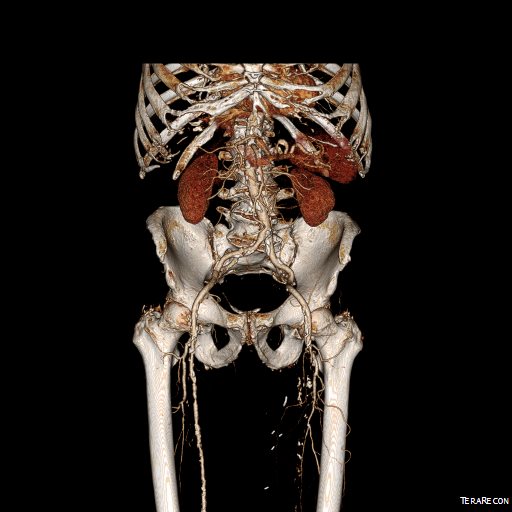
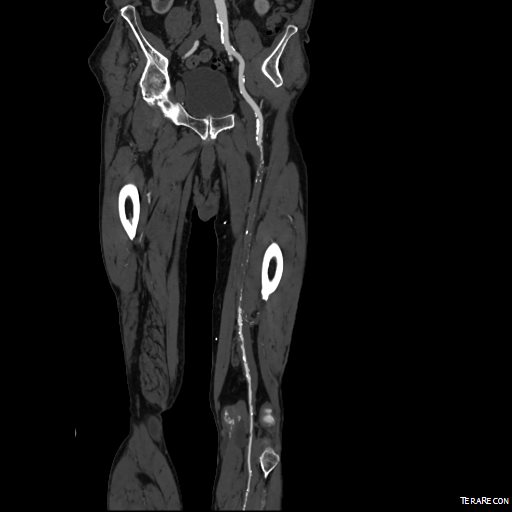
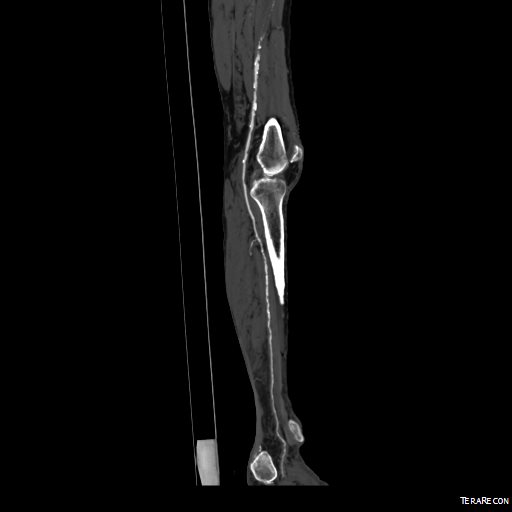
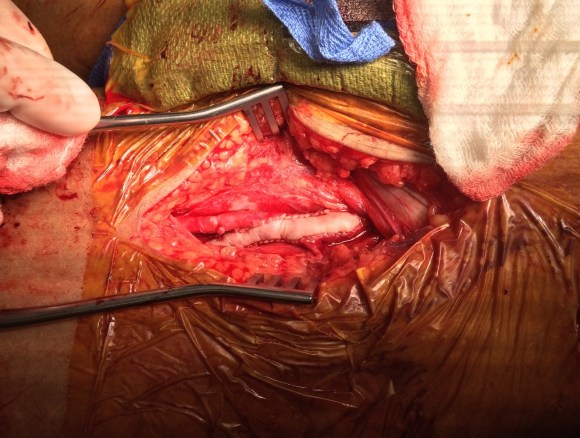
Something that I recently promised Dr. James S.T. Yao, I will be working to publish on my stent removal and extended remote endarterectomy cases and techniques. Meanwhile, here is a talk.
Category: remote endarterectromy
Leriche Syndrome -one of those disease names that adds to our work in a way that an ICD codes and even the “aortoiliac occlusive disease” fails to describe. When I hear someone described as having Leriche Syndrome, I think about a sad, chain smoking man, unmanned, complaining of legs that cramp up at fifty feet, pulseless.
The CT scan will occasionally show an aorta ringed by calcium in the usual places that are targetrs for clamping below and above the level of the renal arteries. Even without the circumferential calcium, a bulky posterior plaque presages the inability to safely clamp the aorta. Woe to the surgeon who blithely clamps a calcified lesion and finds that the rocky fragments have broken the aorta underneath the clamp! The first way to deal with this is to look for ways not to clamp the aorta, by planning an endovascular procedure, but circumstances may necessitate the need to control the aorta despite the unclampability.
The traditional methods of avoiding clamping the calcifed peri-renal aorta are extra-anatomic bypasses including femorofemoral bypass and axillo-femoral bypass. I propose these following options for the consideration when the patient needs a more durable solution while avoiding a heavily diseased aorta.
Not Clamping I:

EndoABF does work to avoid clamping -these are common femoral endarterectomies supplemented by stenting of the aortoiliac segment, including in those with appropriate anatomy, a bifurcated aortic stent graft. This is often not possible to treat both sides, but one side is usually more accessible. Often, people will compromise and perform an AUI-FEM-FEM, but I have found the fem-fem bypass to be the weak link, as you are drawing flow for the lower half of the body through a diseased external iliac artery. The orientation of the proximal anastomosis is unfavorable and in the instance of highly laminar or organized flow, the bypass is vulnerable to competitive flow on the target leg, leading to thrombosis.

The femorofemoral bypass is the option of patients whose options have largely run out. It is made worse when fed by an axillofemoral bypass. Sometimes, you have no choice, but in the more elective circumstance, you do.
Not Clamping II:
The second method is performing a aorto-uni-iliac stent graft into a conduit sewn end to end to the common iliac aftery, oversewing the distal iliac bifurcation.
The conduit is 12mm in diameter, the key is to deliver the stent graft across the anastomosis, sealing it. The conduit is then sewn to the side of a fem-fem bypass in the pelvis, maintaining antegrade flow to both legs. The other option is to sew the conduit to a 14×7 bifurcated graft. Illustrated above is this 12mm conduit sewn end to end to the diseased common iliac artery with wire access into the aorta and a aorto-uni-iliac device. Typically, a small AUI converter (Cook, Medtronic) can be used, but the aorta is often too small even for a 24mm device, and an iliac limb with a generous sized docking segment (Gore) ending in a 12mm diameter fits nicely. Below is a CTA from such a case, where the stent graft is deployed across the anastomosis, sealing it off from anastomotic leaks (exoleaks).

Not Clamping III:
Often, the infrarenal aorta is soft anteriorly and affected only by posterior plaque at the level of the renal arteries. While a clamp is still not entirely safe (I prefer clamping transversely in the same orientation as the plaque with a DeBakey sidewinder clamp), a balloon is possible. I do this by nicking the aorta -simple application of a finger is sufficient to stop the bleeding if you have ever poked the ascending aorta to place cardioplegia line.

