It is a rare day that passes without the announcement of a stroke alert at CCAD. A reflex arc of activity is initiated, as time becomes the critical metric of success. Patients with strokes have a limited window of time to reverse the effects of the arterial occlusion, and the whole hospital is organized around getting the patient into the angiographic suite to open up blood vessels. If you watch it happen, it is the pinnacle of modern medicine, to achieve what only a decade ago was deemed unachievable. It was built around a foundation laid by cardiologists for heart attacks -the STEMI alert. The teams practice like racing pit crews with a stopwatch to get a patient from the emergency room, to CT scan, to angio suite. A long time ago, as a young surgeon, I had to work hard to get institutional support of ruptured AAA and cold legs. Vascular surgery has traditionally struggled to get recognition for its patients, their diseases, and its work, which is nothing less than the most important safety net for any large general multi-specialty hospital, critical infrastructure like oxygen plumbing and backup generators. As I transition to that weird designation of mid-career surgeon (please don’t call me a senior surgeon), I have also appreciated that Steve Jobs aphorism about good artists copying, great artists stealing. It’s only stealing if you don’t give credit. Here is what I borrowed from the neurologists.
Acute mesenteric ischemia is an abdominal stroke. Use it in your conversations with other people as you speed your patients way into the angio suite. The reflex arc is in there. For the emergency department, the operating room, and all the physicians, acute mesenteric ischemia sounds like tummy trouble, but abdominal stroke brings sudden clarity to conversations like:
“Well, you’re in line behind a gallbladder and a cystoscopy. Is the patient NPO?”
Me: “It’s an abdominal stroke. We literally only have a few hours before the patient dies…”
“I’ll bring the backup team in!”
The patient is a middle aged man with risk factors of NIDDM and prior history of DVT who developed severe mid-abdominal pain at 5pm. He came to the ED at around 11pm and had a general surgery consultation who ordered a CT Angiogram showing SMA occlusion (pictured below).
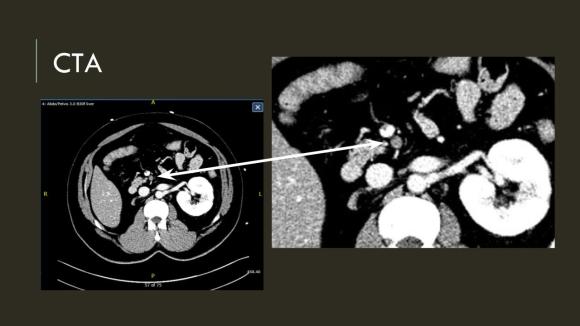
Heparin was started, and at 11:30, vascular surgery was consulted. The patient had a soft, doughy texture to his abdomen, but great pain with palpation -classic pain out of proportion to the exam. Determining the patient to have acute mesenteric ischemia from a thromboembolism, I took the patient to our hybrid angiographic OR suite with the plan for arteriography, possible open thrombectomy, and exploratory laparotomy.
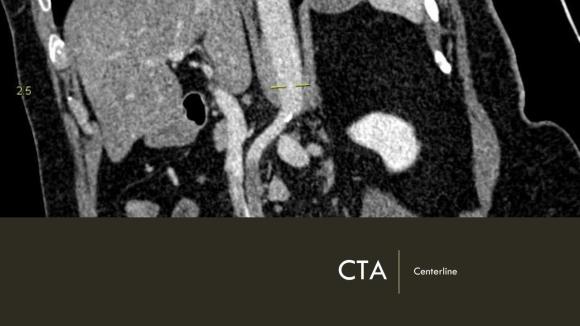
Arteriography from femoral access showed an occlusion of the SMA beyond the middle colic artery, a typical pattern for an embolism that occurs when embolism lodges distally and propogates proximally (image below).
I got Glidewire access into the ileocolic terminus of the SMA, exchanged for a Rosen wire, over which I placed an 8F sheath into the proximal SMA. This was a rather large sheath meant to catch thrombus as I suctioned it out with a 6F Penumbra catheter. This is another technique I borrowed from the neurointerventionalists. Whenever a stroke alert is going on, curiosity drives me to peak in and see what marvelous gadget or gewgaw they are using, and I was impressed by how efficiently the neurointerventionalists were able to get to the smallest thrombus in the furthest branch vessels. I was prepped for open thrombectomy, consented for bypass if necessary, but having experience in suctioning clot through single catheters and sheaths, I thought the simple design of the Penumbra and its efficacy in the cerebral system could easily translate into the mesenteric.The problem with open thrombectomy is the inability to see if you have cleared thrombus from all the branches unless you do an arteriogram after you’ve completed your procedure. This may be a significant contributor to the 20-30% bowel resection rate that occurs on second look laparotomy in my old paper and in the literature since its publication.
