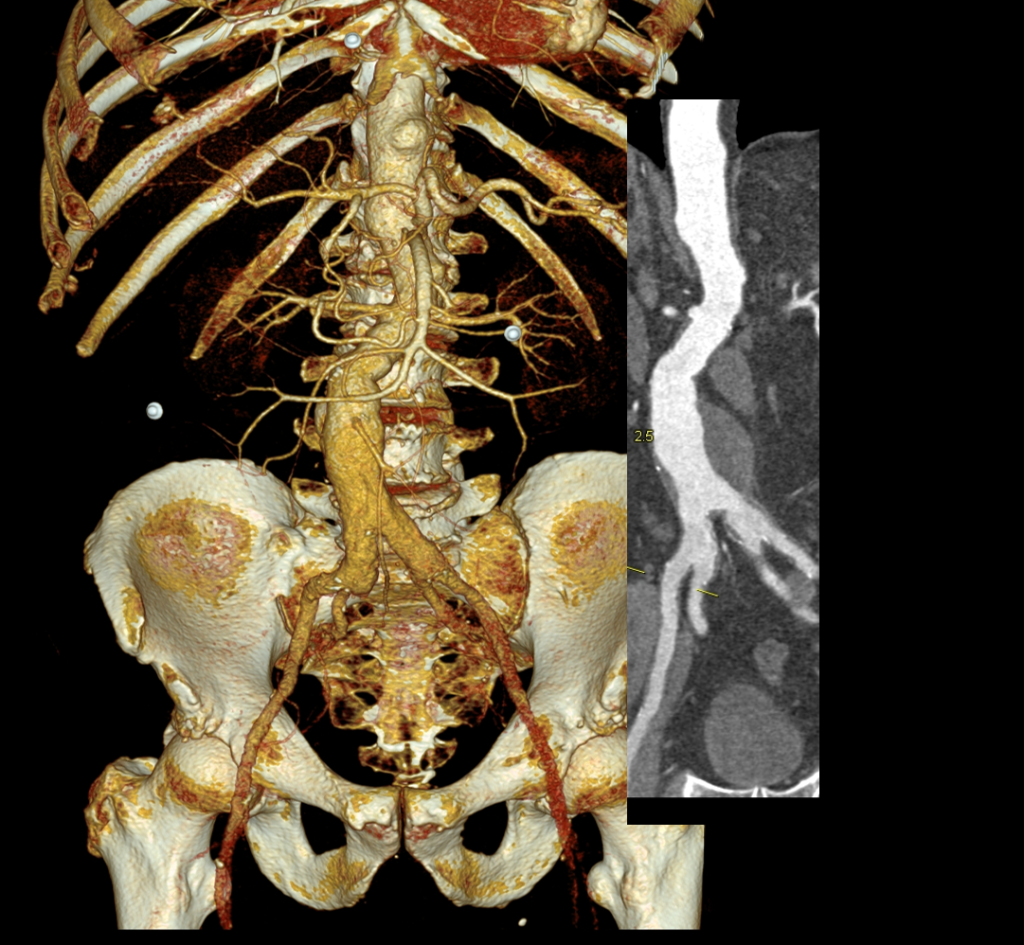
Cardiac risk evaluation revealed an ejection fraction of 35% with reversible ischemia on nuclear stress test. CTA of the coronaries revealed triple vessel coronary artery disease corroborated by catheterization. Off-pump CABG was planned which would eliminate the effects of cardiopulmonary bypass.
Preparations were made for EVAR with IBE of the left iliac aneurysm as a contingency, but there was no question that if the patient recovered well from his CABG, he would undergo open repair. This was because bell-bottoming or IBE must have regular coordinated surveillance which was not going to be easy with the patient leaving for another country in the middle of a pandemic. It is difficult to get followup to happen in normal circumstances (see above cases). I expressed my opinion to the patient and family and we agreed to see how the patient responded to off-pump CABG.