A Foley catheter is inserted and inflated. The Foley’s are more durable and resist puncture better than a large Fogarty. This is usually sufficient for control, although supraceliac control prior to doing this step is advised. The aorta can be endarterectomized and sewn to the graft quite easily with this non-clamp. 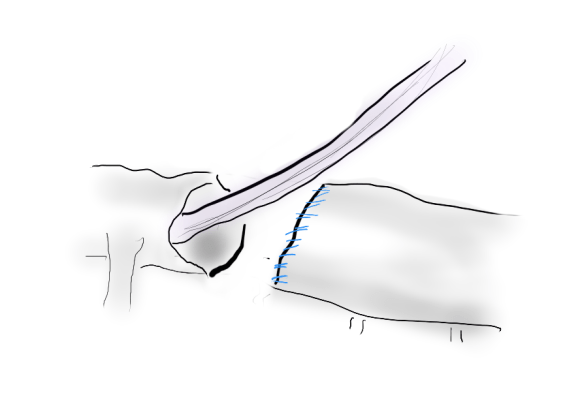
This has worked well, Although pictured above with an end-to end anastomosis planned, it works just as well end-to-side. I actually prefer end to side whenever possible because it preserves the occluded native vessels for future intervention in line.
The Non-Thoraco-Bi-Femoral Bypass
The typical board answer for the non-clampable aorta is taking the inflow from the thoracic aorta or from the axillary artery -neither of which are good options. The first because the patient is positioned in right lateral decubitus and tunneling is not trivial. The second because of long term durability. The supraceliac aorta, technically it is the thoracic aorta, is often spared from severe plaque and clampable. Retropancreatic tunelling is straightforward, and a 12 or 14mm straight graft can be tunelled in this fashion from the lesser sac to the infrarenal retroperitoneum. It then sewn to the supraceliac aorta and then anastomosed to a 12x6mm or 14x7mm bifurcated aorto-bifemoral bypass, of which limbs are tunneled to the groins.
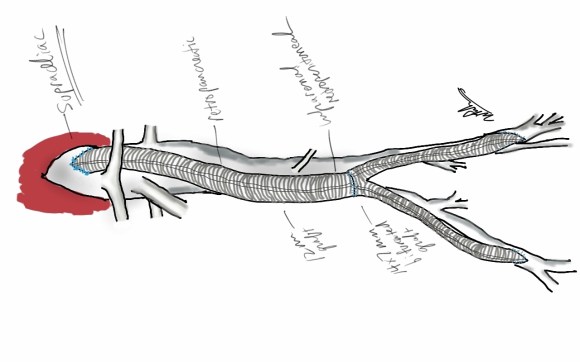
This worked very well recently, allowing a middle aged patient with severe medical problems, occluded aorta and iliac arteries, with critical limb ischemia, survive with minimal blood loss and home under 5 days. It delivers excellent flow to both legs in an antegrade fashion. Dr. Lew Schwartz gave me a list of references showing that this is not novel, but represents a rediscovery as the papers were published in the 80’s [reference], and buttresses the principle that innovations in open vascular surgery are exceedingly rare, largely because we have been preceeded by smart people.
Conclusion: All of these come about through application of some common sense and surgical principles. The most important this is that the aorta is the best inflow source and reconstructing it with the normal forward flow of down each leg and not reversing directions as in a fem-fem bypass gives each of these options a hemodynamic advantage.
References for Supraceliac Aorta to Lower Extremity Bypass
- Surgery [Surgery] 1987 Mar; Vol. 101 (3), pp. 323-8.
- Annals of Vascular Surgery 1986 1(1):30-35
- Texas Heart Institute Journal [Tex Heart Inst J] 1984 Jun; Vol. 11 (2), pp. 188-91.
- Annals of Thoracic Surgery 1977 23(5):442-448

The principles of salvage are in rescuing valuable undamaged goods in the setting of catastrophe. This guided me when a patient was flown in from an outside institution to our ICU with a saline soaked OR towel in his right groin -he had had an aorto-bifemoral bypass for aorto-iliac occlusive disease a year prior, but had never properly healed his right groin wound which continued to drain despite VAC therapy and wound care. On revealing his groin, this is what I saw:

A CT scan was sent with the patient but has been lost to time, and it showed a patent aorto-bifemoral bypass send flow around an occluded distal aorta and iliac arteries. The graft did not have a telltale haze around it nor a dark halo of fluid which signaled to me that it was likely well incorporated and only sick in the exposed part. The patient was not septic, but had grown MRSA from the wound which was granulating from the extensive wound care that had been delivered.
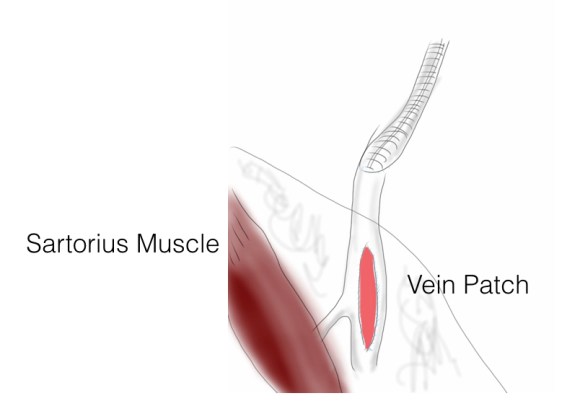
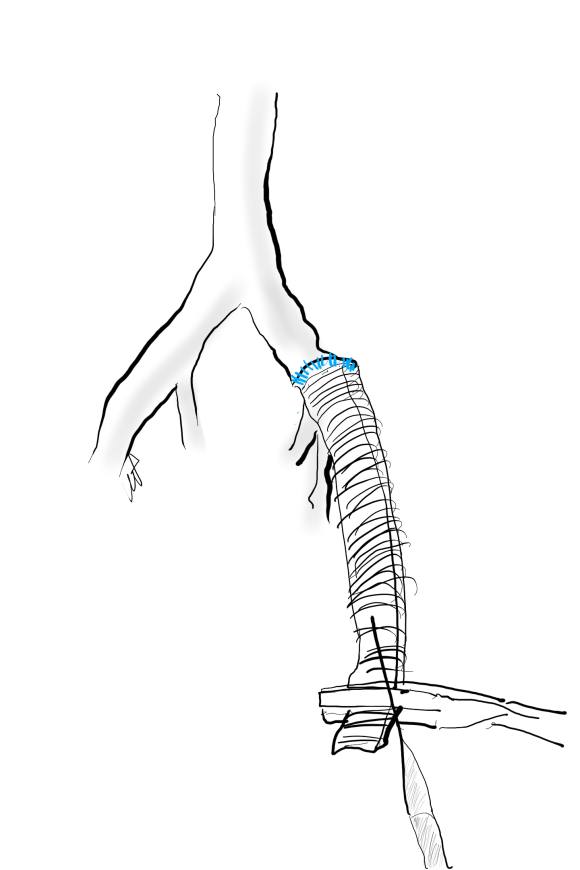
I felt that it would be possible to move his anastomosis point more proximally on the external iliac in a sterile field (figure above), and then close, then endarterectomize the occluded external iliac artery after removing the distal graft, then after vein patching, cover the repair with a sartorius muscle flap. It would salvage the remaining graft and avoid a much larger, more intense operation which was plan B. To prepare for that, I had his deep femoral veins mapped.
The patient was prepped and draped, the groin was excluded by placing a lap pad soaked in peroxide/betadine/saline solution (recipe for “brown bubbly” liter saline, a bottle of peroxide, a bottle of betadine), and covering with an adesive drape. The rest of the abdomen was then draped with a second large adhesive drape. A retroperitoneal (transplant-type) right lower quadrant incision was made (below) and the external iliac artery and graft were exposed. As predicted on CT, the graft was well incorporated.

The external iliac artery was opened and focally endarterectomized of occlusive plaque (image below). The adventitia had good quality despite the longstanding occlusion.

The graft was mobilized and transected and anastomosed end to side to this segment of artery (below). Dissecting was made difficult by how well incorporated it was.
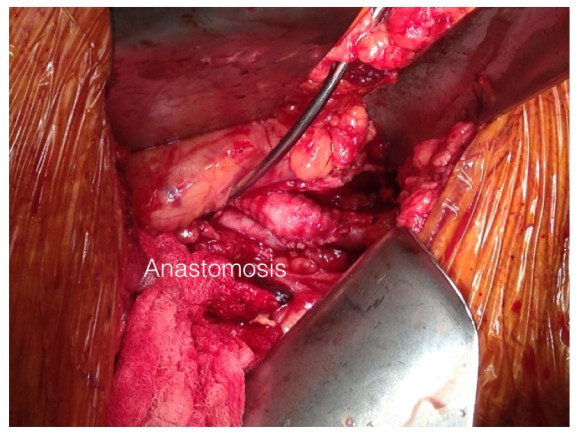
The wound was irrigated (with brown bubbly) and closed, dressed, and sealed over with the adhesive drape. The groin wound was then revealed and the graft pulled out (below).
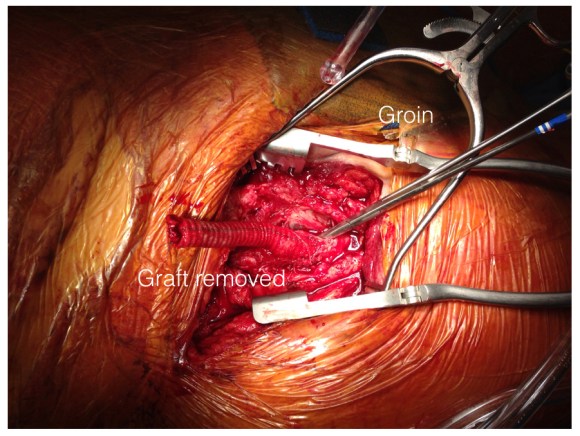
Remote endarterectomy using a Vollmer ring was used -in this case I didn’t use fluoroscopy given the short distance to the terminus of the plaque which i had mobilized in the pelvis.

