Our debranch first technique described by Dr. Niranjan Hiremath and was presented at CX Aortic in Vienna in October. Hoping to collect multicenter experience with this technique.
Category: tbad

Context
Despite all the advances in endovascular repair of thoracoabdominal aortic aneurysms, no data shows their superiority in the mid to long term compared to open repair. That is why branched stent graft programs occur hand-in-hand with robust open surgical programs, to offer durable open solutions to younger healthier patients while mitigating risk in older sicker patients by going with a branched or fenestrated device. Despite these advances, clamp time and visceral ischemia persists as a challenge to safe performance of open thoracoabdominal aortic surgery. Even a straightforward group IV requires the surgeon to be swift. Time on an thoracic aortic clamp results in visceral ischemia with a predictable response of coagulopathy, acidosis, systemic inflammation, and renal insufficiency. Adding cardiopulmonary bypass mitigates some things (distal ischemia, normothermia, hypertension) but brings on other complexities (cannulation, circuitry, coagulopathy). Sewing to a Carrel patch allows one to perform one large anastomosis rather than four individual ones, gaining speed and time, but compromises by leaving aneurysmal tissue which could progress to a troublesome patch aneurysm. TAAA is a condition that demands referral to high volume centers. It is in high volume centers that these apex predator surgical conditions can be subdued. It is in these centers that branched/fenestrated stent graft programs can offer treatment for patients high risk for open repair. It is in these centers that patients can avoid compromises such as snorkels and chimneys. Unfortunately, these centers are long air flights away and the means of the patients may not match the desires. Out here in Abu Dhabi, half a world away from Cleveland, Rochester, Houston, Boston, Chapel Hill, New York, and Seattle, and over six hours from major centers in Europe, we usually have to find our own way. Thankfully, we have the resources in experienced staff and abundant materiel.
During my time at the main campus of Cleveland Clinic, I came to appreciate the hospital as a highly evolved tool for healing, but for open repair of TAAA, it still falls on the operating surgeon and the choices made that shaped the outcome. In this most invasive of operations, simplicity and efficiency translating to speed offers the only consistent path to success. My partner, Dr. Houssam Younes, who trained in Houston, mentioned that Dr. Joseph Coselli, has clamp times approaching 30 minutes for group II TAAA -an almost inhuman speed achieved by doing these cases every day. For this reason, stent grafts are popular because repair of aortic aneurysms can be achieved by more practitioners in widely distributed settings. Because of the marketing of all things minimally being better, patients come asking for endovascular.
Case
The patient is a fifty-something smoker who had a prior type B aortic dissection nearly a decade past who presented with substernal chest pain radiating to the back. He was found on CTA (figure below) to have a 6.5cm extant V TAAA starting above the diaphragm and ending at the level of the renal arteries.

Closer inspection revealed it to be aneurysmal degeneration of the aorta at a large false lumen fenestration. The aneurysm had grown eccentrically into the patient’s right chest and retroperitoneum. The remaining dissection above to the left subclavian artery and to the aortic bifurcation was chronic and thrombosed. His pain waxed and waned with hypertension which initially had to be controlled with parenteral agents. His cardiac workup revealed normal ejection fraction and valve function, and no critical coronary artery disease on coronary CTA. Pulmonary consultation deemed him a low pulmonary risk for major surgery. I offered him open repair, and initially the patient balked, asking for an endovascular repair, but I carefully walked him through the concepts, principles, and data guiding my recommendation. Yes, in a rupture, I have stented and followed with visceral debranching (link) and we have placed multiple snorkels in a very high risk octogenarian with a rupture, but who would offer endovascular repair to an otherwise good risk 50 year old?
The operation was initially planned in the standard way with cell salvage, plan for clamping in the transition point where the descending thoracic aorta took a rightward turn. The sequence of operation was for proximal anastomosis, right renal anastomosis, cooling the left kidney, celiac axis (CA), superior mesenteric artery (SMA), left kidney, and finally distal anastomosis with reimplantation of any prominent intercostal vessels. Preop imaging suggested the one at the T12 level was large. CT surgery was asked to be available for cardiopulmonary bypass via left atrium and femoral vein. Cardiac anesthesia and I had a discussion about intraoperative monitoring and management. Plans were made for CSF drainage -despite recent papers suggesting as much harm as benefit from these drains, I still feel critical time is lost if the patient is ventilated for a prolonged period postop and motor evoked potential monitoring is not available. The culture of CCAD mirrors that of Cleveland Clinic’s main campus in Ohio, and collaboration is ingrained. It was also serendipitous that one of the clinical associates, Dr. Niranjan Hiremath, had a particular interest and training in aortic surgery and suggested something amazing.
The Game is Changed
Dr. Hiremath trained in both vascular surgery and cardiothoracic surgery in Melbourne under Dr. Matalanis. Drs. Matalanis and Ch’ng published a series of 5 patients done with a separate branched bypass to the visceral vessels fed from the cardiopulmonary bypass circuit (reference). A separate aortic bypass was then performed and this truncal visceral graft was anastomosed to the main aortic graft. It was a visceral branch application of what is commonly done for the aortic arch great vessels. It was clear to me that this concept eliminated the need for desperate speed, and minimized clamp time to the 5-15 minutes required for each visceral branch anastomosis. A game changer.
My process for incorporating new concepts to an operation requires comfort and familiarity. The familiarity with this modification had everything to do with my extensive use of shunts during peripheral bypass surgery. Placing the shunt into one of the renal branches of a 4 branch Coselli graft turns this graft into a live blood vessel. By fixing the proximal and distal ends of the graft in the correct orientation and position relative to the branches, each of the visceral branches could be anastomosed to the graft one at a time while the patient’s aorta remained unclamped. This is best described with the sketches I put together for the preop huddle (gif and figure below).