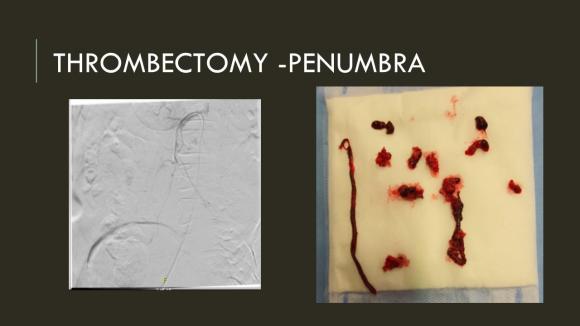
The Penumbra was effective in removing much of the fresh thrombus, but I was also cognizant of the fact that pulling out the catheter will draw clot into the 8F sheath that did not make it into the catheter. I placed a wire, and removed the sheath to expel much of the bulky thrombus (picture below).
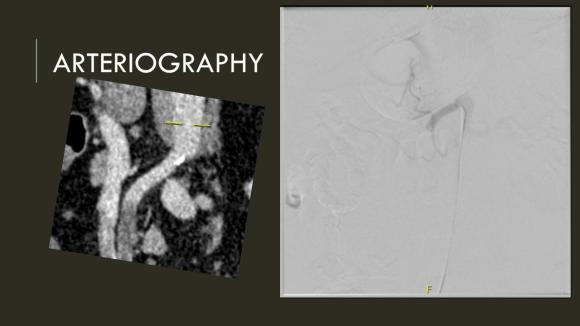
The completion angiogram (below) doesn’t show the intermediate angiograms showing thrombus that embolized to other arteries as I manipulated the catheters and thrombectomized -I was able to successfully retrieve these with selective catheterization, another neurointerventional series of maneuvers that I have successfully borrowed.

After being satisfied with the completion, I removed the sheaths and explored the abdomen finding this segment of infarcted small bowel (next image).
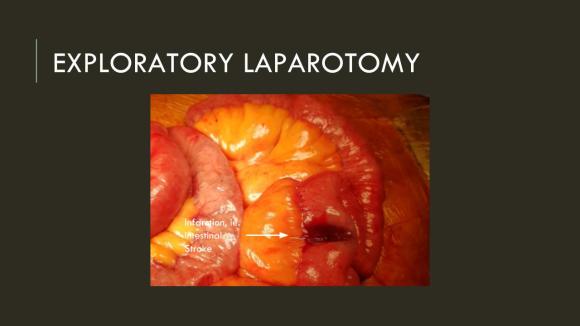
There was no question in my mind that there would be some dead bowel based on the time course described by the patient. Despite my excitement about calling for GIA staplers -I am general surgery boarded- I called in the general surgeons for their help in resecting and anastomosing this segment of bowel. They would be the ones taking the patient back for any second look laparotomy, although in this patient, I determined that there would likely be no need. After the anastomosis was completed, I did a Wood’s Lamp examination (pictured), which is accomplished with a black light after giving the patient an ampule or two of Fluorescein.

The bowel had a splotchy fluorescence pattern which is typical of ischemia-reperfusion. This is where you have to ask the anesthesiologist and any critical care specialist who follows -no pressors please! Edema won’t kill an anastomosis as badly as ischemia will, and the gut is as sensitive to norepinephrine as are the toes. Workup in the hospital including echocardiography and CTA of the entire aorta failed to reveal a proximal source or cardiac shunts or thrombus. The patient recovered and has recently followed up, eating well, and tolerating his anticoagulation which he will be on for life.
I sent out the pictures to my neurointerventional friends with some glee, but also with the purpose of informing them that in the case that the vascular surgeons become incapacitated or quarantined due to the COVID-19 pandemic, their skills would be recruited in the care of an abdominal stroke -a blood vessel is a blood vessel.
Acute mesenteric ischemia should be the first thing on everyone’s differential of sudden onset abdominal pain because of its time dependence, yet it does not have the same resonance to the unfamiliar as abdominal stroke. Survival is dismal when too much time and intestinal death has occurred. When associated with the stroke alert concept, it translates into processes already in place throughout the hospital and it becomes natural for everyone to appreciate the urgency of treating abdominal stroke. This is the system adopted by Roussel et al. in France, where they have regionalized care of intestinal stroke. They report mortality rate of 6.9%, which is in a selected population, but significantly lower than the traditionally reported 30-60% mortality.
I am still an advocate of an open approach, especially when angiographic resources are unavailable, and every trainee needs to be able to describe the exposure of the SMA, and management of acute mesenteric ischemia. Hopefully, everyone will appreciate the urgency of all the various ischemic conditions manifest in the peripheral circulation, but rebranding them as a stroke (leg stroke, hand stroke, intestinal stroke…) is helpful. Finally, there is no survival with dead bowel -it must be found through exploration and resected.
Reference:
Roussel A, Castel Y, et al. Revascularization of acute mesenteric ischemia after creation of a dedicated multidisciplinary center. J Vasc Surg. 2015 Nov;62(5):1251-6. doi: 10.1016/j.jvs.2015.06.204.