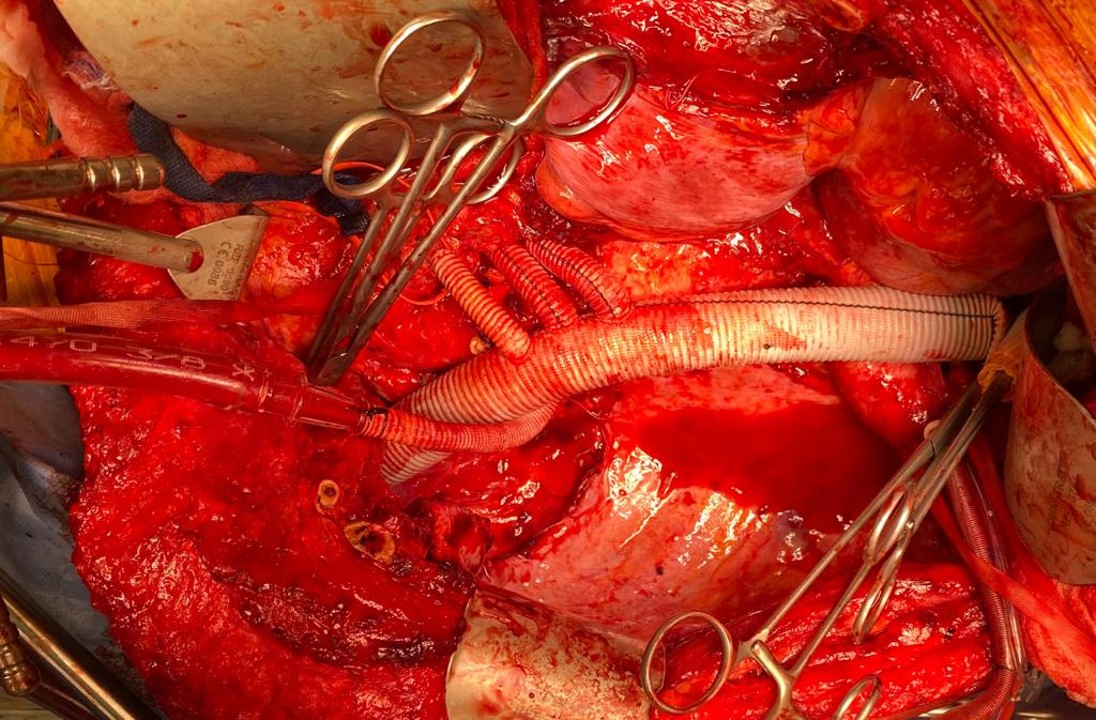
He underwent off-pump CABG with three vessels revascularized. He was extubated POD#1 and mobilized. By POD #4, he was on a regular patient floor, being co-managed by hospital medicine who takes care of all of our patients. The patient expressed readiness for the next operation. His kidney function remained normal. He was transfused 2 units of PRBC to bring his hematocrit to 30%. He was taken off Plavix, but kept on aspirin. On POD#6, he was taken back to the operating room for open aortic bypass. This would not have been possible without close coordination of cardiology, cardiac surgery, and vascular surgery. Choosing off-pump CABG was a critical element in being able to proceed with open aortic surgery.