Even though I have done many of these operations, because of the smaller population at risk, thoracoabdominal aortic aneurysms are a relatively low frequency disease and no assumptions can be made. The fact was, it took very little convincing for me to understand this concept as a game changer, but I took the liberty of modifying it for the particular situation of the patient. Every operation is ultimately another quantum of experience for the people involved to take away priceless information for the next one which will always be unique. We must take these as opportunities for improvement. For once in a long time, I felt this would be a big improvement.
Day of Surgery
Our operations always start with a huddle, but this one was different because of the numbers of people involved. There were the cardiac anesthesiologists, some of the best I have ever worked with in my career led by Dr. Dominique Prudhomme. The cardiac surgeons, Dr. Tareq Aleneiti and Hiremath, who planted this idea, along with the perfusion team, cardiovascular nursing team, and my partner Dr. Younes walked through the steps of the operation with me, with bailout points and plans B and C (CPB with hypothermia and circulatory arrest as a last resort for any unforeseen uncontrollable bleeding). I felt like I was the ringleader in a heist movie, only in this case, rather than jewels, we were grabbing something actually priceless (figure 3).

The patient was placed in the right lateral decubitus position across the table break in the golf backswing position with pelvis relatively flat to the shoulders which were upright. The incision extended from the top of the sixth rib into the abdomen. This dissection is really two -the thoracotomy and separation of the peritoneum from the retroperitoneum linked by the takedown of the diaphragm and cutting of the costal margin. It is a tactile portion of the operation -only the hands really understand when to pull down and separate the two layers. The celiac and SMA are entangled in myenteric plexus which had to be cut to exposure sewable lengths of both. The left renal artery was also tricky in that the prior inflammation of the dissection resulted in stickiness of the tissues. It goes quickly and we have this exposure (figure 4).

Heparinized
The patient is heparinized with a goal ACT around 250. Not enough for CPB, but good enough for rock and roll. The cardiac surgeons placed a cannula within a double ring of advential sutures, cinched with Rummel tourniquets and secured with an 0 silk tie -a maneuver I did many times myself during my cardiac rotations in residency. This was placed on a Y connector, one branch going to the CPB pump in case of a need to go to plan C, and the other to a tubing connector inserted into the right renal artery branch of the Coselli graft -a slight modification of the original plan (figure 5).
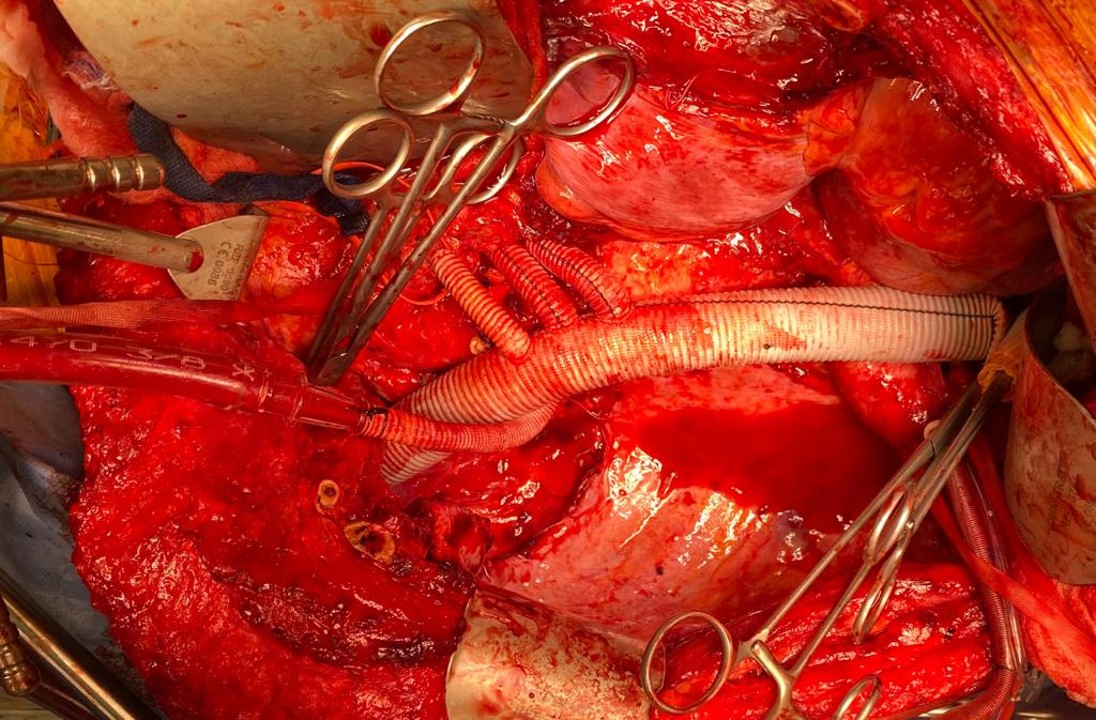
The other branches were simply clamped and the aortic ends of the graft were rolled up with a straight Kelly and secondarily clamped with aortic clamps -this gave weight to the ends letting them be positioned in a way that kept the branches oriented properly. There is need for precision and prediction as everything rotates back 45-90 degrees and the viscera sit on the grafts and the anastomoses. That is why keeping some length is crucial -this length accommodates this rotation. The Coselli graft is opened to systemic pressure by releasing the shunt. The proximal and distal ends of the main graft are twisted and double clamped to position the graft branches in the correct radial and z-axis orientation.
The aortic and branch exposure with takedown of the diaphragm is a standard exposure. One technical difference for this procedure is the need to expose about 2cm of the CA and SMA. Typically, only enough to clamp the vessel is necessary in standard surgery as these vessels are prepared with aortic buttons or anastomosed as an island patch. These vessels are surrounded by myenteric nerve plexus which feels like fascia or scar tissue but can be divide. Use of a hook cautery typically used in laparoscopic surgery along with a Ligasure speeds dissection.
These arteries, starting with the left renal artery, are ligated at the origin and divided for end to end anastomoses to the Coselli graft branches. As these arteries are only briefly clamped for the anastomosis while the remainder are getting flow from the aorta or the shunted Coselli graft, visceral ischemia is minimized. I still chilled the kidneys with manual injections of cold Ringer’s Lactate via large syringes and Stoney injector tips. The final product is shown on figure 6.