The plaque came out easily and was not infected appearing. It is shown below ex vivo.

A segment of saphenous vein was harvested from the patient medially and the arteriotomy was patched. The sartorius muscle was mobilized and applied as a flap over this. The wound was irrigated with brown bubbly and packed open with the intention of VAC application.
The patient healed very rapidly and remains infection free. I had used this approach on several occasions in the past and twice more recently. It truly is salvage as it preserves the uninfected graft while never exposing it to the infection in the process of operating. It avoids having to remove the whole graft which then damages the left side -I have seen other surgeons take this approach elsewhere taking a all-or-nothing approach to graft infection to considerable morbidity to the patient. It avoids having to harvest deep femoral vein -another large operation to which the body responds truculently. The patient recently came by for his 4 year followup, still smoking, but legs preserved.
Followup At 4 Years
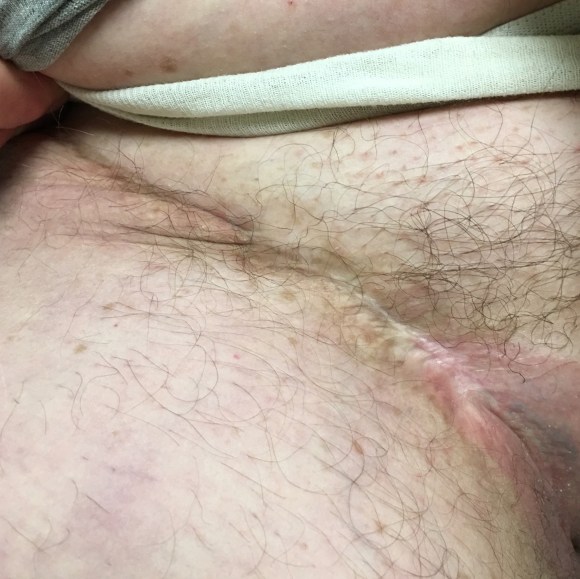
The patient came back in followup -it has been 4 years since his infection was repaired. He was complaining of short distance claudication. His wound healed well and remains closed. CTA shows along with his short segment SFA occlusion which we will treat, a widely patent R. EIA (below).

The remote endarterectomy of the external iliac artery remains patent. Compare this to the preop CTA which I found and wasn’t available when I posted this case originally:
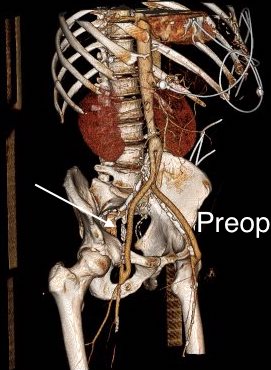
The chronically occluded EIA can be readily seen. The artery shown in the current CTA is that recanalized artery.
The patient is an 70 year old man referred for evaluation of claudication that occurred at under a block of walking. He reported no rest pain or tissue loss. He smoked heavily up to a pack a day, with congestive heart failure with an ejection fraction of 40%, prior history of myocardial infarction treated with PTCA, and pacemaker, and moderate dyspnea on exertion.
On examination, patient had a flaccid abdomen through which the AAA could be palpated, and he had no palpable femoral artery pulse bilaterally, nor anything below. He had a cardiac murmur and moderate bilateral edema. Preoperative risk evaluation placed him in the high risk category because of his heart failure, coronary artery disease, and his mild to moderate pulmonary disease.
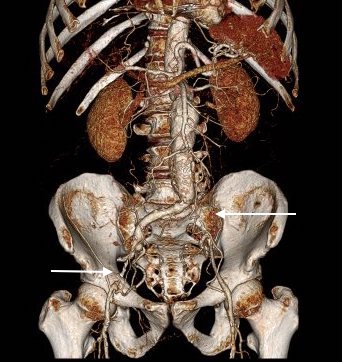
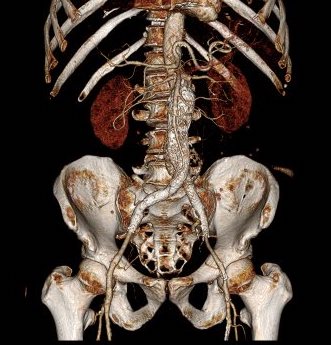
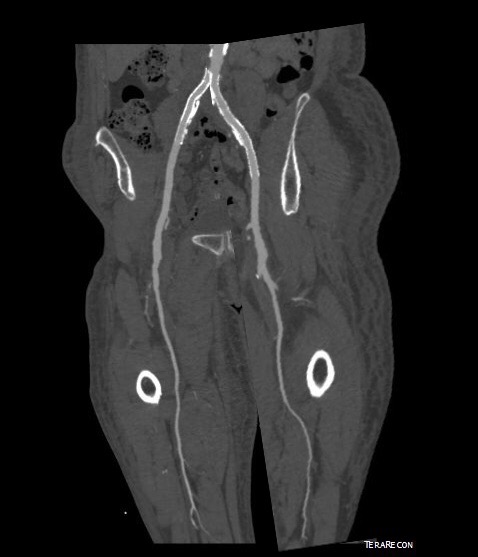
CTA (pictured above and below) showed a 5.1cm infrarenal AAA with an hourglass shaped neck with moderate atherosclerosis in the neck, an occluded left common iliac artery with external iliac artery reconstitution via internal iliac artery collaterals, and a right external iliac artery occlusion with common femoral artery reconstitution. There was calcified right common femoral artery plaque.

Treatment options included open surgical aortobifemoral bypass with exclusion of the AAA, total endovascular repair with some form of endo-conduit revascularization of the occluded segments of iliac artery, or a hybrid repair.
Open aortic repair in patients with heart failure and moderate COPD can be performed safely (ref 1). Dr. Hollier et al, in the golden age of open repair, reported a 5.7% mortality rate operating on 106 patients with severe category of heart, lung, kidney, or liver disease.
Typically, the hybrid repair involves sewing in a conduit to deliver the main body of a bifurcated or unibody stent graft when endovascular access is not possible. Despite techniques to stay minimally invasive -largely by staying retroperitoneal, this is not a benign procedure (ref 2). Nzara et al reviewed 15,082 patients from the NSQIP database breaking out 1% of patients who had conduit or direct puncture access.
Matched analyses of comorbidities revealed that patients requiring [conduit or direct access] had higher perioperative mortality (6.8% vs. 2.3%, P = 0.008), cardiac (4.8% vs. 1%, P = 0.004), pulmonary (8.8% vs. 3.4%, P = 0.006), and bleeding complications (10.2% vs. 4.6%, P = 0.016).
Despite these risks, I have performed AUI-FEM-FEM with good results with the modification of deploying the terminus of the stent graft across an end to end anastomosis of the conduit graft to the iliac artery (below), resulting in seal and avoiding the problems of bleeding from the usually heavily diseased artery

The iliac limbs of some stent graft systems will have proximal flares and can be used in a telescoping manner to create an aorto-uni-iliac (AUI) configuration in occlusive disease. The Cook RENU converter has a 22mm tall sealing zone designed for deployment inside another stent graft and would conform poorly to this kind of neck as a primary AUI endograft which this was not designed to act as. The Endurant II AUI converter has a suprarenal stent which I preferred to avoid in this patient as the juxtarenal neck likely was aneurysmal and might require future interventions
I chose to perform a right sided common femoral cutdown and from that exposure, perform an iliofemoral remote endarterectomy of the right external iliac to common femoral artery. This in my experience is a well tolerated and highly durable procedure (personal data). Kavanagh et al (ref 3) presented their experience with iliofemoral EndoRE and shared their techniques. This would create the lumenal diameter necessary to pass an 18F sheath to deliver an endograft. I chose the Gore Excluder which would achieve seal in the hourglass shaped neck and allow for future visceral segment intervention if necessary without having a suprarenal stent in the way. I planned on managing the left common iliac artery via a percutaneous recanalization.
The patient’s right common femoral artery was exposed in the usual manner. Wire access across the occluded external iliac artery was achieved from a puncture of the common femoral artery. Remote endarterectomy (EndoRE) was performed over a wire from the common femoral artery to the external iliac artery origin (pictures below).
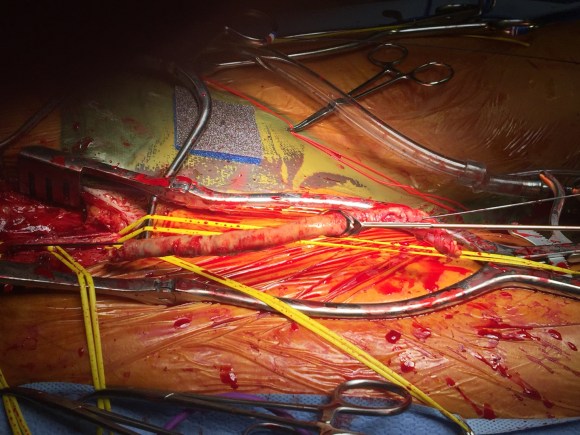
The 18F sheath went up with minimal resistance, and the EVAR was performed in the usual manner. The left common iliac artery occlusion was managed percutaneously from a left brachial access. The stent graft on the left was terminated above the iliac bifurcation and a self expanding stent was used to extend across the iliac bifurcation which had a persistent stenosis after recanalization.
The patient recovered well and was sent home several days postprocedure. He returned a month later with healed wounds and palpable peripheral pulses. He no longer had claudication and CTA showed the aneurysm sac to have no endoleak (figures below).

