The patient is a woman in her forties who works hard and smokes cigarettes to find stress relief. The year prior to presentation, she began to get cramps in her calves while she walked the halls of the building she cleaned, and this became unbearable. A consultation at our hospital revealed moderate to severe diffuse atherosclerosis without a dominant lesion but notable small distal aorta and iliac arteries with a 50% stenosis of the left iliac origin. Recommendations were to quit smoking and exercise. She found this difficult to achieve and went to another hospital nearby.
There, 6 months prior to presentation, she began complaining of painful cyanosis of her toes which was described as blue toe syndrome. These outside studies were not available. She was taken to the OR and her common iliac arteries were stented. This gave her relief, but the soon pain returned three months later -her stents had occluded. This was treated with more stents, extending them proximally into the aorta and distally in the case of the right across the iliac bifurcation. This afforded her relief for three more months until one weekend she found herself unable to walk again for more than minimal distances, and she took herself to my hospital, University Hospital, Cleveland Medical Center.
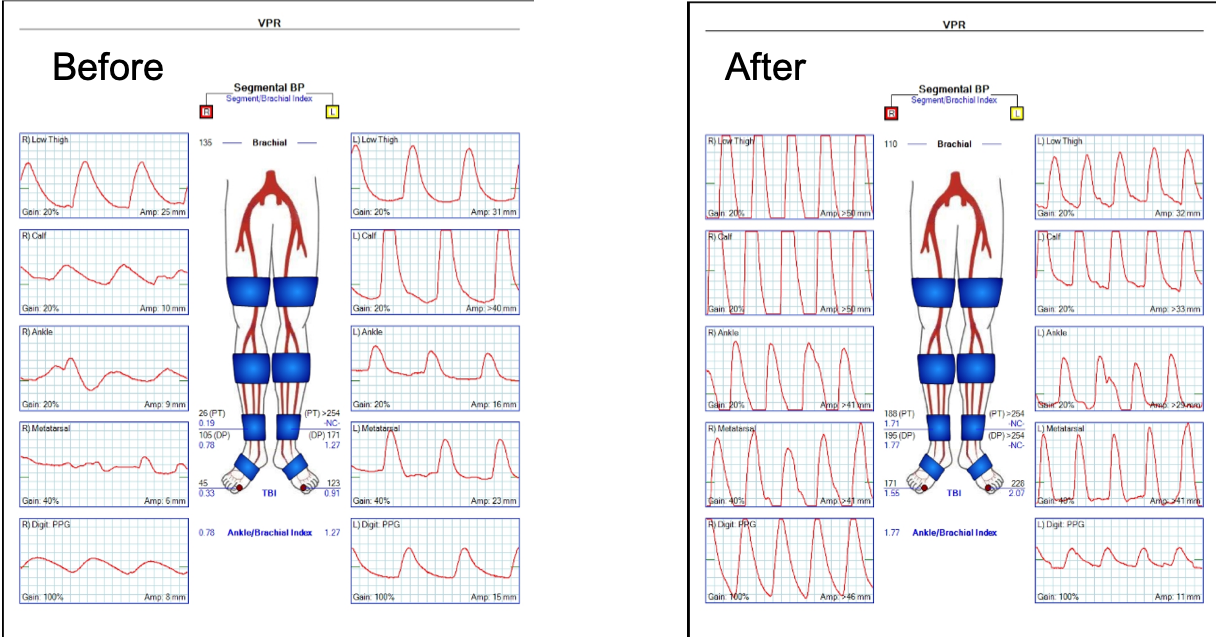
On examination, she was a large woman with no femoral pulses, but signals could be obtained in her popliteal and tibial arteries. Her PVR’s showed inflow disease and poor flow at the feet.

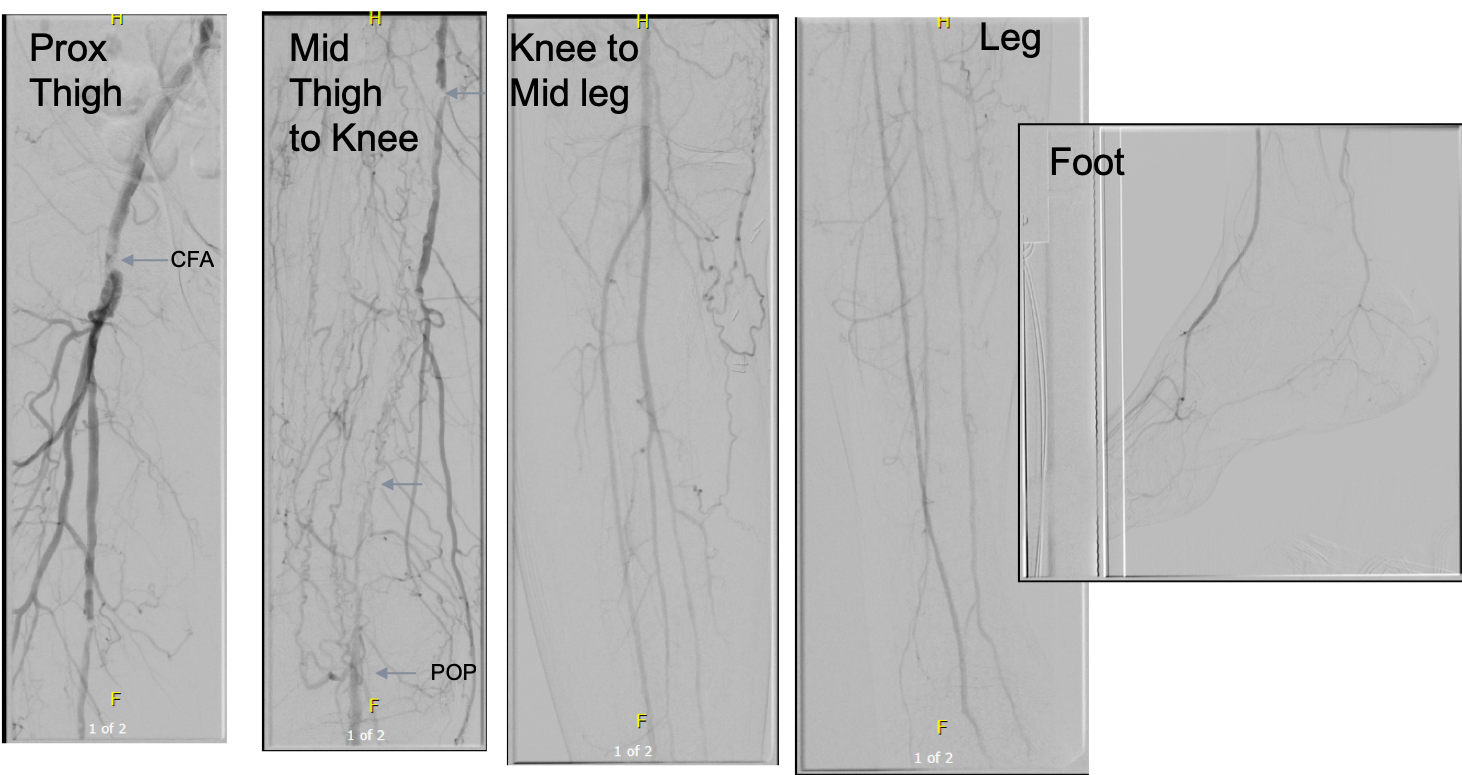
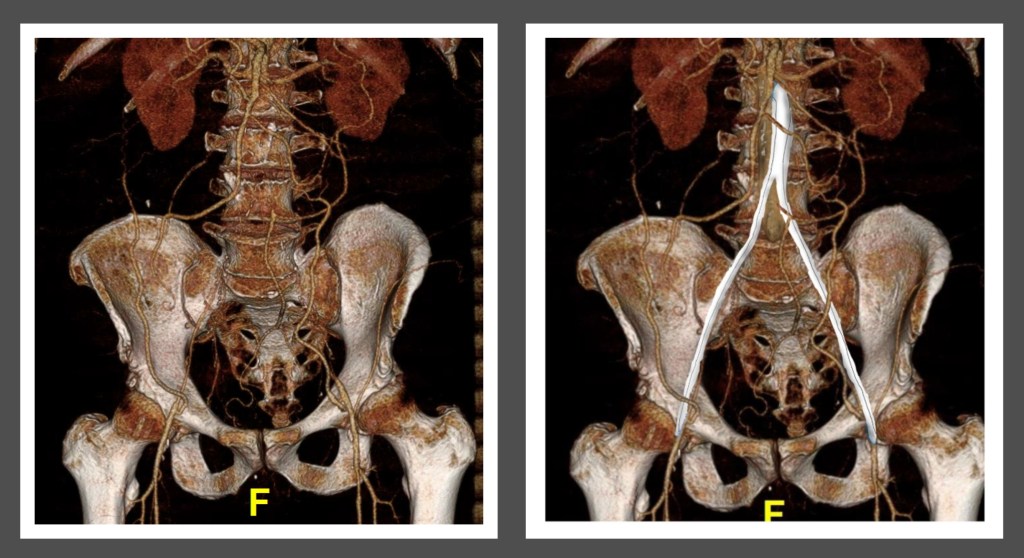
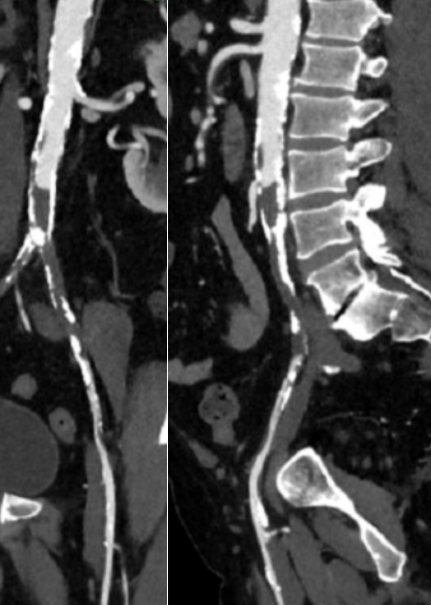
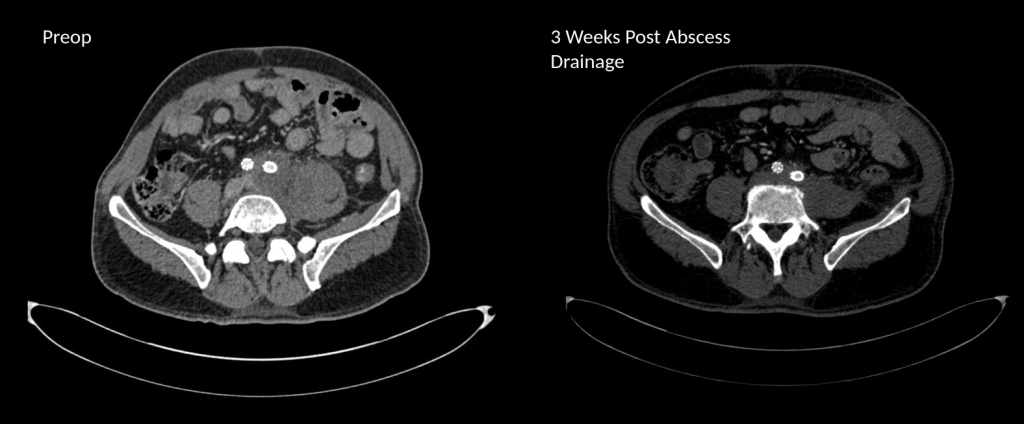
Her baseline CTA in workup of her claudication the year prior to getting stented shows the aorta and iliacs, while open, are small, with aortic lumen diameter reaching 10mm and common iliac lumen diameter at 6mm with diffuse atherosclerosis (below).

