Leriche Syndrome -one of those disease names that adds to our work in a way that an ICD codes and even the “aortoiliac occlusive disease” fails to describe. When I hear someone described as having Leriche Syndrome, I think about a sad, chain smoking man, unmanned, complaining of legs that cramp up at fifty feet, pulseless.
The CT scan will occasionally show an aorta ringed by calcium in the usual places that are targetrs for clamping below and above the level of the renal arteries. Even without the circumferential calcium, a bulky posterior plaque presages the inability to safely clamp the aorta. Woe to the surgeon who blithely clamps a calcified lesion and finds that the rocky fragments have broken the aorta underneath the clamp! The first way to deal with this is to look for ways not to clamp the aorta, by planning an endovascular procedure, but circumstances may necessitate the need to control the aorta despite the unclampability.
The traditional methods of avoiding clamping the calcifed peri-renal aorta are extra-anatomic bypasses including femorofemoral bypass and axillo-femoral bypass. I propose these following options for the consideration when the patient needs a more durable solution while avoiding a heavily diseased aorta.
Not Clamping I:

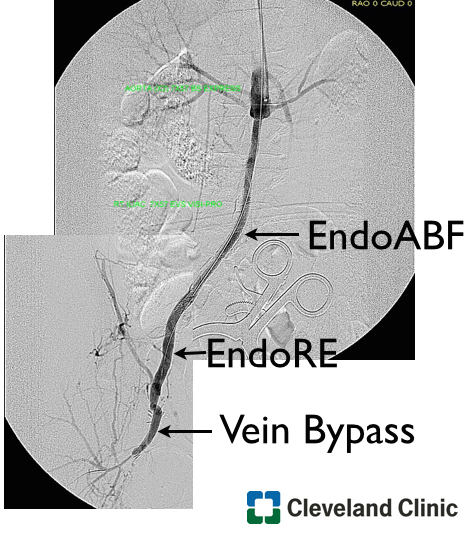