The operation no longer felt like a sprint. The atmosphere was lively and relaxed -something that does not happen in these cases even at closing as exhausted residents or fellows focus on stitching together all the separated layers of the patient’s chest and abdomen. I played a soundtrack of classic Bollywood tracks, alternating between mellow and lively.
What was striking was the absence of the need for blood transfusion -about a liter and half ended up in the cell saver, and the patient received 2 units of plasma, out of tradition. The patient had a minimal brief plasma lactate elevation which did not persist. He was closed with a chest tube and brought to the cardiac intensive care unit, stable, not on pressors. He was extubated that night, and moved all of his limbs to command. The chest tube was removed on POD#2, and he left the unit to recuperate on the floor. Amazingly, his serum creatinine did not rise significantly. Prior to clamping he received the usual cocktail of mannitol but it was likely unnecessary. Most of these patients, even with revascularizing first after the proximal aortic anastomosis in the fastest of hands, there is at least 30 minutes of ischemia manifest postoperatively as a rise in the creatinine with recovery in most. This rise was brief and transient (graph).
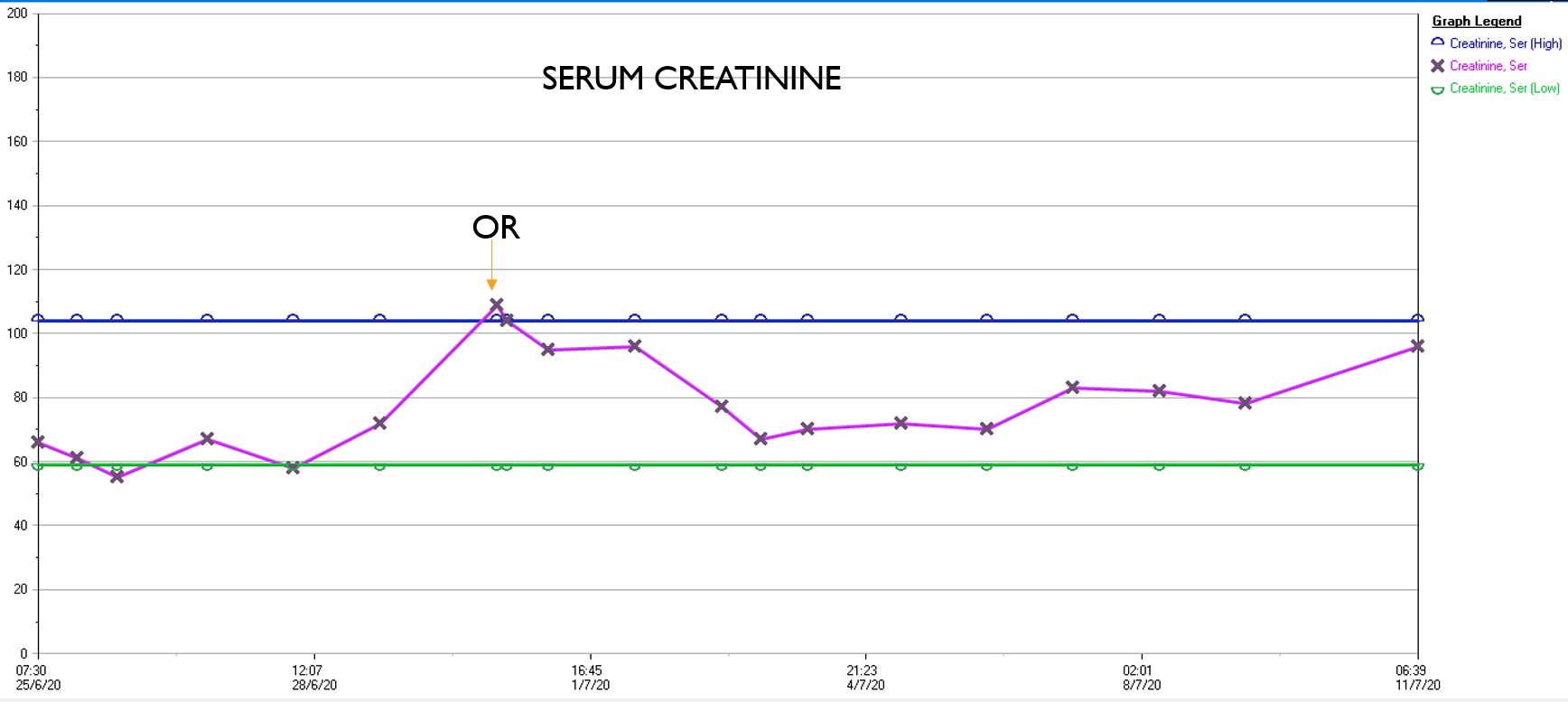
In the visceral circulation, the ischemia in the normal open repair is manifest postoperatively as systemic inflammation requiring pressors, persistent lactic acidosis, and coagulopathy, which at best is transient but at worst, fatal. This patient had no significant shift in any of these parameters. He had his CSF drain removed POD#2 after clamping for 24 hours, and was discharged home POD#11, having to recover from right chest atelectasis and a blood patch placed for persistent headache. CTA prior to discharge showed a good result. The left renal graft had been on stretch but was rendered redundant on repositioning of the viscera. No stenoses were noted.