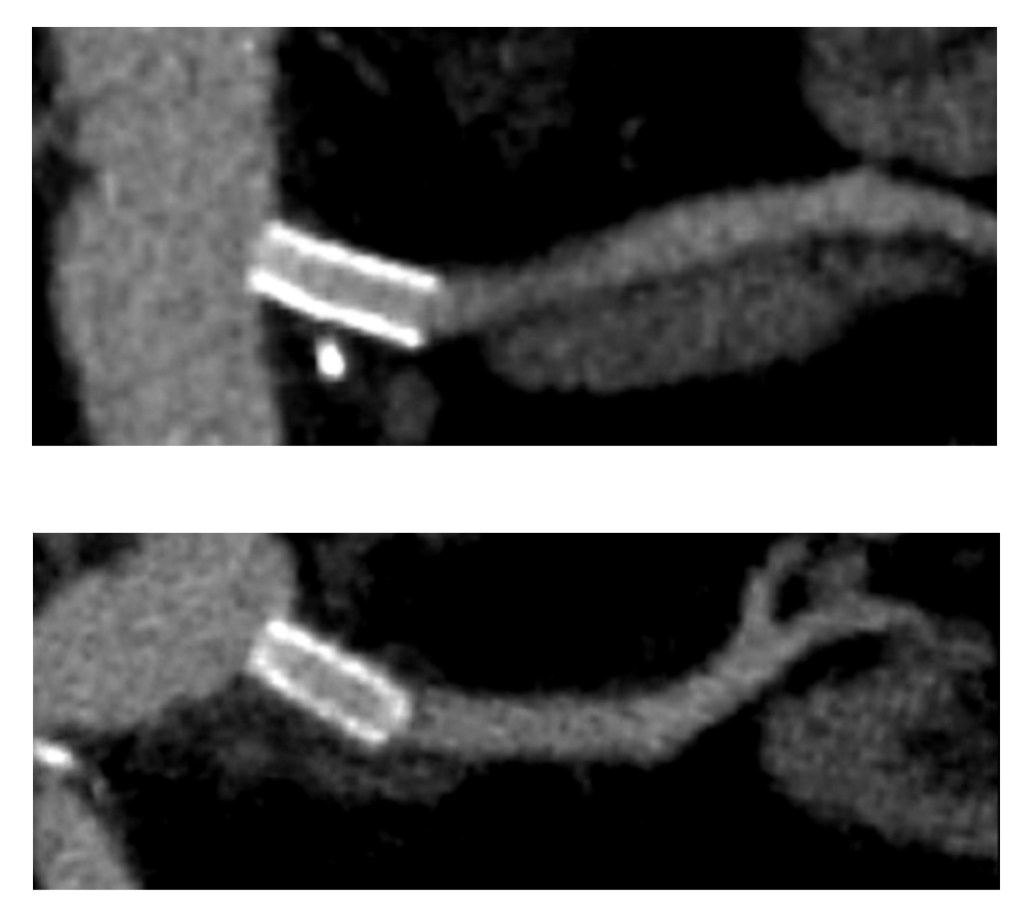
The CTA on presentation shows bilateral stent occlusion. A closer look shows the second set of stents extending the original stents both proximally into the aorta (raising the bifurcation) and distally into the external iliac and across the internal iliac origins (white arrows). The internal iliac arteries, despite the stents and on the right thrombus in the stent, supply flow to the external iliac arteries which have not thrombosed.


The treatment options were
- Exercise and risk factor modification
- Reintervention
- Axillo-bifemoral bypass
- Aortobifemoral bypass
- Aortoiliac endarterectomy and patch angioplasty, stent removal
Although exercise and risk factor modification should be part of the treatment regimen, the best time to institute this was before her first intervention. With the long segment occlusion of her stents, coverage of the right internal iliac artery and occlusion, and acuity of her symptoms, this is no longer feasible.
It reveals a certain kind of bias when we prescribe walking exercise to those who can’t afford gyms or equipment, and whose neighborhoods are unwalkable.
Reintervention, having failed once, will not be durable. Even with anticoagulation, any recanalization -thrombolysis, thrombectomy, balloon angioplasty, atherectomy, lasering, and restenting, would not be durable.
It is likely the patient is frequently vasoconstricted and this is exacerbated by smoking. While never diagnosed with Raynaud’s, she did give a history of easily have numb, cold fingers and toes in the chilly winters in Cleveland. Even normal spectral Doppler signals will show pauses in flow in the peripheral arteries. Combined with any hypercoagulability and injured lumenal surfaces from interventions, and stents will go down.
An Aside on Small Aorta Syndrome in Women
One of the advantages of being a village elder is you remember forgotten concepts that guided treatment “back in the day.” The small aorta syndrome defined as having an aorta smaller than 12mm in diameter is one of those. Best described as not having enough pipe -imagine a small caliber fuel line throttling an engine. For all the muscles involved in standing and walking, there is a minimal diameter necessary for function.
Small aorta syndrome stands up to objective testing. A patient with a small aorta but otherwise patent lower extremity arteries, can present with claudication and demonstrate drops in ABI with exercise. These are typically female smokers with elevated BMI. Along with their small aortas, their external iliac arteries will be small, and I used to wonder if some critical period of inactivity in their early years failed to grow these arteries, or if this process of normal growth and remodeling is retarded by smoking.
Small aorta was a common indication for aortobifemoral bypass (ref). Unlike some abandoned indications for operation like “4.5cm AAA” and “asymptomatic 60% internal carotid artery stenois,” it had a testable finding of drop in ABI with exercise, but its acceptance has waned in the advent of the endovascular era. In a purely open era, I think there was greater emphasis and awareness on engineering the hemodynamics. While endovascular interventions simplify treatment, just stenting a small arteries usually doesn’t fix the problem as illustrated in this case. That is because there is a maximum size that the arteries receiving the stents will allow.
The iliac artery and aortic bifurcation will only tolerate so much upsizing with stents before rupturing. The interventions are constrained by the size of the adventitia. What is also ignored is the concept of elasticity -the 7mm lumen through a reopened and restented artery provides more resistance to flow than a 7mm artery restored via endarterectomy. All stents decrease elasticity of the circuit and decreases flow in a pulsatile circuit because of the increased impedance. Bovine pericardial patches on the other hand add elasticity. Endarterectomy restores elasticity. .
Enough Pipe
In the early 2000’s, I used to live in an pre-war apartment in Riverdale down the hill from Drs. Takao Ohki and Frank Veith. The apartments above and below me all shared this same feature -poor water pressure, because during a restoration twenty years before, the owner used the wrong, smaller size of pipe for this line of apartments. The taps would run, but if more than one apartment ran the shower or dishwasher, the taps would drip. The apartments would claudicate. The pipes were all patent, but inadequate. Not enough pipe. This patient endowed with small vessels, grew a large body, and smoked, and her muscles needed more pipe to support the added load. Not enough pipe.