EndoABF does work to avoid clamping -these are common femoral endarterectomies supplemented by stenting of the aortoiliac segment, including in those with appropriate anatomy, a bifurcated aortic stent graft. This is often not possible to treat both sides, but one side is usually more accessible. Often, people will compromise and perform an AUI-FEM-FEM, but I have found the fem-fem bypass to be the weak link, as you are drawing flow for the lower half of the body through a diseased external iliac artery. The orientation of the proximal anastomosis is unfavorable and in the instance of highly laminar or organized flow, the bypass is vulnerable to competitive flow on the target leg, leading to thrombosis.

The femorofemoral bypass is the option of patients whose options have largely run out. It is made worse when fed by an axillofemoral bypass. Sometimes, you have no choice, but in the more elective circumstance, you do.
Not Clamping II:
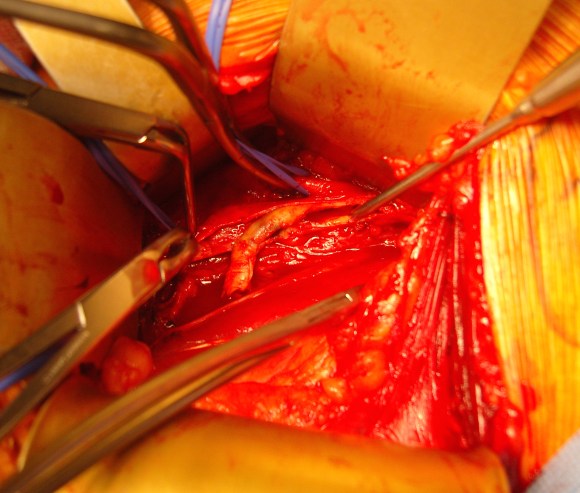
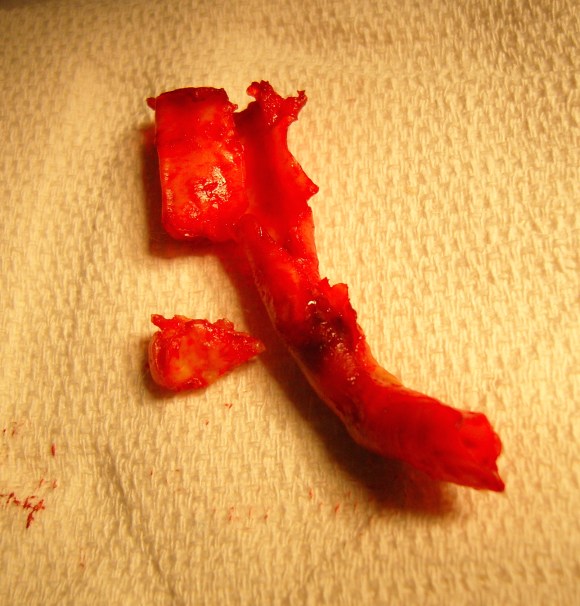
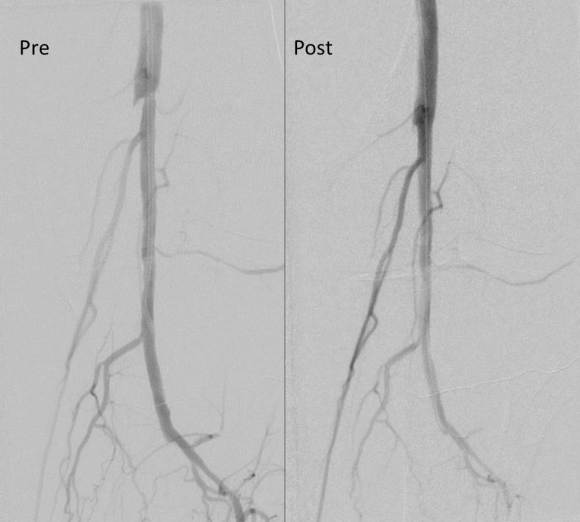
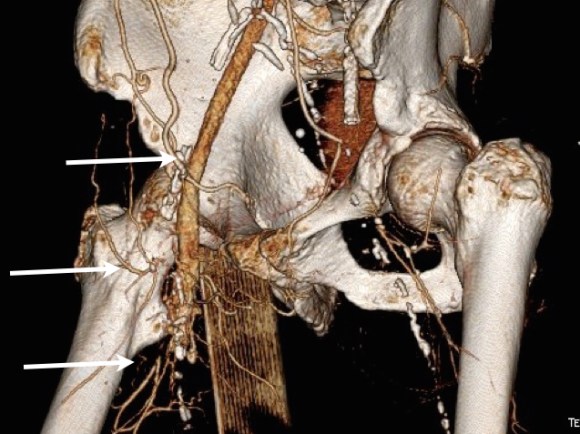
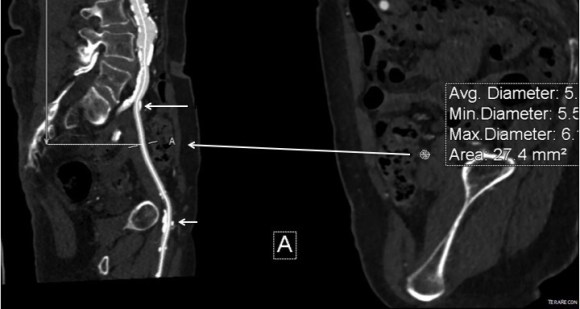
The second method is performing a aorto-uni-iliac stent graft into a conduit sewn end to end to the common iliac aftery, oversewing the distal iliac bifurcation.