Discussion
I have previously posted on using EndoRE (remote endarterectomy) for both occlusive disease and as an adjunct in EVAR. Iliofemoral EndoRE has excellent patency in the short and midterm, and in my experience has superior patency compared to the femoropopliteal segment where EndoRE is traditionally used. This case illustrates both scenarios. While the common iliac artery occlusions can be expected to have acceptable patencies with percutaneous interventions, the external iliac lesions typically fail when managed percutaneously especially when the stents are extended across the inguinal ligament. The external iliac artery is quite mobile and biologically, in my opinon, behaves much as the popliteal artery and not like the common iliac. Also, the common femoral arterial plaque is contiguous with the external iliac plaque, making in my mind, imperative to clear out all the plaque rather than what can just be seen through a groin exposure.
On microscopy, the external iliac artery is restored to a normal patent artery -I have sent arterial biopsies several months after endarterectomy and the artery felt and sewed like a normal artery and had normal structure on pathology. This implies that the external iliac can be restored to a near normal status and patients that are turned down for living related donor transplantation of kidneys can become excellent recipients. In this case, this hybrid approach effectively treated his claudication but also sealed off his moderate sized AAA while not precluding future visceral segment surgery or intervention with a large suprarenal stent.
Reference
- Hollier LH et al. J Vasc Surg 1986; 3:712-7.
- Nzara R et al. Ann Vasc Surg. 2015 Nov;29(8):1548-53
- Kavanagh CM et al. J Vasc Surg 2016;64:1327-34

One of the nice things about practicing at the Clinic is being able to offer unique solutions. A severely diseased or occluded external iliac artery (EIA) can be a vexing problem, particularly if bilateral, in this endovascular era. Many cardiovascular devices require femoral access that has to traverse compromised iliac arteries -those with large (>16F) delivery systems require a sufficiently wide path to get the devices to the heart and aorta. Also, living related donor kidney transplantation is predicated on minimizing risk to maximize results and having significant iliac plaque negates one as a recipient for this high stakes elective procedure. In situations where the EIA is too small to accommodate devices because of atherosclerotic plaque, the typical solution is placement of a conduit to the common iliac artery or the aorta. The practice of “endopaving” with a covered stent graft and ballooning is also described, but its long term outcomes are not reported and the internal iliac artery is usually sacrificed in this maneuver.
This patient presents with lifestyle limiting claudication and an absent right femoral pulse. ABI is moderately reduced on the right to 0.57, and he had no rest pain. CTA at our clinic revealed an occluded EIA bracketed by severely calcified and nearly occlusive plaque of the common iliac artery (CIA) and common femoral artery (CFA).
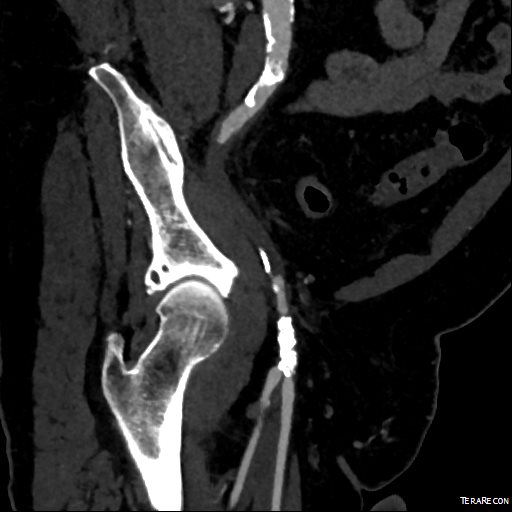
The patient was amenable to operation. Traditionally, this would have been treated with some form of bypass -aortofemoral or femorofemoral with a common femoral endarterectomy. While endovascular therapy of the occluded segment is available, one should not expect the patencies to be any better than that of occlusive lesions (CTO’s) in other arteries. Hybrid open/endovascular therapy is an option as well with CFA endarterecotmy and crossing CIA to EIA stents, but I have a better solution.
The common femoral endarterectomy rarely ends at the inguinal ligament, and is uniquely suitable for remote endarterectomy, a procedure from the early to mid twentieth century.
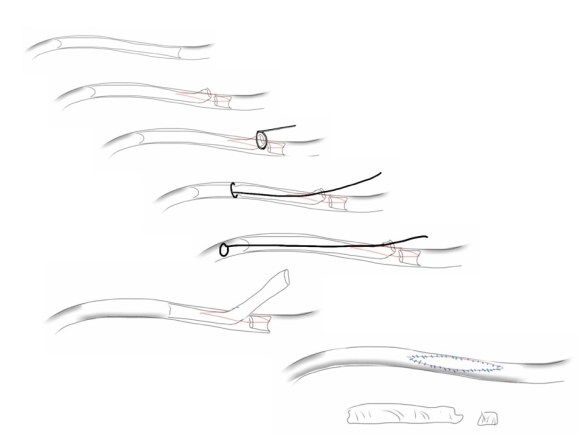
The addition of modern fluoroscopic imaging and combining with endovascular techniques makes this a safe and durable operation.

The patient was operated on in a hybrid endovascular OR suite. A right groin incision was made to expose the common femoral artery for endarterectomy and left common femoral access was achieved for angiographic access, but also to place a wire across the occlusion into the common femoral artery.
All actions on the external iliac artery plaque are done with an up-and-over wire, allowing for swift action in the instance that arterial perforation or rupture occur. This event is exceedingly rare when the operation is well planned. With this kind of access, an occlusive balloon or repairing stent graft can be rapidly delivered.
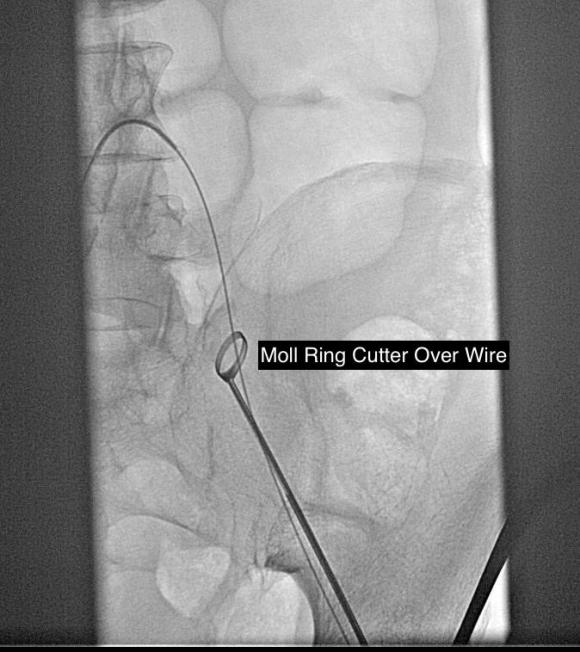
The common femoral endarterectomy is done from its distal most point and the Vollmer ring is used to mobilize the plaque. A Moll Ring Cutter (LeMaitre Vascular) is then used to cut the plaque.