Aftermath
Of course we are writing this case up, but case reports by their form cannot be overly enthusiastic whereas on my personal blog I can be excited. The normal course of postoperative recovery, the ebb and flow taught in surgical critical care books, is a result of ischemia, blood loss and replacement, fluid resuscitation, and cardiopulmonary support. Add to that cardiopulmonary bypass and you get an additional hurdle for the patient to recover from. This technique of shunting reminds me most of the temporary axillofemoral bypass. When I was a fellow at Mayo, I assisted Audra Noel in taking an elderly patient with a 25% ejection fraction through open aortic surgery with nearly miraculous recovery largely by avoiding the factors that trigger the ebb and flow. This technique is easier but mandates a strong normal segment of thoracic aorta to serve as inflow, otherwise an axillary artery will need to be cannulated.
Spinal cord protection is made easier with this technique by avoiding the massive fluid shifts, the pressors, and the acidosis in a typical thoracoabdominal aortic aneurysm repair. The blood pressure and cardiac output were never seriously perturbed. Several large intercostals and lumbar arteries were encountered and they backbled so avidly, after the short operation that I really felt there was no need to revascularize them. The spinal drain was kept open only for a day, and kept another day clamped to ensure that it would not be needed before removing it.
If you accept that spinal cord ischemia is multifactorial, we had avoided those factors. The absence of massive blood loss, negligible pressor use, no fluid shifts, minimal ischemia, no significant acidosis, no prolonged OR and clamp times, no blood transfusions, and the presence of avid back bleeding suggesting strong collateralization, compelled me to end the operation without revascularizing these intercostal vessels.
The patient recuperated for an ten days after his operation but was walking from postoperative day #1. He had atelectasis due to mucus plugging in his right lung base and was treated for aspiration, but clinically did not have a pneumonia and his atelectasis cleared with chest physiotherapy and nebulizers and was discharged home. Gratefully, he has given us permission to discuss and study his case.
There is no success in these cases without a team, and we are blessed with talented caregivers. This technique greatly reduces the physiologic impact of this surgery on the patient, reducing the injury to the equivalent of a broad sword cut from chest to abdomen that missed all the vital organs and vessels. In the right hands, this concept will broaden the appeal of open repair of these challenging aneurysms.
Reference
Matalanis G, Ch’ng SL. Semin Thoracic Surg 31:8:708-12.
A basic recipe for treating complicated aortic dissection
#aorticsurgery #tevar
It was only last month when I came across a post of an aortic aneurysm in a difficult spot (link) and I couldn’t help chiming in some comments. Reading it now, I sound insufferable, because I wrote,
“Depends on etiology and patient risk stratification. Also assuming aneurysm goes to level of SMA and right renal artery origins and involves side opposite celiac. Options depending on resources of your institute: 1.Open repair with cannulation for left heart bypass and/or circulatory arrest 2.Open debranching of common hepatic, SMA, R. Renal from infrarenal inflow and then TEVAR 3.FEVAR from custom graft from manufacturer on protocol 4.Parallel grafting to CA, SMA, R. RA with TEVAR 5.Surgeon modified FEVAR 6.Open Repair We would have a multidisciplinary huddle around this patient -Vascular, Cardiac Surgery, Cardiology, Anesthesia, and ID (if needed) to help choose. Be ready to refer to a center with more resources -including prepping patient for transfer and imaging -including uploading images to the cloud for transfer with patient’s permission. More info please”
More Info Please, Indeed
The post I commented on was of a saccular aneurysm in the transdiagphragmatic segment of aorta. Ironically, only a few weeks later, I got called from hospital transfer center about a patient with a leaking aortic aneurysm, a type V thoracoabdominal aortic aneurysm as it turned out, from an outside hospital, needing urgent attention, and we accepted in transfer based on the conversation I had with the tranferring physician. And that was the problem -usually in taking these inter-hospital transfers, you have to pray that the precious CT scans come along with the patient burned correctly onto a CD-ROM. You can buy and watch a movie in 4K resolution over the internet -about 4 gigabytes, but a patient’s CT scan which is about 200 megabytes -because of various self imposed limits, overly restrictive interpretations of laws, and lack of computer skills, these life saving images get transferred on CD in 2019. That last time I purchased a CD for anything was over 15 years ago.
An Interested Party
The technical solution –to use the internet to transfer critical life saving information between hospitals – came about when our IT folks took an interest in my quarterly complaint email about using the newfangled internet for sharing files. After mulling various solutions ranging from setting up a server to using commercial cloud solutoins, we came upon the compromise of using our internal cloud with an invitation sent to the outside hospital. I would send this invitation to upload the DICOM folder of the CD-ROM to an outisde email address. It was simple and as yet untried until this night. “Would the patient agree to have his CT scan information transmitted to us electronically?” I asked the other physician. He assured me that the patient was in agreement.
It Takes Two to Tango
Of course, being able to transfer these pictures requires a willing partner on the other side, and the referring physician made it clear he did not have the technical expertise to do so. It took a bit of social engineering to think about who would have that ability. Basically, aside from myself, who spend all their time in dark rooms in the hospital in front of giant computer monitors? The radiologists! I got through to the radiologist who had interpreted the report and explained the simple thing I needed. Gratefully, he agreed, and I sent him a link to our cloud server. I explained to him, “When you receive this, clicking the link opens a browser window and then you open the CD-ROM and find the DICOM folder and drag and drop it on the browser window.” The 200-500 megabytes of data then get sent in electronic form, as it was meant to in 2019.
The Internet Saves a Life
The brutal truth is that in locking down a computer system, many hospitals make it impossible to even load an outside CD-ROM, creating many self imposed barriers to care. Thankfully, at CCAD, we were able to work together to find a secure solution. With the CTA on our servers, I was able to review the study within 15 minutes of accepting the patient, and arrange for the right team to be assembled, and confirm that we had the right material (stent grafts) for treating the patient. When the patient arrived, OR was ready to go, saving hours of time that normally would have been required if the CT scan had to be reviewed from the CD-ROM that came with the patient. Sometimes, the CD-ROM does not come, and in a critical situation, the CTA has to be repeated with some risk to the patient for complications of the contrast and radiation.
What to Do
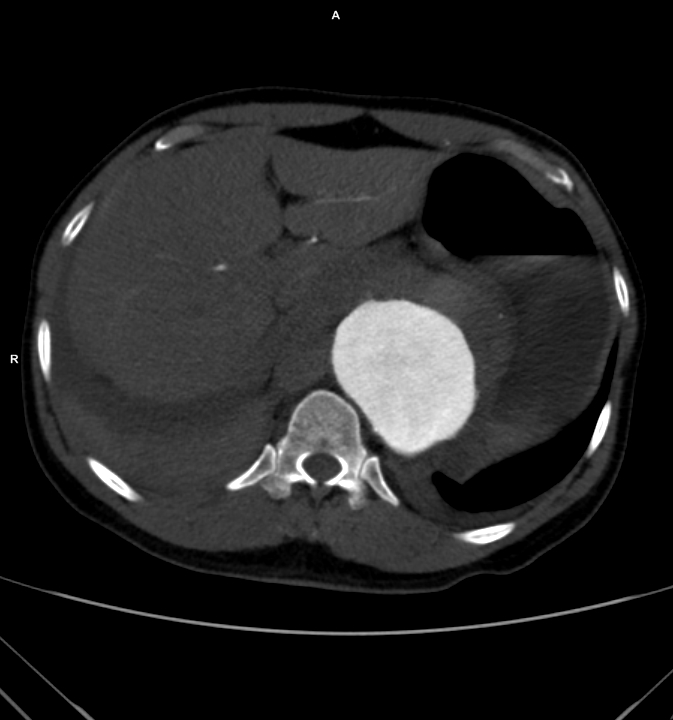
The patient had a 8cm sphere shaped aneurysm arising in the transdiagphragmatic aorta, leaking into the right pleural cavity.