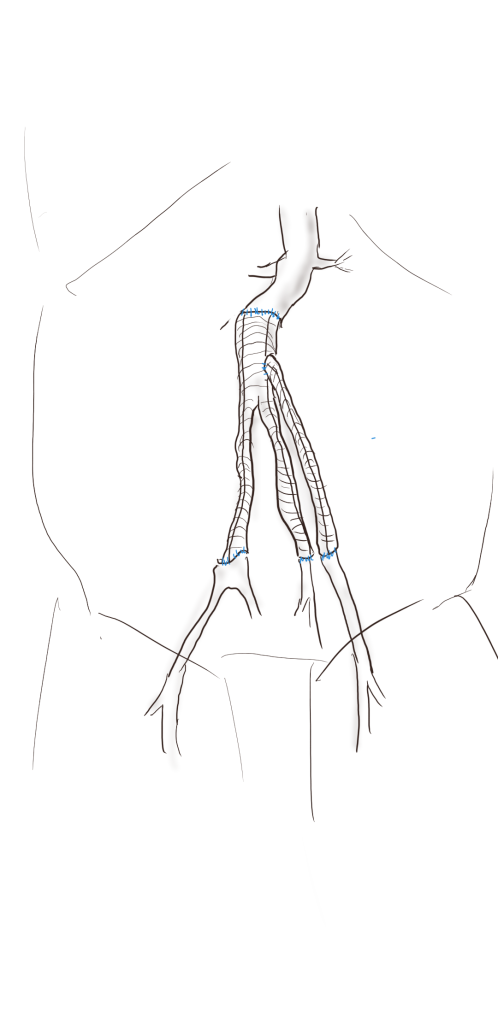
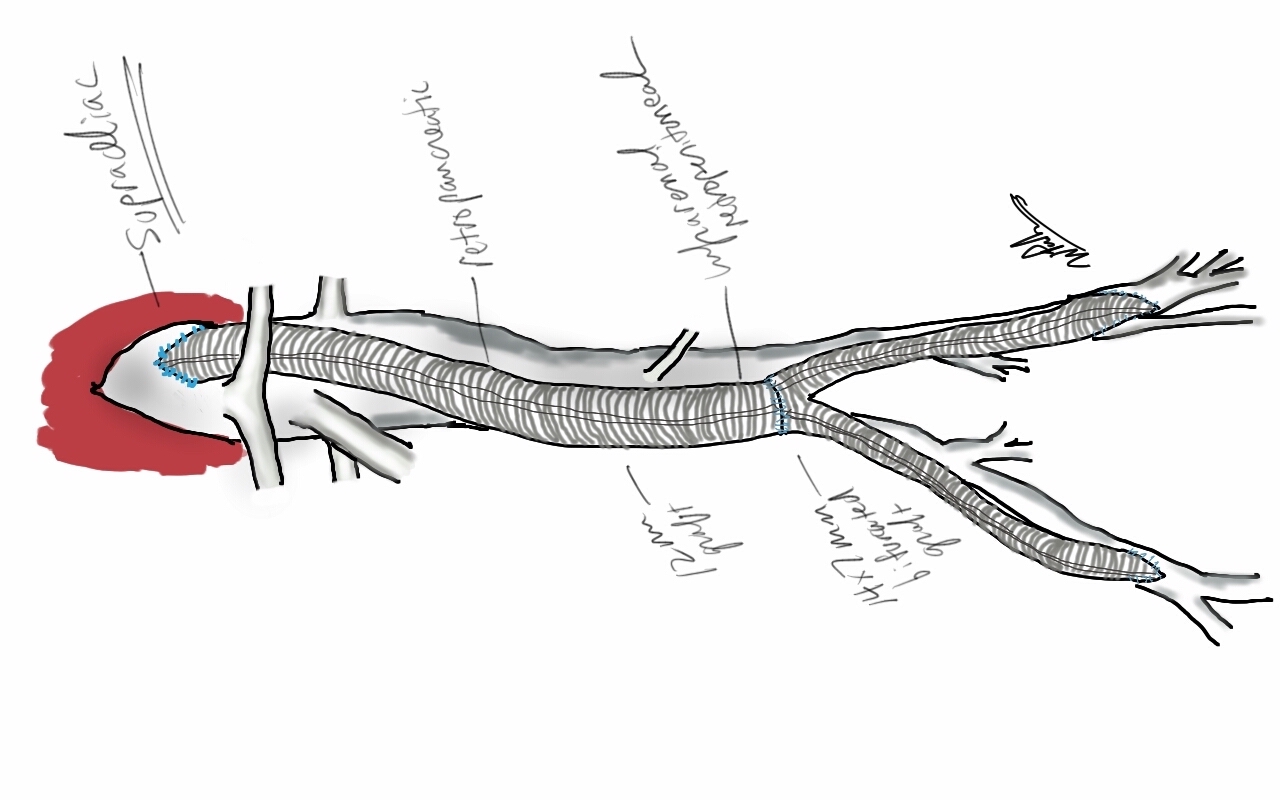
I do several things to decrease the physiologic impact of the operation. First is keeping all the viscera retracted under the skin. This simple move has the effect of decreasing the rate of intestinal paralysis and amount of fluid shifts that occur postop, akin to going retroperitoneal. This decreases the space you potentially have if you use standard clamps, but I use the Cherry Supraceliac clamp (image), DeBakey Sidewinder (transverse), or just a Satinsky clamp oriented transversely. This minimizes the occupation of volume over the anastomosis which always happens with standard aortic cross clamps. The anastomosis is easier without the clamp taking up valuable space.
