A basic recipe for treating complicated aortic dissection
#aorticsurgery #tevar
A basic recipe for treating complicated aortic dissection
#aorticsurgery #tevar

Type B aortic dissections (TBAD) are frequently seen here at the Clinic as we serve as a regional referral center. As a trainee, I read the chapters discussing all the classifications and discussions of the biomechanics and felt quite intimidated by the all the moving parts involved in an aortic dissection, and I missed the main point about TBAD. Aside from the rupture risk due to the attenuation of the adventitia and hypertension, the acute TBAD is a rapidly developing stenosis of the aorta due to the inflation of a wind sock balloon created by the dissection flap. You can assume any flow that occurs in the false lumen is limited by the area of the proximal tear which is always smaller than the area of the aortic lumen. The true lumen is still perfusing the lower half of the body, and because of the volume filling effect of the flap, flow is restricted. The equivalent physiology is seen in aortic coarctation. Long term, the false lumen behaves like a pseudoaneurysm and may thrombose, continue to grow, or both.
Our group looked at CT’scans on 80 consecutive patients and found that the true lumen to false lumen ratio of less than 0.37 is predictive of the need for intervention.

This makes hemodynamic sense as it approximates the 70% critical stenosis borderline for other arteries. It explains why closing the opening of the dissection, the opening of the wind sock, and expanding the true lumen effectively treats malperfusion.

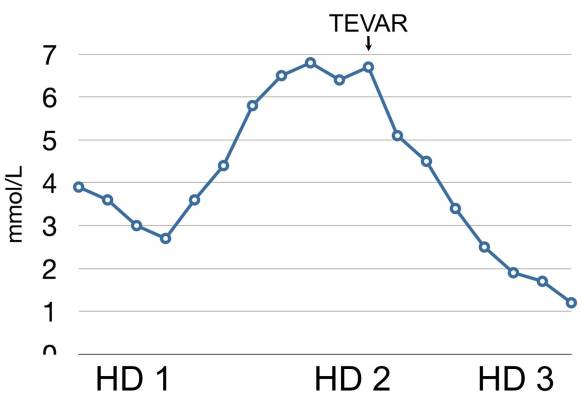
This patient whose CTA is shown above was transferred with increasing abdominal pain, inability to control blood pressure, and worsening lactic acidosis.

There was nearly complete obliteration of the true lumen throughout the aorta and occlusion of the left renal artery and dissection into the celiac and superior mesenteric arteries.

Aortography showed the dissection, and absence of visceral vessels from the injection which was from the aortic root. True lumen position was confirmed with IVUS.

A thoracic stent graft was delivered across the left subclavian artery origin up to the innominate artery origin -the patient had a bovine arch. Immediately, there was filling of the visceral vessels with re-establishment of true lumen flow.