The patient was otherwise a healthy middle aged man with risk factors of smoking and hypertension. The centerline reconstructions showed the thoracic aorta above the aneurysm to be around 20mm in diameter and same below, with the celiac axis and superior mesenteric artery in the potential seal zone of a stent graft. The only plaque seen was around the level of the renal arteries and was focal and calcified. Looking at the list I had made as a comment to the Linked-In post, I realized that I really only had one viable option.

Open repair, usually the most expeditious option, was made challenging by the right hemothorax, making a left thoracotomy hazardous if the lung had to be deflated. Cardiopulmonary bypass would have to be arranged for, and that adds a metabolic hit that greatly raises the stakes. Of the endovascular solutions, the only viable option was TEVAR to exclude the rupture and debranching of the celiac axis and superior mesenteric artery. To those who would advocate for parallel grafts, there was no room in the normal 20mm diameter aorta. And branch systems for rupture are some time in the future. Also, the patient was becoming hypotensive. So the planned operation was first TEVAR to stop the bleeding, and then open surgical debranching. A hybrid repair.

The smallest stent graft we have is a 21mm graft, but it would not be suitable for this aorta. In practice, the normal aorta is quite elastic and will dilate much more than what is captured on a CTA. The next size we have is 28mm graft and I chose this to exclude the rupture, which was done percutaneously.

As seen below, the graft excluded the celiac and SMA. Late in the phase of the final aortogram (second panel) there was an endoleak that persisteed despite multiple ballooning. The timing suggested the intercostals and phrenic vessels contributed to a type II endoleak, but it was concerning.
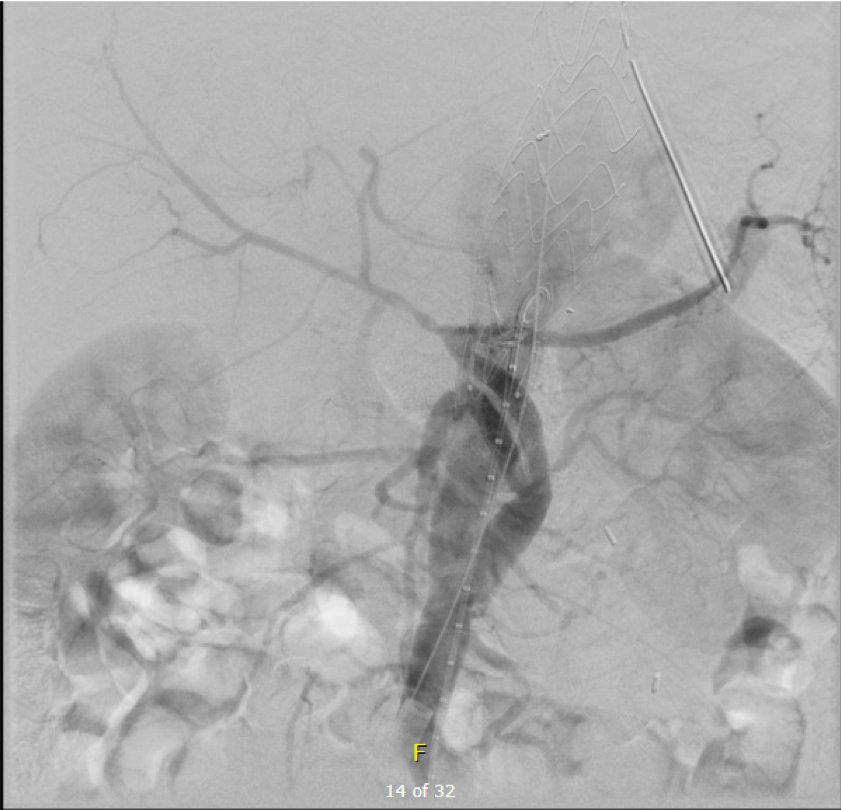
The bypasses were sent from the infrarenal aorta to the common hepatic artery and the SMA close to its origin, and the origins of the celiac axis and SMA were clipped. The bypasses were then done with a 10x8mm bifurcate Dacron graft originally for axillofemoral bypassing. It had spiral rings which I removed at the anastomosis and this resulted in a kink at the closer bypass. Usually, I loop this for iliomesenteric bypass but there was not enough distance from the infrarenal aorta. I have to add a little trick I modified from my pediatric surgery experience as a resident -a Heinecke-Mikulwicz graftoplasty:

This worked to relieve the kink as evidenced on the aortogram above. After closing the laparotomy, I placed a chest tube in the right chest. The patient had a course prolonged by a classic systemic inflammatory response syndrome, with fevers, chills, and leukocytosis. He bled for a while but stopped with correction of his coagulopathy. All blood cultures were negative, but a CT scan was performed out of concern for the endoleak, and the possibility of continued bleeding.
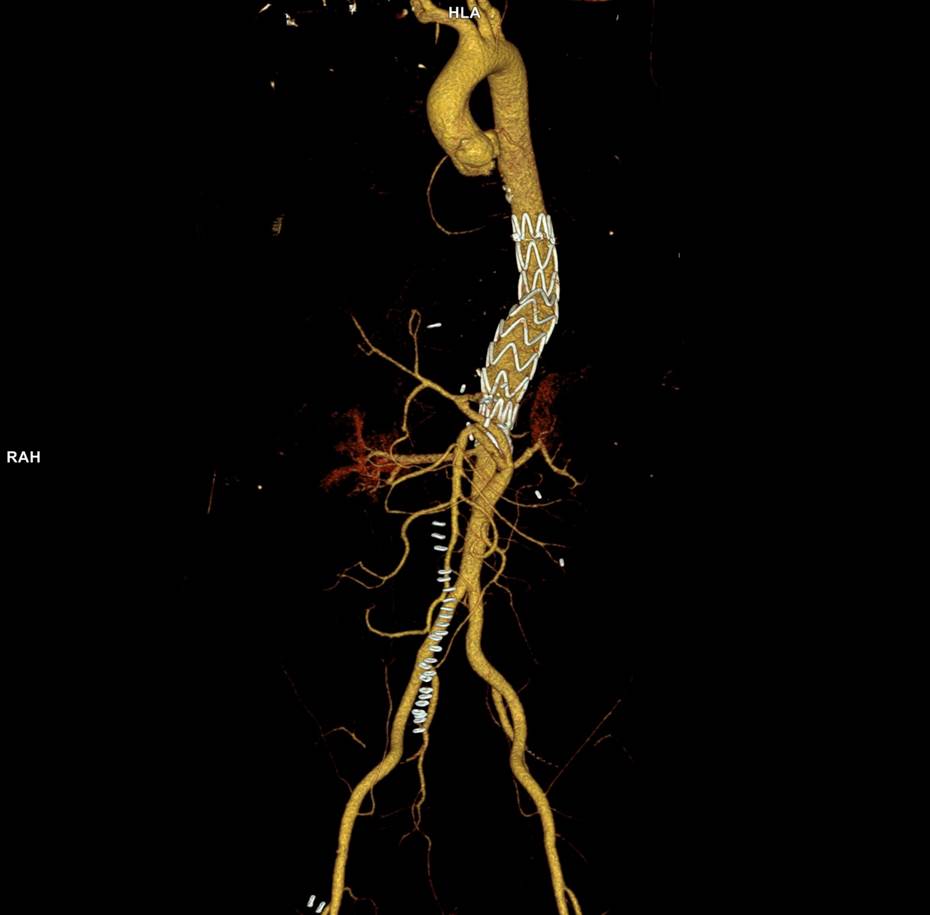
No endoleak was detected as the sac was fully thrombosed. There was a consolidation of the blood in the right chest, but it resolved with fibrinolytic therapy.
Discussion
This case illustrates several points I have been making on this blog.
- Hybrid repairs are not some kind of compromise but the full realization of a complete skill set. When students ask me how to judge a training program, one of the best metrics is how frequently are hybrid operations performed. It means either the endovascularist and open surgical operator are in complete synchrony or there are individual surgeons competent in both open and endovascular surgery. Hybrid operations, rather than being a compromise, are an optimization.
- Time -Laying the stent graft across the celiac and SMA origins starts a warm ischemia clock. The liver and intestines, in my reckoning, should be able to tolerate the 2 hours of work to get the bypasses working. Cardiopulmonary bypass may give you less ichemia but at the metabolic cost of the pump time. These negative factors add up, but were surmounted by the fact that early control of hemorrhage was achieved. Stopping the bleeding and restoring flow are the core functions of vascular surgery.
- Planning and preparation. The ability to see the CT images and prepare the teams and materiel before the patient transferred was lifesaving. This is where our IT gets credit for responding to a critical need and formulating a solution that meets internal policies, external regulations, and saves a life. It illustrates so many opportunities particularly with electronic medical records and their processes which focus more on documentation for billing. A discharge summary should be multimedia like this blog post and it should be normal and easy to generate. And finally, as clinicians, we should mind technology with as much attention as we give to the latest medical devices and techniques.



I was invited by Dr. Martin Maresch to speak on complicated type B aortic dissections. Should be an exciting day.

The innovation of sabremetrics in baseball management and finance as described in Michael Lewis’ wonderful book Moneyball wasn’t just the ability to quantify skill to predict outcomes, it was the ability to assemble that skill without overpaying. For a baseball team on a budget, spending all your payroll on a superstar makes no sense when you can get equivalent quants of skill in a statistical aggregate of no-name players with proven metrics. Rather than pay for an A-Rod, you can recruit, and pay for, 5 players that in aggregate, statistically achieve what you would get with a healthy A-Rod, so the thinking goes. How does this translate into vascular surgery? Can we arbitrage complication rates?
The open repair of type II thoracoabdominal aortic aneurysms is a heroic endeavor, putatively best done by surgeons wearing cowboy boots, and classically comes with sobering complication rates that exceed 20% for death and paralysis. Is it possible to reduce this risk by subdividing this most enormous of cardiovascular operations into component parts?
The patient is a middle aged man in his 50’s who presented with a type B aortic dissection. His dissection flap spanned from his left subclavian artery to the infrarenal aorta. He was a long time smoker and had hypertension that was difficult to control, made much worse after his dissection. He had a moderate dilatation of his thoracic aorta, maximally 36mm and tapering to 35mm in visceral segment. There was a 4.9cm infrarenal AAA where the dissection terminated.