So is the solution an aortobifemoral bypass? It is the board answer and a durable one, but it shares with axillobifemoral bypasses the risk of groin infections, particularly with a large body habits (below). The outflow arteries, all patent, are small and likely subject to vasoconstriction. My choice of ABF graft in this patient is a 14x7mm bifurcate which is on the small side, but I would be afraid that a 16x8mm graft would be too large on both the aortic and iliac side, resulting in mural thrombus formation.


Axillo femoral bypasses, aside from the groin issues, suffer from poor long term durability and is not a great choice for a 40 year old. Her axillary artery was 6mm and sourcing flow to the lower torso from that is never great. Also, supplying a long 8 or 10mm graft would recapitulate the original problem of a small aorta. Not enough pipe.
For me, the best option would be to remove the stents and restore the distal aorta and iliac arteries to their original elasticity and slightly larger than original diameter. I would then be able to reopen flow to the occluded right internal iliac artery. Not just enough lumen, but enough and correct pipe.
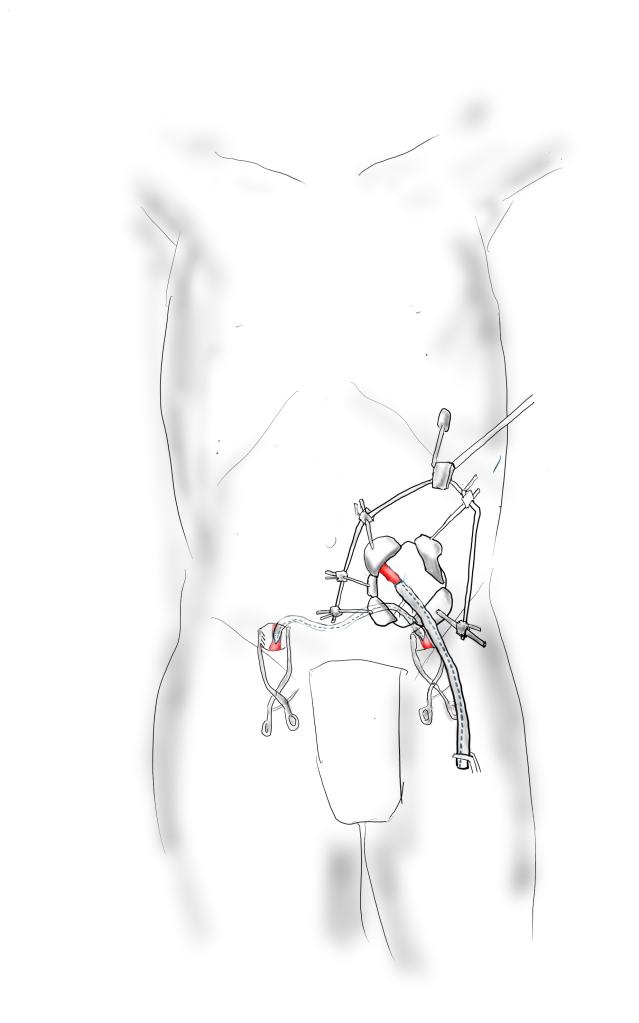
Technique: Exposure
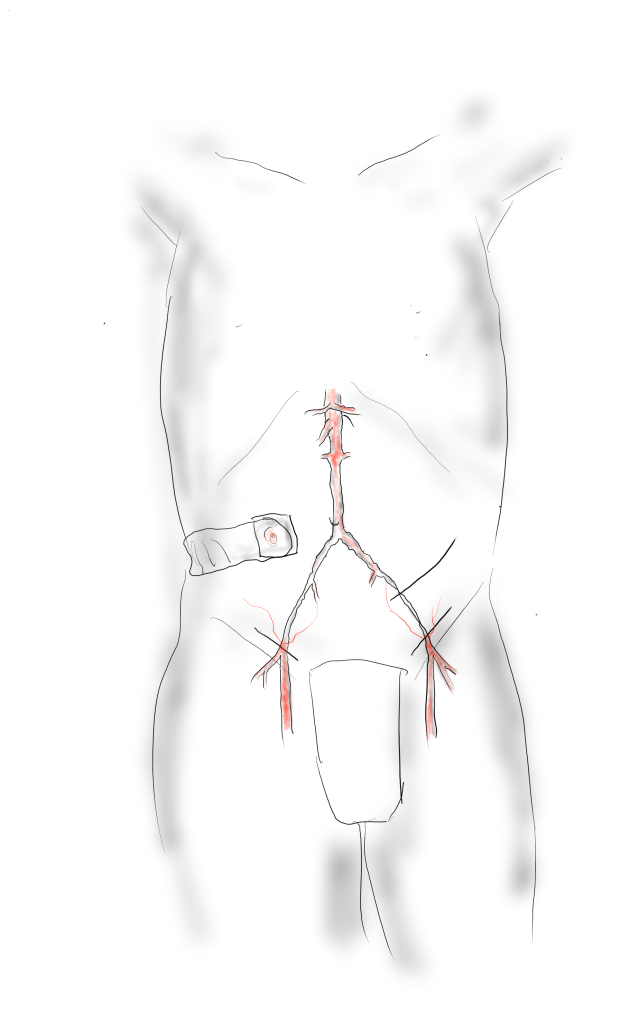
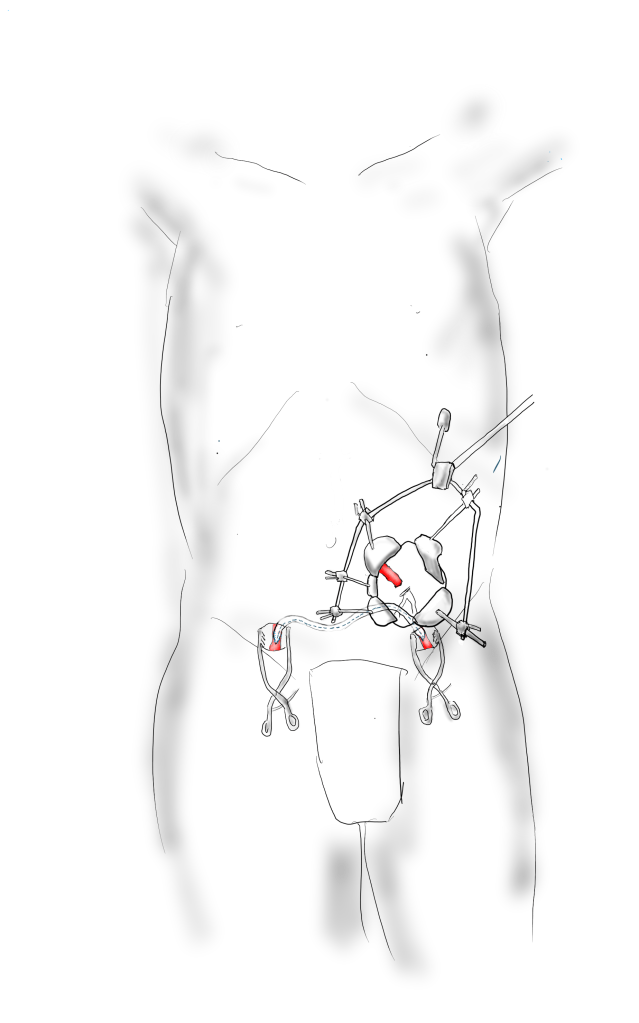
Exposure is predicated on the planned extent of the endarterectomy, place for clamping, and plans for aortobifemoral bypass if the endarterectomy results in poor adventitia. In a woman, the iliac bifurcations are easier to reach. A midline laparotomy is the incision of choice here. Let me digress here about the laparotomy. Over the three decades since the launch of laparoscopic surgery and subsequently endovascular surgery, the midline laparotomy has gotten an undeserved bad rap. Laparotomies are well tolerated and should not be viewed as a rare bailout or outright failure of laparoscopic therapy. Rather, it is still the gold standard exposure.