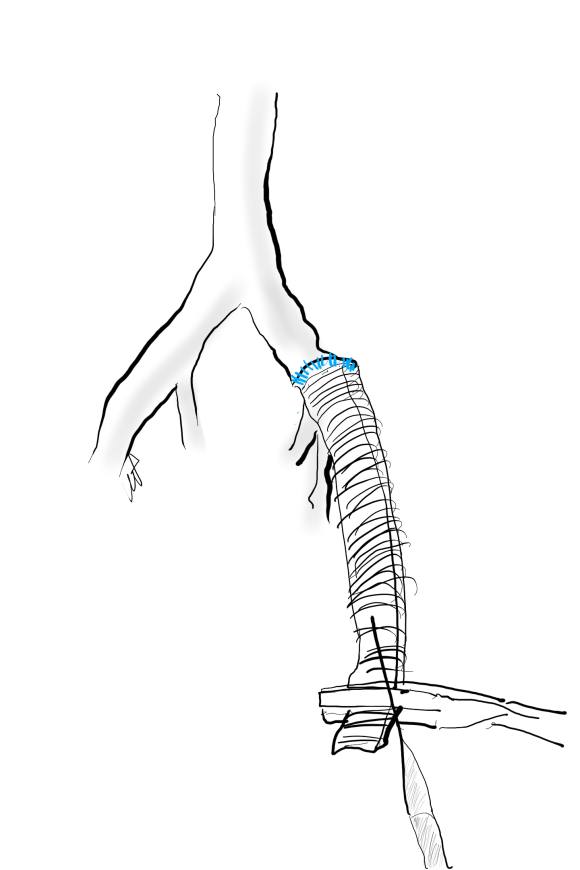
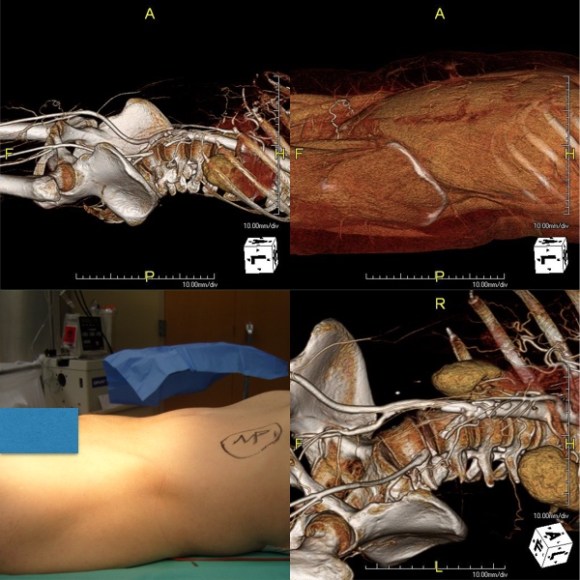
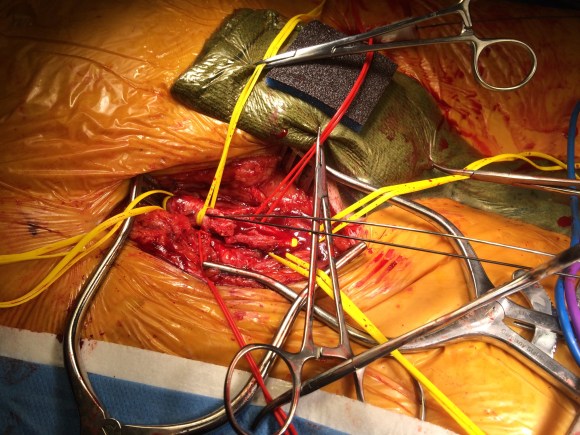
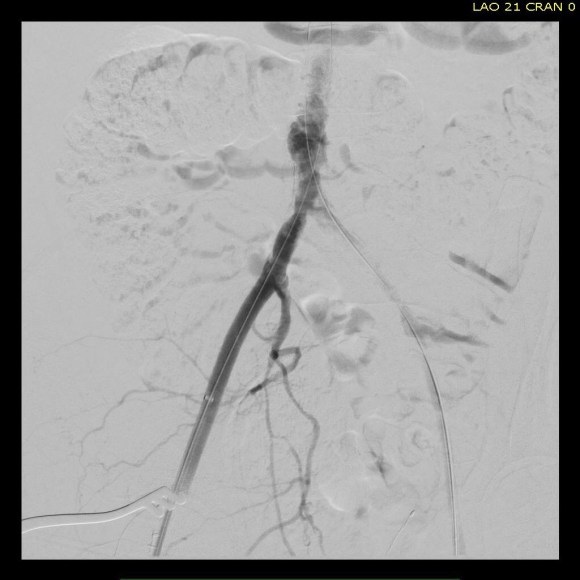
The conduit is 12mm in diameter, the key is to deliver the stent graft across the anastomosis, sealing it. The conduit is then sewn to the side of a fem-fem bypass in the pelvis, maintaining antegrade flow to both legs. The other option is to sew the conduit to a 14×7 bifurcated graft. Illustrated above is this 12mm conduit sewn end to end to the diseased common iliac artery with wire access into the aorta and a aorto-uni-iliac device. Typically, a small AUI converter (Cook, Medtronic) can be used, but the aorta is often too small even for a 24mm device, and an iliac limb with a generous sized docking segment (Gore) ending in a 12mm diameter fits nicely. Below is a CTA from such a case, where the stent graft is deployed across the anastomosis, sealing it off from anastomotic leaks (exoleaks).