His chest pain resolved with blood pressure control and he was discharged, but in followup his thoracic aortic segment grew and his blood pressure worsened, never getting below a 150mmHg systolic despite multiple agents. CTA two months after presentation, showed growth of his TAA to 44mm from 36mm in two months and the visceral segment showed that his dissection flap impinged on flow to the right renal artery. His AAA remained the same. He continued to have bouts of chest pain related to hypertension.
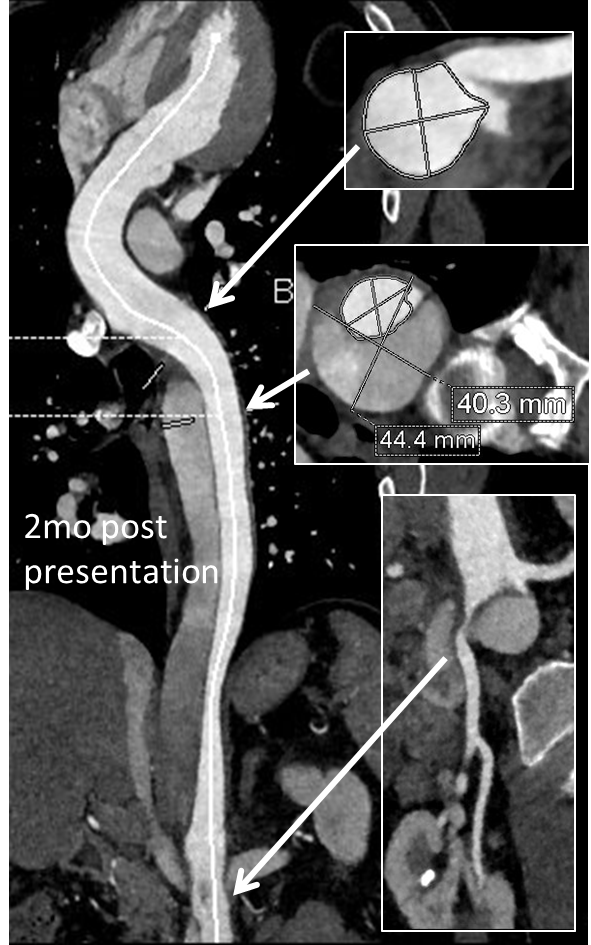
Twenty years ago, the board answer would have been to replace the whole aorta. In young, otherwise healthy man who had been working in road construction up to the dissection, he would have been considered a candidate for a direct open repair of the type II thoracoabdominal aortic aneurysm. From the landmark paper out of Houston by Dr. Svensson in 1993, open type II TAAA repair was associated with about a 10% death rate and 30 percent paralysis rate. Waiting a few months for the aneurysms to grow further in this patient, in the 90’s this patient would probably have ended up with an open TAAA repair. Good thing we have better options.
The goals of modern therapy are to treat the urgent indication while holding off repair of less critical segments of the aorta, and to do so in a way that each operation builds on the previous one.
This patient needed a left subclavian artery debranching and then TEVAR of his dissecting thoracic aortic aneurysm, and intervention on his right renal artery. We did this in one setting performing first a left carotid subclavian artery transposition and then percutaneous TEVAR from the left common carotid artery origin to the supraceliac abdominal aorta.
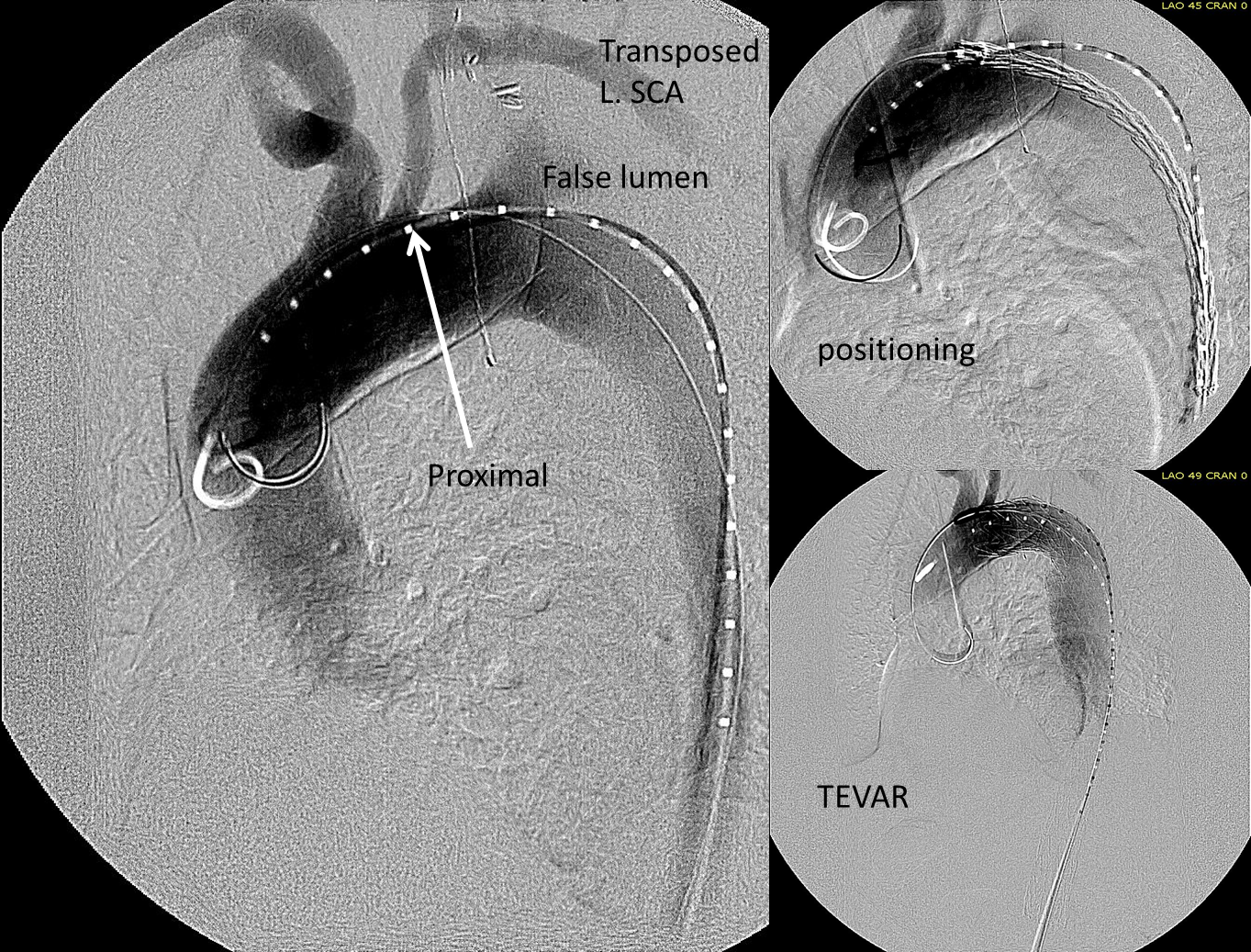
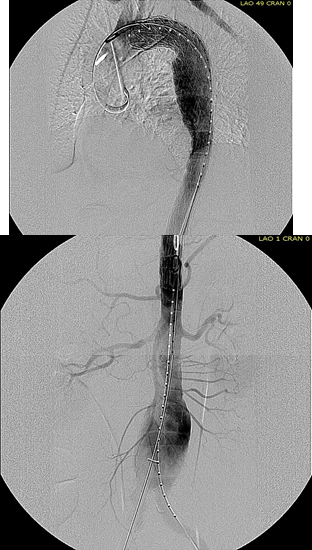
The completion aortography showed good deployment of the CTAG device from the left common carotid artery origin to the celiac axis origin. The false lumen was no longer visualized. The right renal artery which was narrowed was treated with a balloon expandable stent.
The distal thoracic aorta, the true lumen was constrained by a chronic dissection flap. It is here I gently dilate the distal thoracic stent graft with the hopes of eliminating the distal false lumen. This is different from the acute dissection where I rarely balloon.
The TEVAR was done percutaneously, minimizing the overall time in the operating room. The technical details of the transposition can be found in the excellent paper by Dr. Mark Morasch.