The infrarenal aorta to the right external iliac artery is exposed as well as the common iliac. In this patient the sigmoid mesentery was fatty and did not readily expose the iliac bifurcation so a separate exposure of the distal left common iliac artery was performed by mobilizing the left colon.
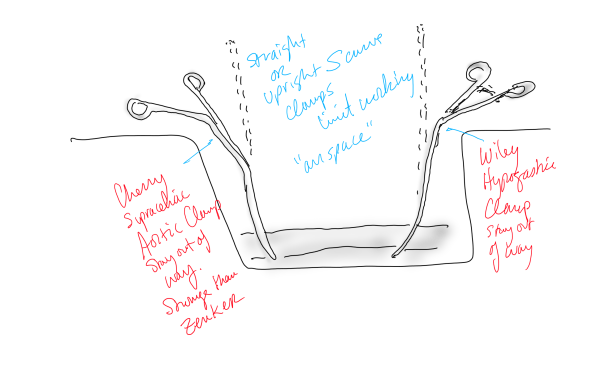
The aorta above the bifurcation was prepared for clamping. This involves circumferential exposure as I prefer a transverse aortic cross clamp. The lumbar arteries are clamped with bulldogs or aneurysm clips. The right external iliac well beyond the stent is controlled and the internal iliac is exposed and controlled. On the left the internal and external iliac arteries are expose and controlled.
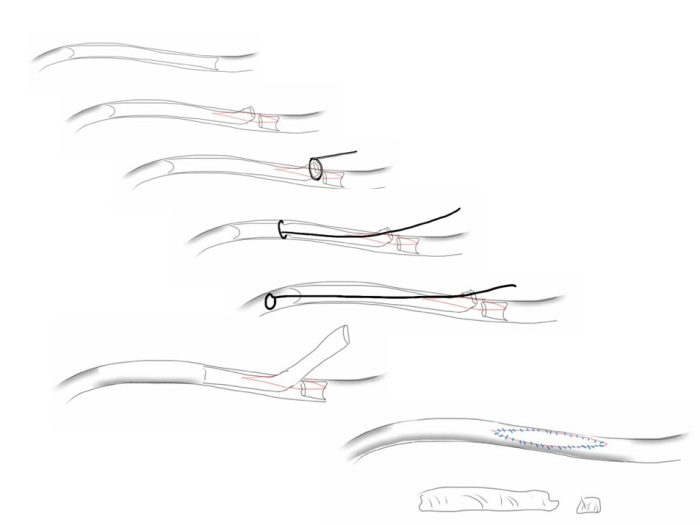
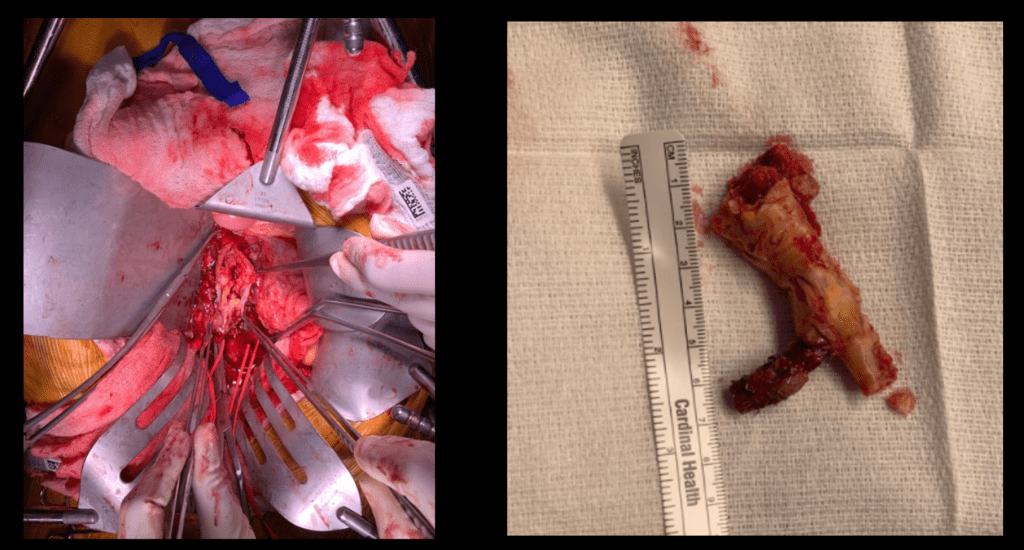
The patient is heparinized clamps applied, and I make the arteriotomy with a 15 blade cutting down to the stent. The aorta is cut to a point about a centimeter below the clamp. The external iliac is cut to where there is patency of the artery and the plaque is mild. The endarterectomy is performed in the same way one does a carotid or femoral, with care to find the correct endarterectomy plane outside the plaque and good end points where the plaque adheres well. The internal iliac plaque on the right was chronically occluded but was successfully removed via eversion resulting in back bleeding. I sound the artery with a dilator to make sure a dissected plaque isn’t occluding it then reclamp.