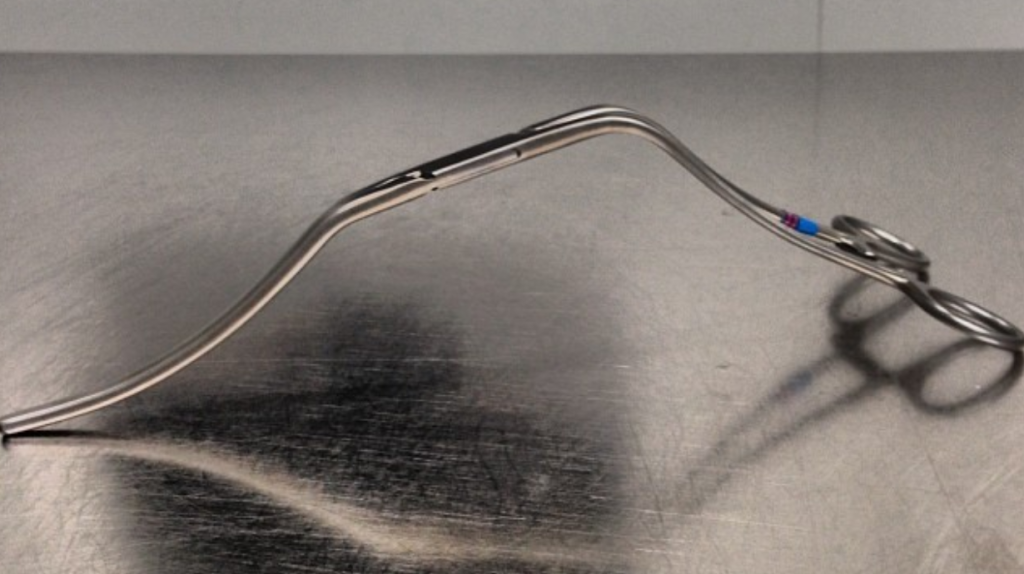
The iliacs are always clamped with Wylie Hypogastric clamps, again, with the principle of eliminating clamp overhang. Suturing is done with 4-0 Prolene on SH needles -this is plenty (link). The proximal anastomosis wants to bleed, and sewing to a fully cut ring of aorta ensures good posterior bites but also allows for sliding a band of graft down over the anastomosis (Dan Clair calls this a gusset) which works well at creating a hemostatic proximal anastomosis rapidly-trust me, getting this done well is the key step of the operation. Before closing, I infiltrate the rectus sheath and preperitoneum bilaterally with local anesthesia -lidocaine 1% with epinephrine 1:1 with bupivicaine 0.5%. The skin is closed with absorbable dermal sutures because staples create as many problems as they solve. The patient had cell salvage through the case and no extra units of transfused blood.
The patient was extubated that night and started on clear liquid diet. The next day his lines were removed and he was moved to the floor and started on regular diet when he expressed hunger. On POD#2, he was pacing floor, asking when he could be discharged.