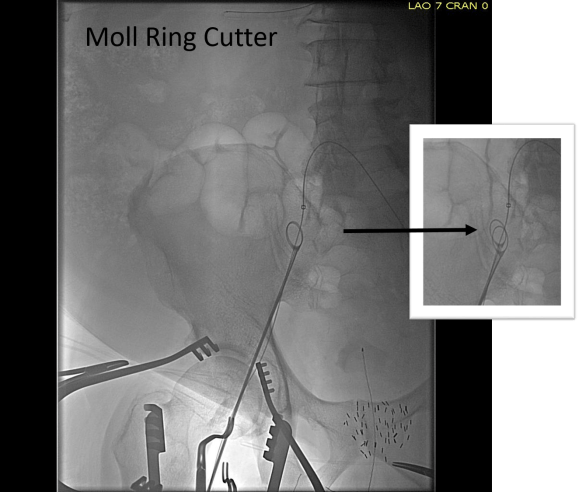
The plaque is extracted and re-establishes patency of the EIA.

The plaque end point is typically treated with a stent -in this case, the common iliac plaque was also treated.

What is nice about this approach is that this artery has been restored to nearly its original condition. I have taken biopsies of the artery several months after the procedure in the process of using the artery as inflow for a cross femoral bypass, and the artery clamped and sewed like a normal artery and the pathology returned normal artery.
This has several advantages over conduit creation which can be a morbid and high risk procedure in patients who require minimally invasive approach. A graft is avoided. The artery is over 8mm in diameter where with stenting up to 8mm with an occlusive plaque, the danger of rupture is present, and often ballooning is restricted to 6mm-7mm. This is insufficient for many TEVAR grafts and TAVR valves.
For patients being worked up for living related donor transplants who are turned down because of the presence of aortoiliac plaque, those with the right anatomy can undergo this procedure and potentially become candidates after a period of healing.

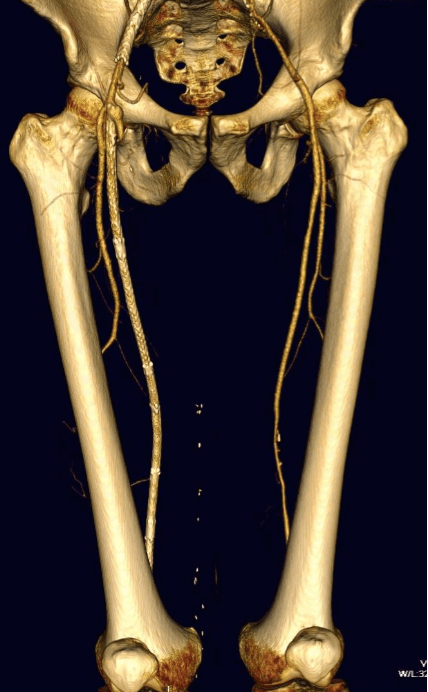
The patient presented with complaints of leg and foot pain with sitting and short distance calf claudication, being unable to walk more than 100 feet. This is unusual because sitting usually relieves ischemic rest pain. He is in late middle age and developed claudication a year prior to presentation that was treated with stent grafting of his superficial femoral artery from its origin to Hunter’s canal at his local hospital. This relieved his claudication only briefly, but when the pain recurred a few months after treatment, it was far worse than what he had originally. Now, when he sat at his desk, his foot would go numb very quickly and he would have to lie down to relieve his pain.
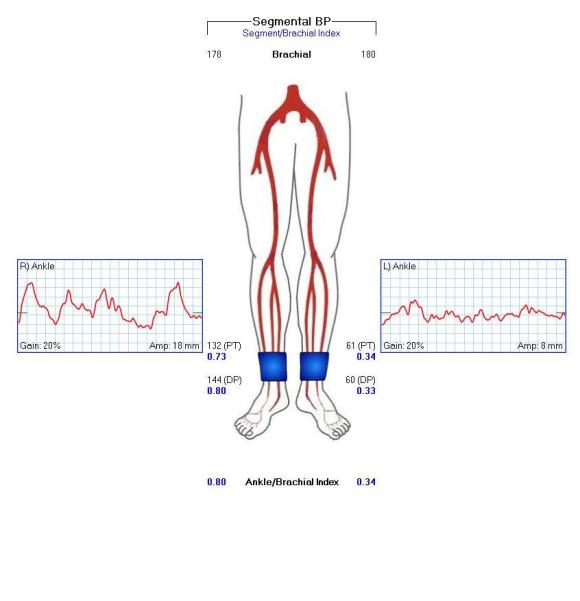
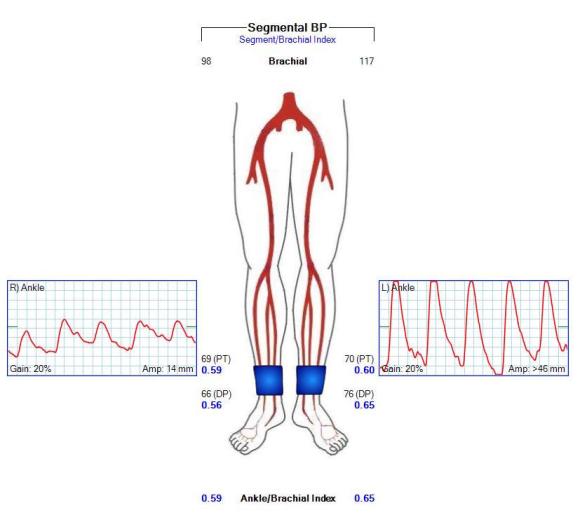
On examination, the patient was moderately obese with overhanging belly. He had a palpable right femoral pulse, but nothing below was palpable. He had multiphasic signals in the dorsalis pedis and posterior tibial arteries. The left leg had a normal arterial exam. Pulse volume recording and segmental pressures were measured:

These are taken with the patient lying down which was the position that relieved his pain, and the PVR’s show some diminishment of inflow. It would be easy at this point to declare the patient’s pain to be due to neuropathy or spinal stenosis, but because of his inability to walk more than a hundred feet and because of his severe pain with sitting, I went ahead and obtained a CTA.
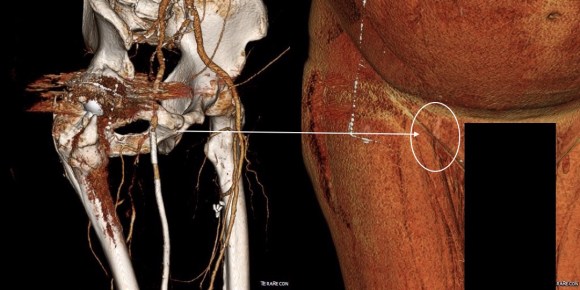
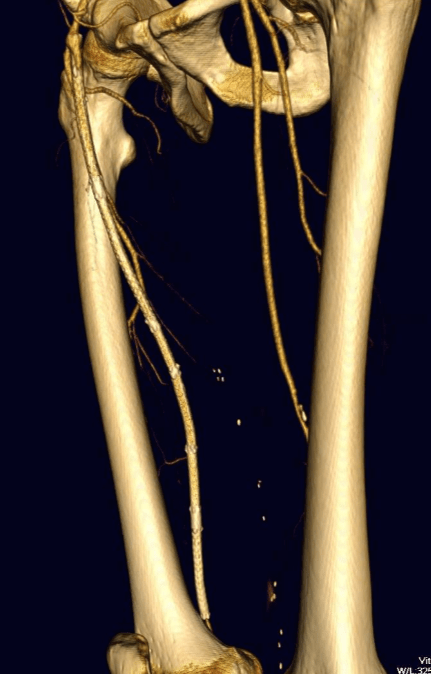
The CTA showed he had an occluded superficial femoral artery (SFA) with patent profunda femoral artery (PFA) with reconstitution of an above knee popliteal artery with multivessel runoff. The 3DVR image showed his inguinal crease to be right over the femoral bifurcation which is not an unsual finding, but his stent graft was partially occluding his profunda femoral artery.
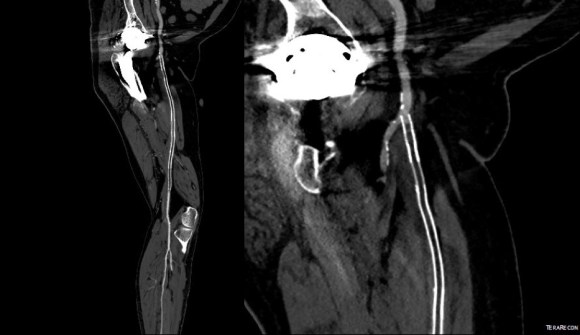
I decided to take him to the operating room to relieve his PFA of this obstruction. My plan was to remove the stent graft at the origin of the SFA and at the same time, remove the plaque and occluded stent graft from his SFA to restore it to patency.
In the OR, on exposing his SFA, I discovered that because of his overhanging belly, his inguinal ligament had sagged and was compressing his femoral bifurcation.