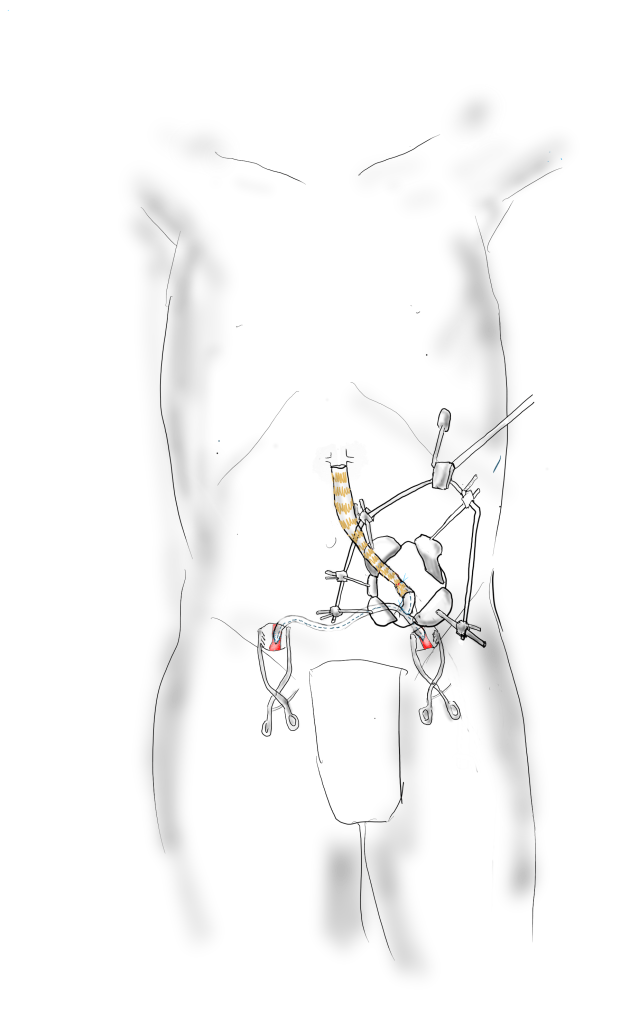
The left common iliac artery is opened via a separate arteriotomy as I find tailoring a Y-shaped patch laborious. The arteriotomy is extended under the sigmoid mesentery and then moving the left colon medially the arteriotomy is finished slightly beyond the external iliac origin. The endarterectomy is finished short of the iliac bifurcation and any narrowing at the bifurcation is treated with the patch.
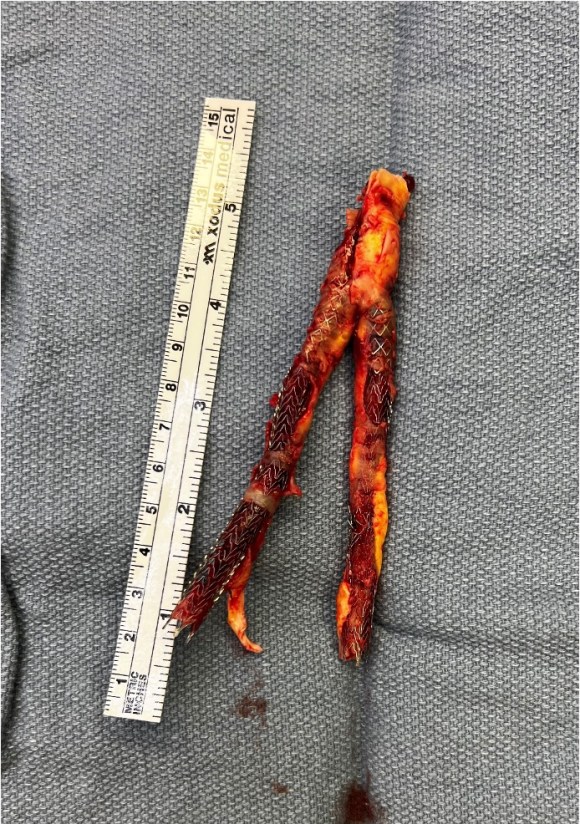
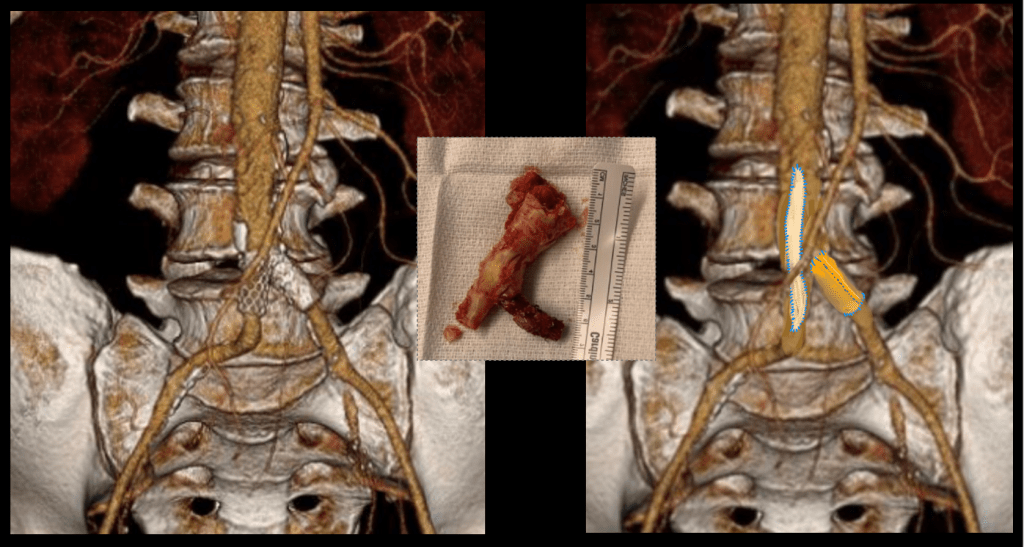
The specimen shows that the adventitia remains separated from the stents by the plaque. I rarely use tacking sutures as I feel a properly performed endarterectomy results in no plaque or well adherent mild plaque.