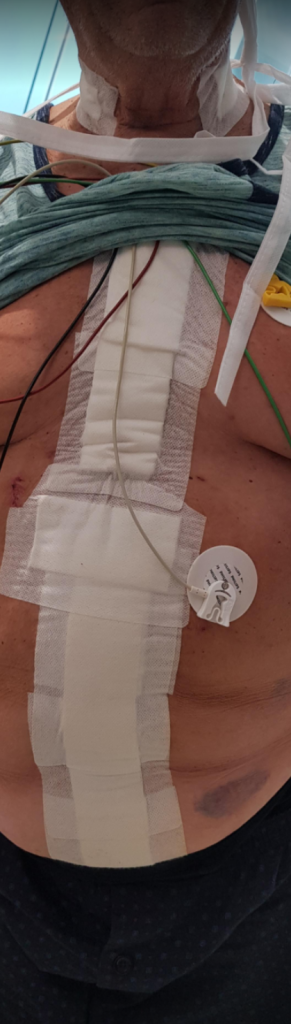
As he was eating, walking, talking, breathing, evacuating bowel and urine, and pain free (well controlled), I saw no reason to keep him beyond POD #3 AAA/#9 CABG. I have kept in touch with him and his family and he is doing well and has given permission for this posting.
Satisfaction
This final case has confirmed several of my beliefs. First, calling something high risk can drive one to make bad choices and in fact endanger patients. This last patient would qualify as high risk on any international criteria, and you would not be wrong in quoting upwards of 30% major morbidity and mortality for cardiac revascularization and AAA repair, but you would also be tying your hands from offering the best solution for this man who fortunately was able to undergo two prodigious operations. He will not require much in the way of followup. Coronary revascularization with arterial conduit and open aortic grafting frees him from the need for close followup and reassures us that his repair is durable.