The renal occlusion appeared improved but there was still a stenosis due to deflated dissection flap and this was stented (panel right above).
His abdominal pain remitted and his lactate normalized. His creatinine stabilized and has since normalized.
Again, if the true-lumen is compressed, the aorta is stenotic because there is a wind sock inflated in it. TEVAR offers a minimally invasive option, frequently percutaneous, for treating this.
The figure above shows the summarizes the problem that brought the patient to his local hospital and triggered his transfer to our acute aortic syndrome unit. The concept is that all chest pain of cardiovascular origin gets intake through a vast intensive care unit staffed by cardiovascular intensivists. Stabilization, workup, transfer to operating room or interventional suite all happens in an ICU that encompasses almost a city block.
The patient is an older middle aged man with sudden onset of back and abdominal pain. He was on coumadin for a prior SMV thrombosis and treatment for a ruptured appendicitis -antibiotics with plan for staged appendectomy. CT at his local hospital revealed a type B aortic dissection (TBAD) that extended into his superior mesenteric artery.
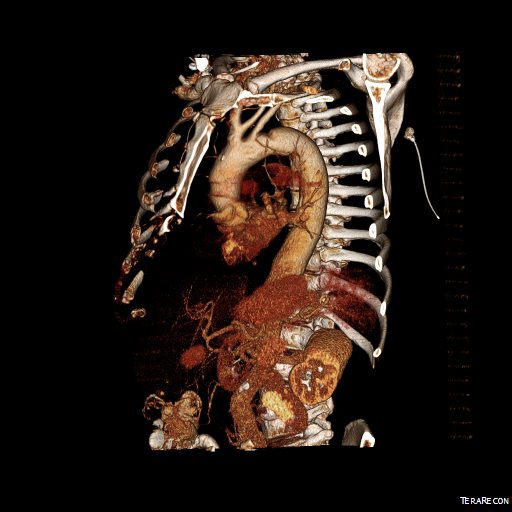
The dissection started at the left subclavian artery origin. The false lumen compressed the true lumen up at the proximal descending thoracic aorta. This is an important finding because in this configuration with much of the filling of the dissection from the distal reentry sites, the false lumen acts like a pressurized, compressive lesion. With time, the adventitia around the false lumen may become aneurysmal if the false lumen fails to thrombose or obliterate. When the dissection is acute, the flap may cause a direct obstruction to flow or a dynamic one that depends on the pressure difference between true and false lumen.
In this patient, thrombosis occured in the SMA beyond the origin due to dissection and decreased flow. This was consistent with the patient’s complaint of generalized abdominal pain and examination findings of pain out of proportion to the exam, indicating acute mesenteric ischemia.
His laboratory findings were within normal ranges, indicating this was early in the process. It is important to remember that no lab value correlates with acute mesenteric ischemia except very late in the process, and acute mesenteric ischemia remains a clinical diagnosis (reference 1) that is associated with a high mortality rate.
He was taken to the hybrid operating room. Right groin access was achieved and wire access to the arch was achieved. IVUS (Intravascular ultrasound, Volcano) was used to confirm the location of the wire -I believe this is an important adjunct as simply passing the wire doesn’t guarantee travel up the true lumen.




Wire access was achieved across the thrombus. At this point, I had a range of options for thrombectomy including simply aspirating which retracting a catheter. This was not optimal as I could lose subsequent wire access or reenter the false lumen. The other option was an open thrombectomy and patch angioplasty -the thighs were prepped in case we had to harvest vein. Again, in the setting of dissection and going into the mesentery, an open revascularization while feasible, is challenging.
Thrombectomy catheters like Angiojet were available, but I chose to try the Export aspiration catheter (Medtronic). It is simple to set up and goes over a 0.14 wire. It is designed for the coronaries which have a similar lumenal diameter as the SMA. It worked well in this setting in retrieving thrombus which had a pale element that may have indicated some chronicity.
The completion arteriogram was satisfying.
The SMA completely filled as did the celiac axis and both renal arteries. I opted not to treat the right renal artery as we had given 250mL of contrast, and it was filling well without intervention. The patient was making excellent urine and his blood pressure had been maintained with mean arterial pressures above 70mmHg. At this point, IVUS confirmed good deployment of the stent.
The sheath was removed and the access site repaired. The general surgeons explored the patient and found all the bowel to be well perfused with pulsatile flows seen in the mesenteric arcade. The appendix was removed.
On waking, the patient was noted to not move his legs. A spinal drain was expertly placed by our cardiac anesthesia staff and his blood pressure was raised to MAP’s above 80. He recovered motor function in his legs soon after. I usually don’t place preop CSF drains in this scenario in the presence of good pelvic circulation, no history of infrarenal aortic interventions, and patency of the left subclavian artery. That said, with TEVAR of TBAD, there is a small incidence of paraplegia (1-5%) which I emphasize in my preoperative discussion.
He was started on heparin anticoagulation postop because of his history of SMV and now SMA thrombosis, interrupting it briefly to remove the CSF drain. A CTA was obtained to confirm absence of bleeding showing obliteration of the dissection in the aorta and good patency through the true lumen of the SMA.
Most importantly, he had complete relief of his abdominal pain.
The treatment of acute mesenteric ischemia has greatly evolved since I presented my paper in 2002. While open revascularization remains a gold standard, it is becoming increasingly apparent that good to better results may be obtained with an endovascular approach. Dan Clair, our chair, has made the comment that early revascularization with endovascular technique is analogous to emergent PTCA in occlusions of the coronary system and that re-establishing flow is a critical first step.
Open exploration still is the mainstay of managing acute mesenteric ischemia, but laparoscopic exploration remains feasible. This patient underwent open conversion after an initial laparoscopic exploration to remove a ruptured retrocecal appendix that had been treated for over a month on antibiotics. Without bowel necrosis, a second look is usually unnecessary, but is critical when threatened bowel is left behind.
Reference