The patch needs to be thoughtfully applied. An overly large one will billow, and at worse case, create an artificial aneurysm. For example, for a 7mm iliac artery, the circumference is 22mm. Adding an 8mm patch with 1mm suture bites results in a 26mm circumference and an 8.3mm end diameter. Narrower is okay, but much larger will result in size mismatch that the body compensates for by laying mural thrombus. A long 8mm wide patch can be cut from a large swatch of bovine pericardium, remembering to add a slight angulation onto the iliac artery.

This operation avoids the groins with exposure or access into a small artery with a large sheath. A 4mm artery with 12.6mm circumference receiving an 8F sheath receives a 2.6mm hole, a 40% defect across the anterior hemicircumference of 6.3mm. This is not trivial, particularly because the arteries are often atretic after prolonged occlusion, may tear with a closure device, most of which are off IFU for such a small vessel. Avoiding the groins altogether is a great benefit to this type of procedure.
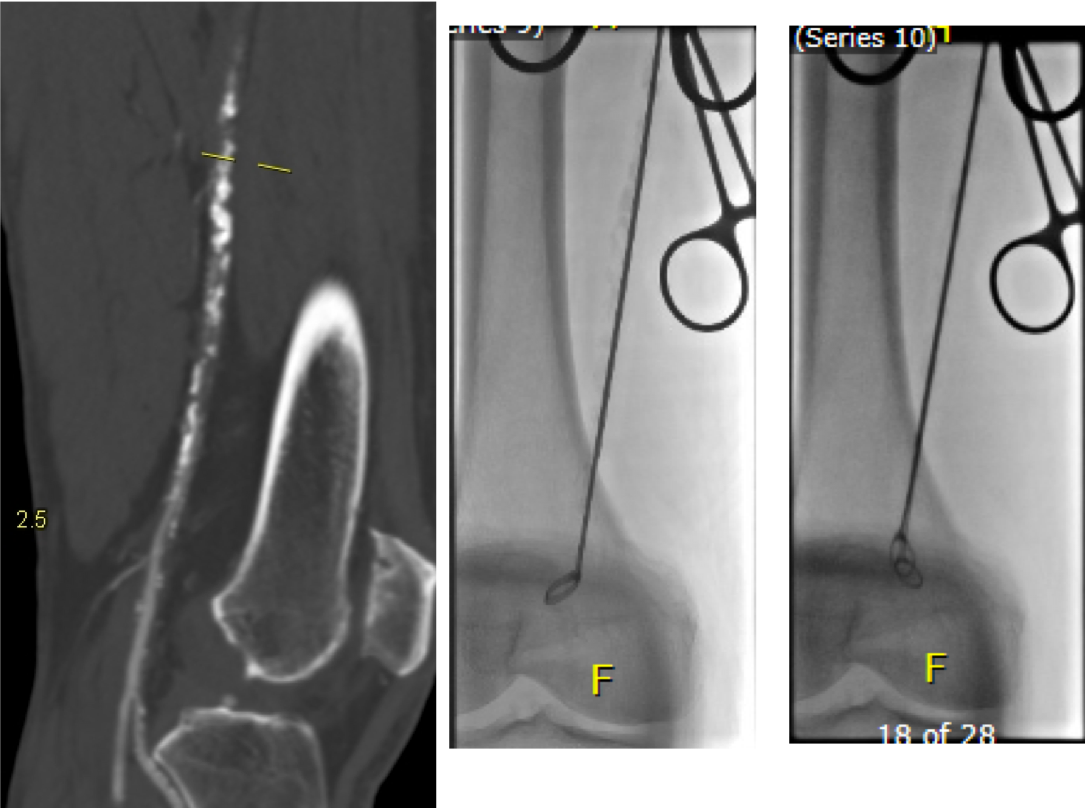
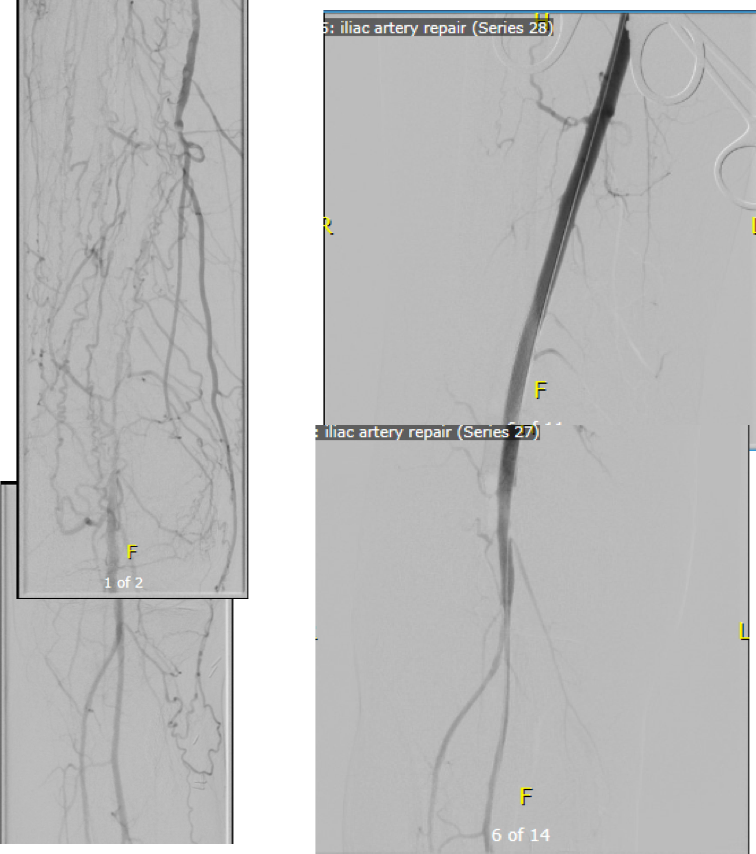
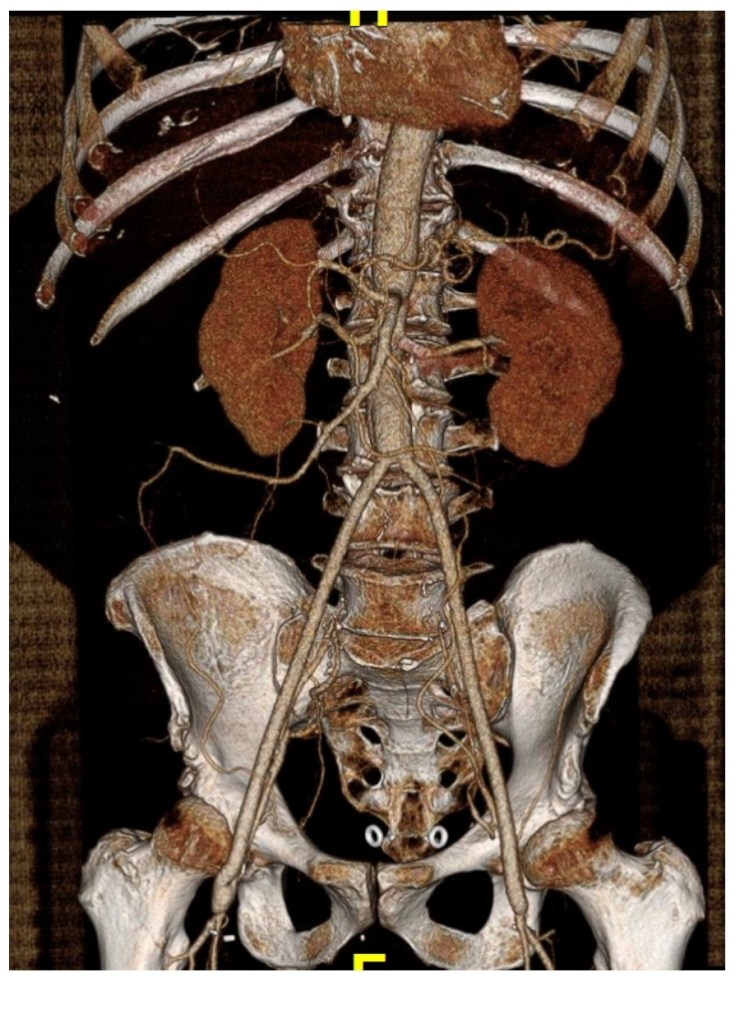
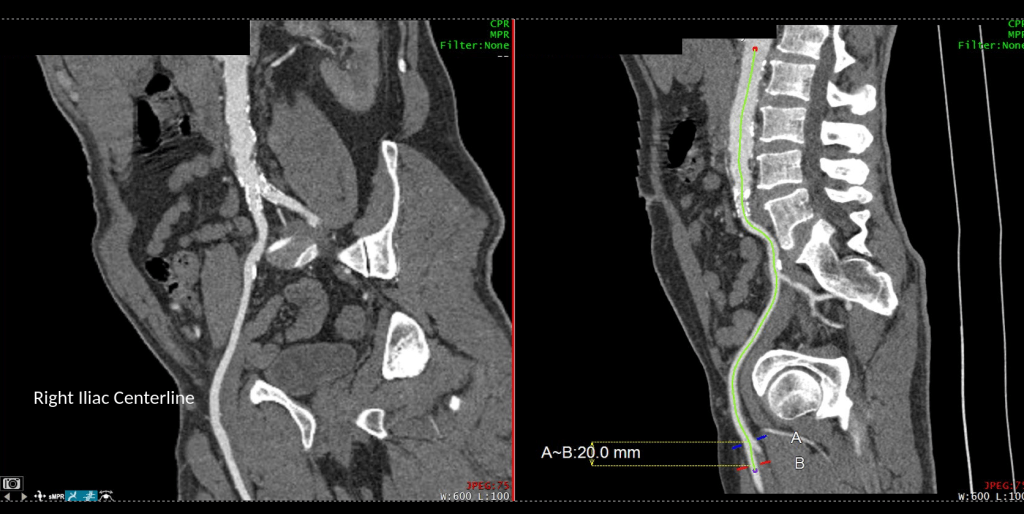
A postoperative CTA showed wide patency of the restored aorta and iliac arteries.

At followup several months after the procedure, the patient was walking well without claudicating and was ready to return to work. PVRs showed excellent flows down to the toes.

Hemodynamic Engineering
A surgical trainee has to develop a sense for flow. Looking at a circuit, she has to ask “how does blood get from point A to point B?” Merely providing a pipe does not mean a cure. For example, replacing a blocked artery with a steel pipe would provide flow but it has a hemodynamic impact that is different from the native vessel. Flow stoppages during the cardiac cycle that is modulated in a normal artery by the elasticity of its wall. While we don’t deliver steel pipes, we do something similar in ballooning heavily calcified arteries, or stenting them. ABF with prosthetic bypass offers a safe, broadly available method of treating this, but is fraught with problems for patient who develop groin infections or occlude a bypass for the reasons previously mentioned. Endarterectomy and patching with bovine pericardium allows for more precise restoration which would be a laudable goal in a young patient.