Second, calling something advanced and minimally invasive gives one cachet in the marketplace but forgoes careful discussion and consideration of what is being abandoned. The first two patients survived their ruptures but now face the consequences of having stent grafts. It is a shame when podium speakers at international symposia declare surgery to represent failure because this affects training by encouraging abandonment of hard to acquire skills. It seeps into patient perceptions and expectations. I hope that a balanced approach prevails. You have to be capable of both open and endovascular approaches to be able to offer the best treatment for a particular patient.
Finally, these old operations are cost efficient and there is a lot of room to improve these procedures with new perspectives, techniques, and data. I don’t operate the same way I trained, and it is only through continued application of operations that improvements can come about. As budgets tighten and economies are stressed, cost efficacy will rein in much of the interest and demand in new stuff unless it adds value. That said, I am grateful to our stent graft representatives who have worked to get us bell bottoms and IBE’s for when they will be needed. These grafts will be used when the time is right.

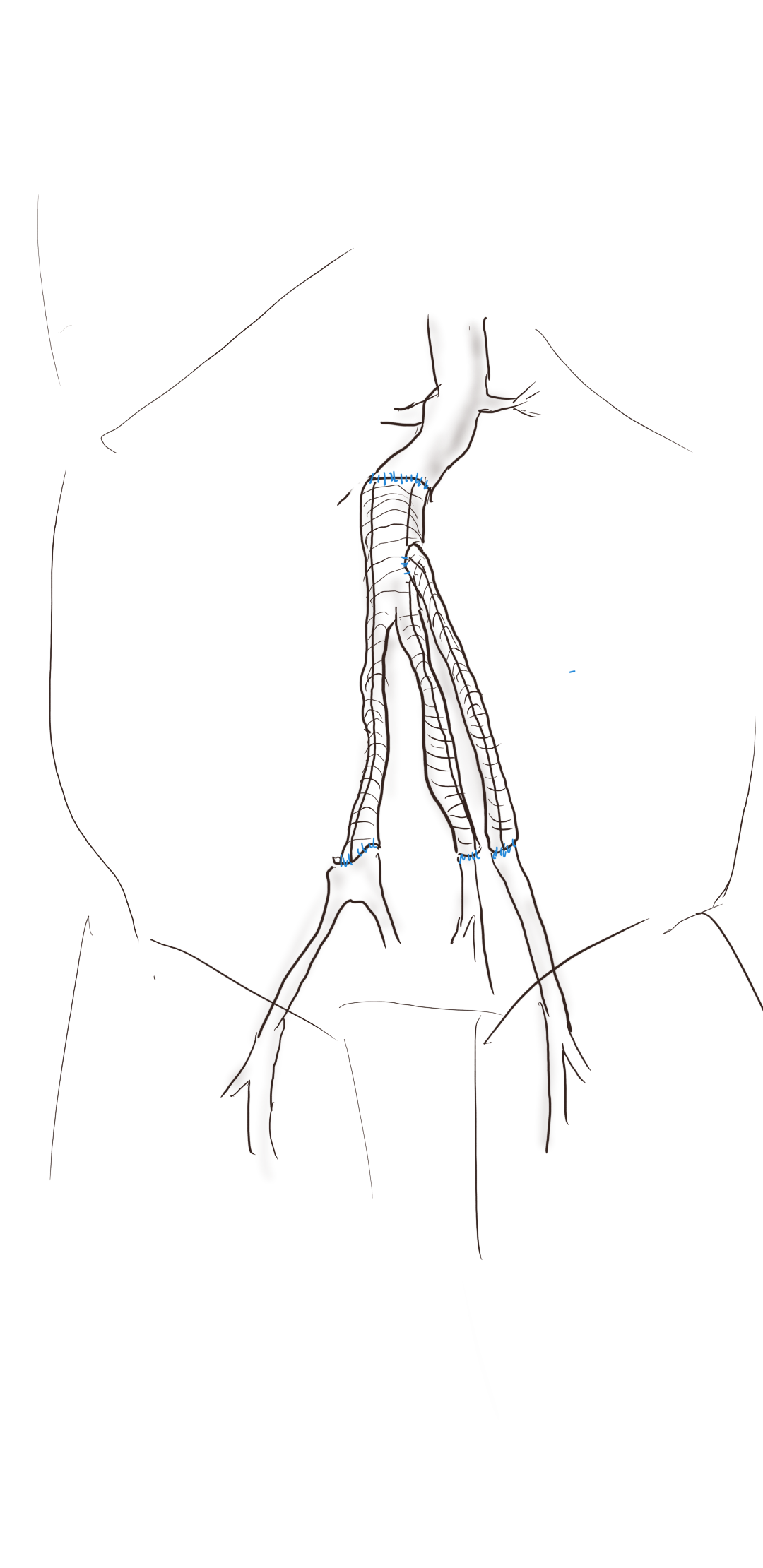




























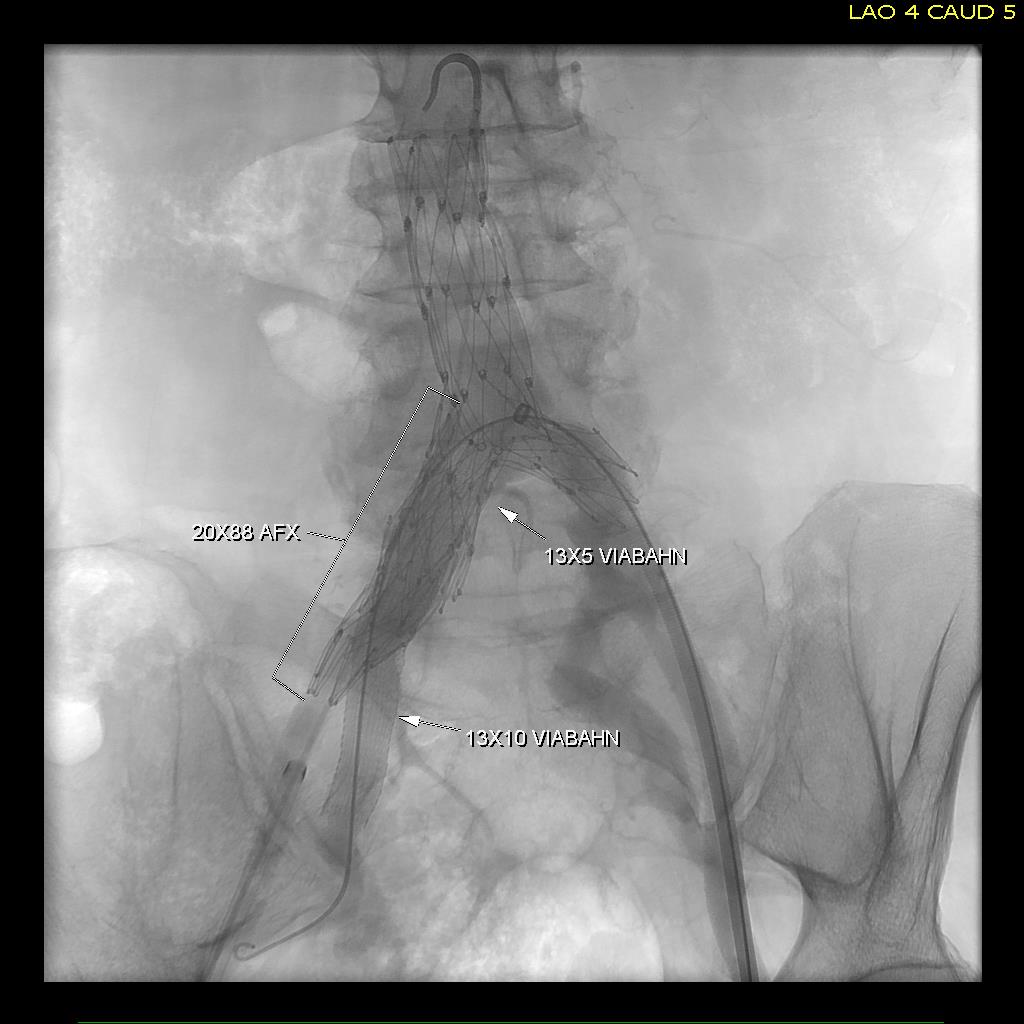
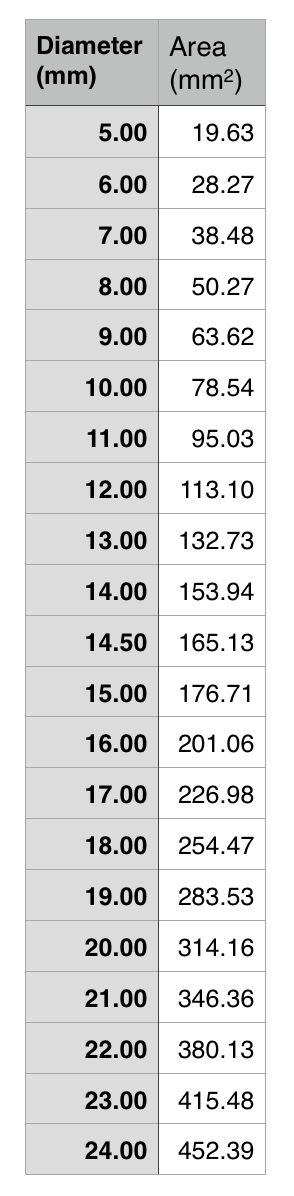 The inflow graft area is taken from its measured diameter. Then usually one or the other artery has an obligate size -a size the graft has to be while the other has more “wiggle room.” The other thing that comes from experience is that the AFX graft’s iliac limb extension don’t get the B-infolding that can affect an oversized stent graft placed in a small artery and it accomodates a neighbor well.
The inflow graft area is taken from its measured diameter. Then usually one or the other artery has an obligate size -a size the graft has to be while the other has more “wiggle room.” The other thing that comes from experience is that the AFX graft’s iliac limb extension don’t get the B-infolding that can affect an oversized stent graft placed in a small artery and it accomodates a neighbor well.