Not Clamping III:
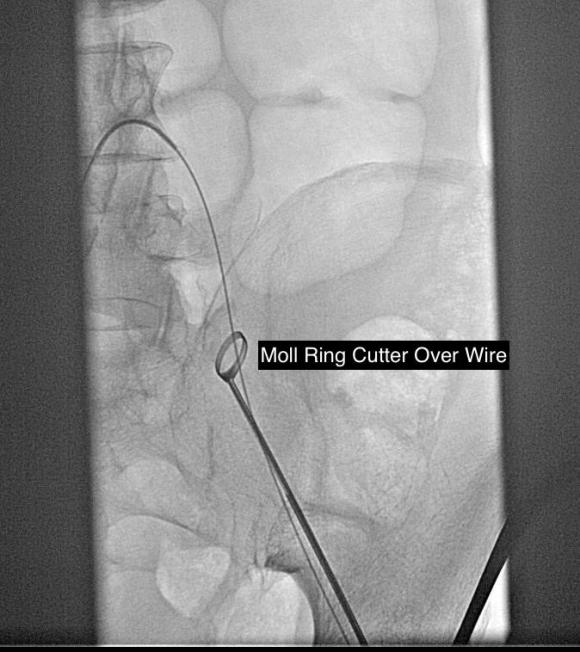
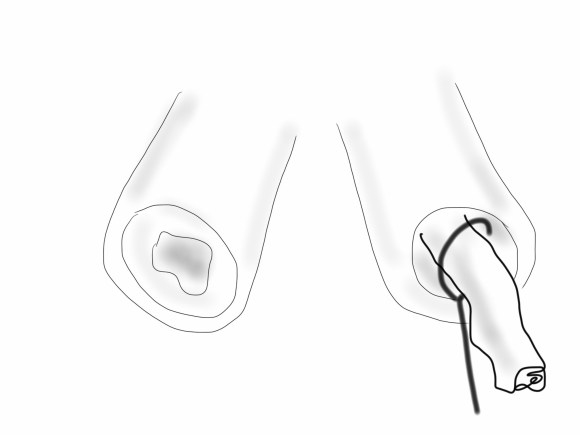
Often, the infrarenal aorta is soft anteriorly and affected only by posterior plaque at the level of the renal arteries. While a clamp is still not entirely safe (I prefer clamping transversely in the same orientation as the plaque with a DeBakey sidewinder clamp), a balloon is possible. I do this by nicking the aorta -simple application of a finger is sufficient to stop the bleeding if you have ever poked the ascending aorta to place cardioplegia line.