This explained his presentation. The stent graft really had no chance as when he sat, the belly and ligament compressed it at the origin, and because it partially occluded the origin of the PFA, sitting probably pinched off flow completely. The 3dVR image shows the mid segment of PFA to have little contrast density -this is not because of thrombus, but because of the obstruction, the PFA was getting collateral flow from the hypogastric artery.
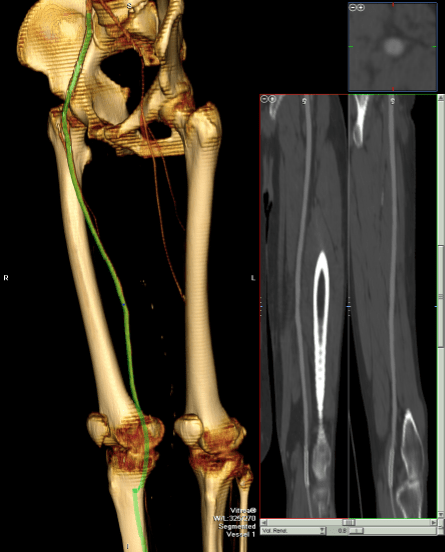
The stent graft was removed at its origin via a longitudinal arteriotomy after remote endarterectomy of the distal graft.
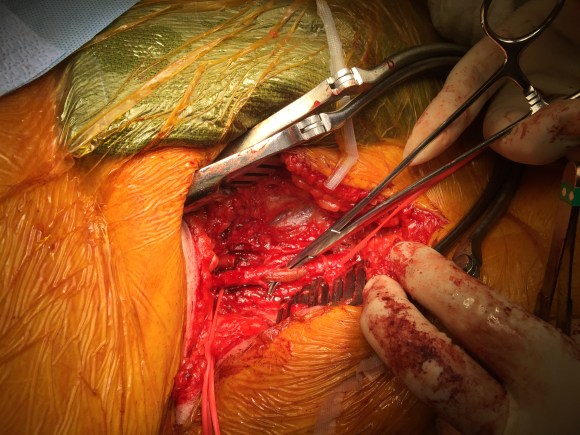
In this case, the Multitool (LeMaitre) was useful in dissecting the plaque and stent graft because of its relatively stiff shaft compared to the standard Vollmer rings. The technique of EndoRE has been described in prior posts (link).
The stent graft came out in a single segment -they come out easier than bare stents.


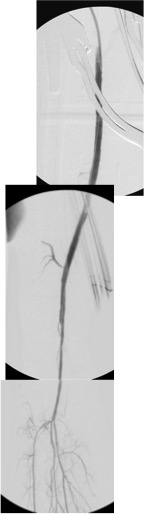 The patient regained palpable pulses in his right foot and recovered well, being discharged home after a 4 day stay.
The patient regained palpable pulses in his right foot and recovered well, being discharged home after a 4 day stay.
While one could argue that just taking out the short piece of occlusive stent graft over the PFA was all that was necessary, I feel that there is no added harm in sending down a dissector around the stent, and in this patient there was restoration of his SFA patency which was the intent of the original procedure.
Unlike PTFE bypasses that sometimes fail with thromboembolism, SFA EndoRE fails with development of focal stenoses. From a conversation I had with Dr. Frans Moll at the VEITH meeting, I found that he has had good experience with using drug coated balloons in the treatment of these recurrent stenoses.
At the time of discharge, the patient was relieved of his rest pain, and was no longer claudicating. The common femoral artery, its bifurcation, and the profunda femoral artery remain resistent to attempts at endovascular treatment, and remain in the domain of open surgery. And in retrospect, the history and physical examination had all the clues to the eventual answer to the oddities of the patient’s complaints. The combination of inguinal crease, abdominal pannus, and low hanging inguinal ligament meant these structures acted to crush the stent graft and femoral bifurcation.
In 2007, at which I had performed about 20 standard EndoRE (Remote Endarterectomy, LeMaitre Vascular) cases over about two years, this patient in his later 40’s presented to me with ischemic rest pain of his right foot. He was a current heavy smoker who initially had severe claudication and a TASC D occlusion of his right superficial femoral artery. Prior to being referred to me, he had undergone a mitral valve replacement from which he recovered uneventfully. He then had treatment of claudication starting with iliac stenting and a vein bypass. He had undergone a femoral artery to below knee popliteal artery bypass with reversed greater saphenous vein which became occluded after being complicated by MRSA wound infections. When this graft developed problems at the distal anastomosis, he underwent revision with a jump graft from the arm. This graft went down after he developed MRSA infection of the cephalic vein harvest site. He then underwent SFA stenting with 5 femoral stents (at that time, long stents were not available), but these occluded and his access site was the nidus of MRSA based sepsis. He had had multiple hospitalizations for MRSA infection from phlebotomy sites when he presented. He had reintervention for in-stent restenosis, first with balloons, then an extra stent, then cryoballoon therapy, each episode complicated by MRSA infection. He presented with severe claudication and nocturnal rest pain. On exam, he had dependent rubor, elevation pallor and absence of pulses, despite having fairly benign anatomy on CTA.
There was two vessel runoff below a reconstituted popliteal artery, with stent occlusion and visible stump of the vein bypass.
My options included bypass with PTFE, cadaveric vein, endovascular recanalization of the occluded stents, or EndoRE. While considering the MRSA which had been extensively worked up prior to presentation by ID including TEE and multiple cultures, it was decided that he was firmly colonized with MRSA despite efforts at eradication, and PTFE was not an option. Cadaveric vein I have used in infections with acceptable short term results -never great long term except for one individual who I inherited from a surgeon in Kansas who maintained a decade of patency of a cadaveric vein to tibial artery bypass with coumadin alone. This patient was not likely to be so lucky. Endovascular recanalization with atherectomy versus laser was considered, but I had at that point become disillusioned with those modalities in such extensive disease.
EndoRE made the most sense because it was my observation in a prior patient in whom I had unintentionally removed a 4cm stent with plaque that stents are placed inside plaques and when you remove plaque, theoretically, the stents have a layer of plaque between them at the adventitia. Also, he had none of the extensive calcium that made regular EndoRE challenging. Also, it would be repaired with native tissues through a single groin incision, and covered with a sartorius flap. And that is what I did.
The common femoral artery was exposed and the SFA controlled. The plaque dissection was started and the ring fitted around plaque and stent. There was a little more friction than expected, but I did inject via a catheter cold LR with the idea that it would shrink the nitinol a bit. Also, the wire that guided the catheter did double duty as a dissector as I was subintimal with it. The rings traveled well to the end point which I achieved with little difficulty.

The end point was dissected and required a short self expanding stent. The patient recovered well and was discharged, but as in prior admissions, developed a cellulitis on the groin wound that resolved with Vancomycin, presumably with MRSA. A CTA done at that admission showed excellent patency and he had palpable pulses.
Three years later, he underwent intervention by one of my partners in cardiology at that time for a restenosis in the mid SFA and had ballooning and a stent -the second set of stents in this patient, and by the time I left Iowa, he was still patent and walking.
This operation fails with randomly distributed TASC A lesions that develop in sites of remnant smooth muscle. I think today, I would treat with a drug eluting balloon. Thrombosis is the other failure mode, but unlike PTFE grafts, there is no thromboembolism of the outflow, rather, the SFA thromboses with reconstitution of the original state, and is amenable to thrombolysis. Smokers such as this patient and those with limited outflow are anticoagulated with warfarin.
The Europeans call this now arterial restoration. The vessel is returned to its baseline state with a full complement of collaterals which are revived. Also, compliance is restored and I believe this plays a significant role in maintenance of patency. Also, as the native tissues heal, they return to a normal ultrastructure -I have taken pathology specimen with aortic punches to perform bypasses to the other leg from external iliacs treated so, and they were microscopically and visibly normal.