And my final point is this. This patient can yet undergo aortobifemoral bypass. Ironically, even larger stents may be safely placed than was previously possible. One of the principles laid out by Dr. Jack Wylie in his peerless surgical atlases was of leaving a patient in a condition to allow for future necessary operations. For a forty year old patient with many decades left, this is a critical concept.
This case represents the second aortoiliac endarterectomy I performed to remove failed stents. The third just happened today with resection of failed CERAB stents which I did with my chair and fellow Mayo Alum, Dr. Jae Cho. I think that there is a room for this operation which should not remain in the history books.
For a video presentation from Dr. Pat O’Hara: https://vascsurg.me/2022/10/15/aortoiliac-endarterectomy-removing-occluded-stents-is-possible/
References
Cronenwett, JL, Davis JT, et al. Aortoiliac occlusive disease in women. Surgery 1980; 88:775-84.

























 If you work long enough, you will not only see everything, but you may end up doing something that you say you would never do. You will be confronted with a scenario that would test not just your skills but also your boundaries. The adage, never say never is a warning that all of us may face a choice -to remain rigidly consistent with some earlier proclamation or to excuse a little hypocrisy for the sake of the patient.
If you work long enough, you will not only see everything, but you may end up doing something that you say you would never do. You will be confronted with a scenario that would test not just your skills but also your boundaries. The adage, never say never is a warning that all of us may face a choice -to remain rigidly consistent with some earlier proclamation or to excuse a little hypocrisy for the sake of the patient.