A Foley catheter is inserted and inflated. The Foley’s are more durable and resist puncture better than a large Fogarty. This is usually sufficient for control, although supraceliac control prior to doing this step is advised. The aorta can be endarterectomized and sewn to the graft quite easily with this non-clamp. 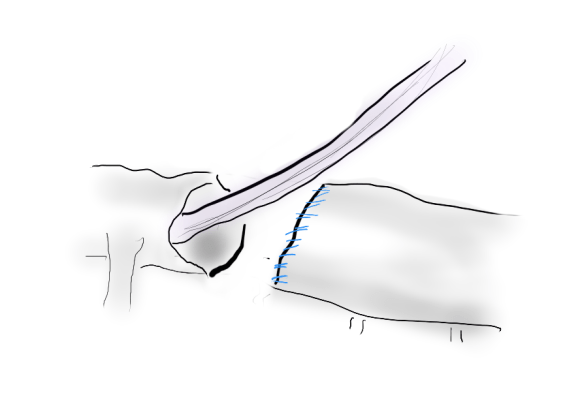
This has worked well, Although pictured above with an end-to end anastomosis planned, it works just as well end-to-side. I actually prefer end to side whenever possible because it preserves the occluded native vessels for future intervention in line.
The Non-Thoraco-Bi-Femoral Bypass
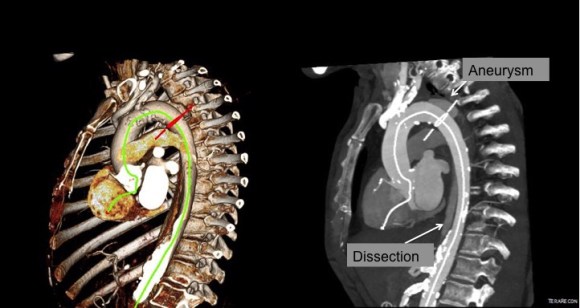
The typical board answer for the non-clampable aorta is taking the inflow from the thoracic aorta or from the axillary artery -neither of which are good options. The first because the patient is positioned in right lateral decubitus and tunneling is not trivial. The second because of long term durability. The supraceliac aorta, technically it is the thoracic aorta, is often spared from severe plaque and clampable. Retropancreatic tunelling is straightforward, and a 12 or 14mm straight graft can be tunelled in this fashion from the lesser sac to the infrarenal retroperitoneum. It then sewn to the supraceliac aorta and then anastomosed to a 12x6mm or 14x7mm bifurcated aorto-bifemoral bypass, of which limbs are tunneled to the groins.
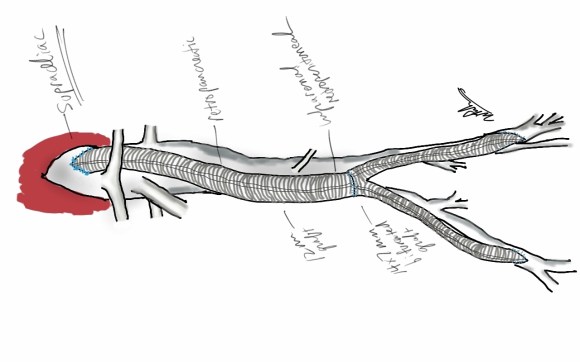
This worked very well recently, allowing a middle aged patient with severe medical problems, occluded aorta and iliac arteries, with critical limb ischemia, survive with minimal blood loss and home under 5 days. It delivers excellent flow to both legs in an antegrade fashion. Dr. Lew Schwartz gave me a list of references showing that this is not novel, but represents a rediscovery as the papers were published in the 80’s [reference], and buttresses the principle that innovations in open vascular surgery are exceedingly rare, largely because we have been preceeded by smart people.
Conclusion: All of these come about through application of some common sense and surgical principles. The most important this is that the aorta is the best inflow source and reconstructing it with the normal forward flow of down each leg and not reversing directions as in a fem-fem bypass gives each of these options a hemodynamic advantage.
References for Supraceliac Aorta to Lower Extremity Bypass
- Surgery [Surgery] 1987 Mar; Vol. 101 (3), pp. 323-8.
- Annals of Vascular Surgery 1986 1(1):30-35
- Texas Heart Institute Journal [Tex Heart Inst J] 1984 Jun; Vol. 11 (2), pp. 188-91.
- Annals of Thoracic Surgery 1977 23(5):442-448