This patient is a 90 plus year old man who developed ever worsening claudication to the point he was disabled and more worryingly, had developed pain over his left heel. His ABI’s were severely diminished.
CTA showed that he had an occluded SFA with above knee reconstitute, but also had only single vessel runoff to the foot via a heavily diseased posterior tibial artery that had serial mild to moderate stenoses.
An attempt at endovascular recanalization was performed at an outside institution, but the SFA lesion could not be crossed. Bypass was not a good option -the ipsilateral saphenous vein had been harvested for CABG, and a long operation was going to have a significant impact on this patient who also had mild dementia and drank 2-3 glasses of wine a day. It is not uncommon to have a successful operation, but have the patient lose 2-3 months in recovering from the physical effects of a long operation as well as from perioperative delirium.
I felt that removing the occlusive plaque from his arteries offered a minimally invasive solution. The plaque was easily accessible via an oblique, skin line incision in the groin, and clearance could be performed from the external iliac artery origin to the planned endpoint slightly beyond Hunter’s canal. While the outflow was not perfect, in my experience, aside from a single native vein bypass, long segment restoration of vessel elasticity results in very acceptable patency rates.
Remote endarterectomy is a bit of a lost art from the early days of vascular surgery. A ring dissector (Vollmer Ring Dissector, LeMaitre Vascular) is used to liberate the plaque from the remnant adventia. A cutting device (Moll Ring Cutter, LeMaitre Vascular) shown third from left below is used to divide the plaque.
The common femoral artery plaque is usually contiguous with plaque in the external iliac artery and surgeons who perform a lot of CFA endarterectomy have various maneuvers to remove as much plaque as possible, up to stenting the end point of the plaque down to the endarterectomy patch. I have never been satisfied with this because the EIA behaves differenty than the CIA (am looking into this!) in my experience and placing stents even minimally across the inguinal ligament is not desirable. Sending the dissector up to the EIA origin frees the plaque to be removed completely with the CFA plaque. The clip below shows the Vollmer Ring dissecting plaque up to the EIA origin. I do this over a wire in the pelvis because in the rare instance of leak or rupture, rapid control is possible without having to open the abdomen.
Once freed, the cutter is used to transect the plaque and the end point is tacked down with a stent at the distal common iliac/EIA origin which is a better place for a stent than the inguinal ligament.
The PFA in this patient did not require endarterectomy and reconstruction, but if it did, I would have made the arteriotomy go onto the profunda from the CFA. The SFA plaque is then mobilized with the Vollmer ring. I don’t do this over a wire, but have a definite end point in mind based on what I see on CTA.
The CTA (images earlier) shows that the above knee popliteal artery has no significant calcified plaque. This doesn’t mean there isn’t fibrotic plaque. Cutting the plaque as in the clip below results in a coned in antegrade dissection which has to be crossed in the true lumen.
This is technically the most difficult part of the EndoRE procedure and it requires good imaging and wire skills. The trick here is that an ultrasound guided puncture of the popliteal or tibial vessel can give you distal true lumen access if needed. It was not necessary in this patient. The better maneuver is if the end point is surgically accessible is to cut down and tack down the plaque and patch the arteriotomy.
The patient regained multiphasic PT and DP signals at the end of the case, after the common femoral artery was patched and flow restored. The small groin incision was closed with a running absorbable monofilament after multilayer deep closure. The patient had a blood loss of 50mL. An ilioinguinal field block and local anesthesia provided excellent pain control. Postoperative ABI was improved to 0.82 from 0.34 and all pain was relieved. The patient felt good enough to go home on postoperative day 1.
This illustrates what I feel to be a best application of both open and endovascular techniques. The above knee popliteal stent is short and in a position that is not going to result in fracture. The external iliac stent is in a protected position in the pelvis and quite large -10mm, which I expect will stay open for the life of the patient. The profunda femoral artery, the rescue artery, is widely patent, and numerous collaterals off the SFA have been restored to patency which I feel aid in maintaining the patency of this repair, along with the restored elasticity of the artery which mimics the biomechanics of autologous vein.
In most patients with compromised outflow, I start warfarin along with ASA at 81mg. Because of his age, I opted for Plavix+ASA. These fail with the development of random TASC A restenoses along the SFA which are amenable to balloon angioplasty. The role of drug eluting balloons in this situation is unknown but theoretically promising. Occlusion through thrombosis does not result in embolization and limb loss as in failure of prosthetic bypass grafts (another option in this patient), but rather leaves a situation where endovascular thrombectomy or lysis is technically feasible.
The great thing is that this is by far superior to stenting of a TASC D femoral arterial lesion.

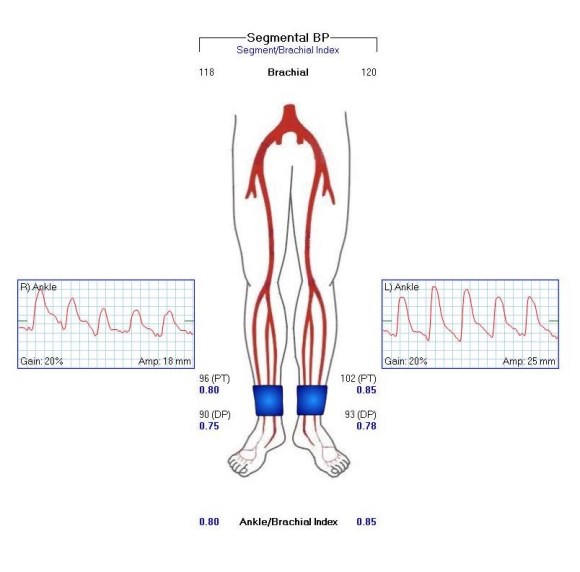
The patient is 70 year old woman with prior history of smoking who developed severe claudication and near rest pain. She was unable to walk more than 50 feet before having to stop due to severe leg pain. On exam, neither femoral artery pulses were palpable. PVR’s (pulse volume recordings) and ABI’s (ankle brachial index) are shown below.
PVR’s demonstrate the presence of severe inflow (aortoiliac occlusive disease or AIOD). CTA was acquired and the findings were consistent with the PVR’s.
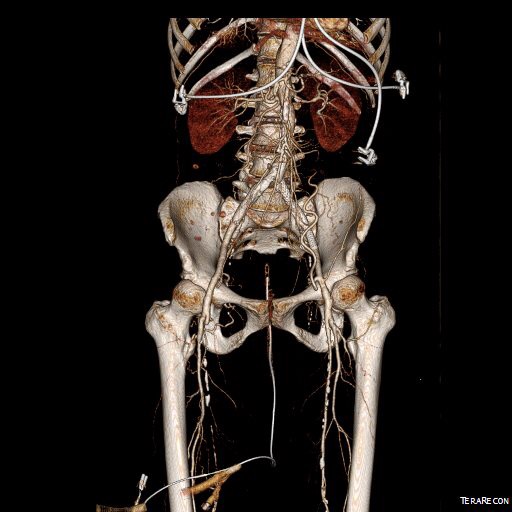
There was diffuse bilateral iliac atherosclerotic plaque with occlusion of the right common femoral artery and left common and external iliac artery. The 3DVR (three dimensional virtual reality) reconstruction image below shows this as well as the abdominal and pelvic wall collaterals feeding the legs around the occluded iliofemoral system.
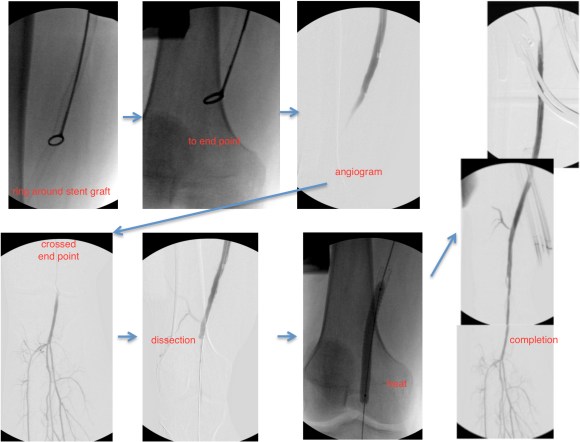
Plans were made to perform a hybrid common femoral and profunda femoral endarterectomy, remote external iliac artery endarterectomy (EndoRE), and common iliac artery stenting. The specific challenges to this case was getting into and staying in the true lumen. Typically, this is easiest to achieve from a left arm access with wires being pushed antegrade, but in a smaller person, particularly woman, this increases the chances for access site complications. My plan was to expose both common femoral arteries and get control of the external iliac arteries at the inguinal ligament and the profunda femoral arteries at the point the proximal plaque dissipated -typically at the second branch point, and then get micropuncture access of the right iliac system by accessing from the common femoral plaque. This would give me true lumen access, and with a sheath and curved catheter (VCF in this case, but a similarly shaped OMNI Flush catheter would do as well), wire access up and across the occluded left iliac system could be achieved and the wire retrieved from the left common femoral artery. This up and over access with the wire allows for control of the aortic bifurcation and both iliac systems.
I perform EndoRE over this wire -this allows for quick access if the artery is ruptured. To minimize blood loss, I gain control of the common femoral artery in the following fashion -a 4cm segment of common femoral artery is left intact and looped above the inferior epigastrics -this loop is brought out in the lateral lower quadrant of the abdomen so that the loop doesn’t travel distally over the arteriotomy. The second loop adjacent to the arteriotomy is sent through periadventitial tissues behind the artery to keep the loop migrating over the arteriotomy. The arteriotomy is created from the distal CFA (common femoral artery) onto the profunda femoral artery (PFA) where the endarterectomy is started. A separate arteriotomy on the superficial femoral artery (SFA) allows me to divide the plaque and mobilize the proximal segment up to the SFA origin, freeing the CFA plaque in this manner. It also gives me the option to perform EndoRE of the SFA if warranted. The dissected plaque and system of loops which I call the blood lock is shown below:

The yellow loops are major control points (the blood lock loop is drawn in the picture above) and the red loops are around smaller branch arteries. At this point, micropuncture access through the plaque core was achieved into the true lumen of the yet patent EIA (external iliac artery, picture below).

The right EIA plaque was mobilized with a Vollmer ring dissector, and cut with a Moll ring cutter (LeMaitre).
This allowed for cutting and removal of the plaque.

Up and over access and control of the wire from the contralateral (left) arteriotomy allowed for EndoRE on the other side. The occluded left common iliac plaque was ballooned and wire access into the aorta from the left was achieved.
Kissing balloon angioplasty was performed with revascularization of the aortoiliac bifurcation and common iliac arteries.
The stents were extended across the dissected end points of the external iliac artery origins. The arteriotomies were closed with bovine pericardial patches. Because the PFA were of small caliber, to avoid narrowing the distal end of the patch, the patches were sewn over Argyll shunts which also allowed perfusion of the legs during the suturing of the patches. The loops made this a straighforward maneuver.
The completed CFA to PFA patch on the left is shown below:
Closure involved reapproximating the Scarpa’s type investing fascia of the femoral triangle and a running dermal layer of absorbable monofilament, dressed with a surgical glue. No drains were used, but if needed, they would be exited through the counter incisions created for the EIA loops.
The patient recovered well. I always use cell salvage -sometimes, profundaplasties can be bloody, particularly if they are in reoperative fields. The ABI’s and PVR’s at the ankles improved significantly.
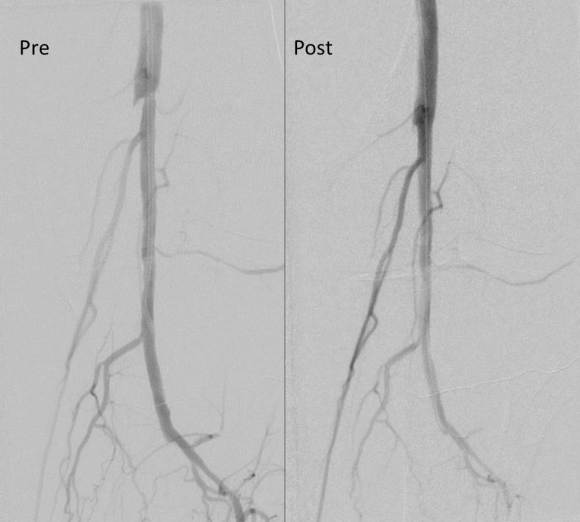
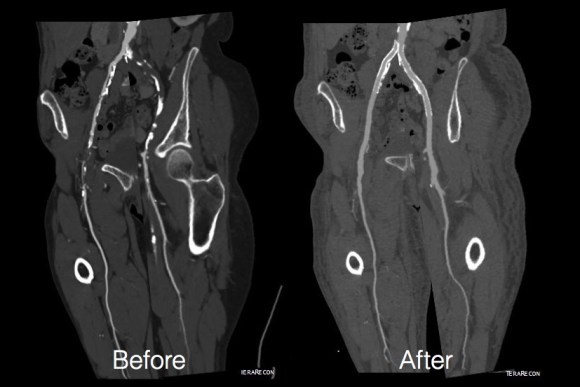
 The postoperative CTA shows good results as well. Below is the composite right and left centerline from aorta to the PFA’s.
The postoperative CTA shows good results as well. Below is the composite right and left centerline from aorta to the PFA’s.
The 3DVR reconstruction images are shown below, with the comparison to preop shown in the first image of this blog entry:
The pre and postoperative images of the centerlines (composited) are shown below:
EndoABF is an established hybrid procedure involving an open endarterectomy of the common femoral and PFA/SFA with iliac balloon angioplasty and stenting, often taking the stents distally into the CFA and the patch to deal with complex distal EIA plaque. This procedure, which would be an EndoRE ABF, offers some advantages in eliminating the need for EIA stents which are often placed across the inguinal ligament and into the patch during EndoABF. In my experience, the EIA EndoRE performed as an extension of a CFA endarterectomy is safe, and made even safer by performing the EndoRE over a wire. Published results from Europe shows for TASC C and D disease, EIA EndoRE has excellent patency, and I would expect the same here. EndoRE and Endo ABF both offer advantages over traditional ABF, particularly in patients with medical comorbidities.
The patient is a 70 year old man who arrived with complaints of worsening claudication, worse on the left leg. He smokes over a pack a day. On exam, he only had femoral pulses, nothing was palpable below. PVR showed multilevel disease with an ABI of 0.42 on the left leg.
CTA was done showing that both his SFA and PFA were occluded, along with occlusion of his AT in the mid leg, and tibioperoneal trunk.
There is a reconstitution point on the PFA, and there is also SFA constitution. Looking at this, it was apparent to me that it would be possible to endarterectomize the whole of the iliofemoral and femoropopliteal system from a single groin incision, but the question being, would a profundaplasty be sufficient.

The textbook answer is profundaplasty, but given my experience with endarterectomy, it has become apparent that removing all the plaque, including CFA and iliofemoral plaque reduces the chance that clamp injury and stenosis occur, and that placed in the common iliac system have better patency than those placed in the external iliac, particularly crossing the inguinal ligament into a patch.
The other observation is that with this exposure, SFA remote endarterectomy is very simple to do, but becomes more difficult in a redo situation. The only problem with going ahead with it is that the runoff is poor -all three tibial vessels occlude, but a very robust posterior tibial artery reconstitutes proximally from well developed collaterals.
The CFA, PFA, and SFA were exposed as shown in my sketch at the beginning of the post. Wire access up and over from the right side allowed for secure control of the aortoiliac segment. The endarterectomy was started from the PFA reconstitution point and the CFA plaque was mobilized. The SFA plaque was transected in a proximal arteriotomy and the plaque was mobilized with a ring to its origin. The CFA plaque then was mobilized with the ring dissector over a wire (for security in case of rupture), up to the EIA origin and cut.
The distal SFA plaque was endarterectomized to the planned end point above the knee joint.
The specimen is shown below.
The arteriotomies were repaired with patches. The common iliac artery was stented to improve the flow. The SFA end point was managed with a stent, placed proximal to the first large geniculate collateral.
Completion angiograms show widely patent EIA, CFA, PFA, and SFA
The patient recovered and was discharged on POD#3. His postop ABI’s are shown below.
They are improved compared to preop, with ABI’s of 0.65. Notably, he did have a weakly palpable posterior tibial artery pulse, and multiphasic signals in all three tibial vessels. While I don’t know if the SFA revascularization will stay open, I am confident the PFA will, and this will keep him from his symptoms recurring and is a durable procedure.
Ideally, if he had needed a distal revascularization, a vein bypass would be the answer, but in the setting of inadequate conduit, it is very simple to endarterectomize from the below knee popliteal artery the remaining plaque and either patch to the patent tibioperoneal trunk or perform a short POP to posterior tibial artery bypass. He did not require this.
I don’t know the answer to the titular question, but in the setting of an inflow procedure, the best chance at opening the SFA is during the inflow procedure because of the exposure, and it is very simple to do when the lesion is minimally calcified.