When I do this procedure for acute dissection, I quote the patient a general risk of stroke, paralysis of about 2-5% and death of 1-2 percent for someone with low cardiopulmonary risk like this patient had. He recovered rapidly and went home post op day 5.
Followup post TEVAR
He at 6 month post TEVAR followup, CTA showed stablility in his thoracic aorta. in infrarenal AAA grew from 5.0 to 5.7cm between the 1 month CT and the 6 month CT.
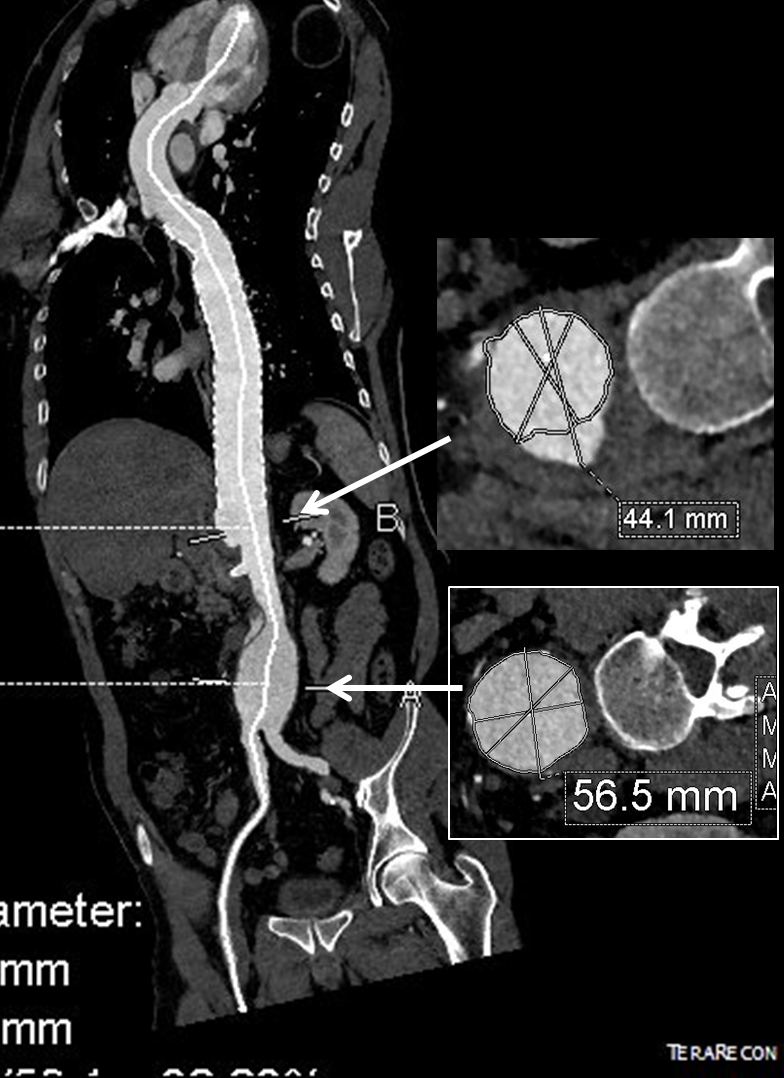
The terminus of the stent graft excluded the false lumen in the thoracic aorta but also resulted in filling and pressurization of the false lumen beyond and can be seen as a 44mm lateral dilation of the visceral segment of the aorta which had developed in the 6 month interval since the TEVAR.
The infrarenal neck continued the dissection and had dilated to about 36mm, but was parallel for a good length above the AAA. I decided to treat the inrarenal aorta with direct transabdominal repair. This would allow me to fenestrate the aorta, and possibly prevent further growth of the viseral segment while reserving the retroperitoneum for the visceral segment repair if it came to it. The neck diameter was 36mm.
 His operation was performed via an anterior approach with the patient supine. A tube graft repair was performed expeditiously and included resecting the dissection flap up to the clamp. Care was taken to avoid injury to the renal stent. The proximal anastomosis went well – the dilated aorta yet had strong tissue strength. A felt strip was used to buttress the aortic side of the anastomosis. The estimated risk of paralysis was less than 1% and risk of death was less than 2%. The patient recovered uneventfully and went home on POD 5.
His operation was performed via an anterior approach with the patient supine. A tube graft repair was performed expeditiously and included resecting the dissection flap up to the clamp. Care was taken to avoid injury to the renal stent. The proximal anastomosis went well – the dilated aorta yet had strong tissue strength. A felt strip was used to buttress the aortic side of the anastomosis. The estimated risk of paralysis was less than 1% and risk of death was less than 2%. The patient recovered uneventfully and went home on POD 5.
He did well in subsequent followup, having successfully quit smoking. He retired early on disability and was becoming more active, but the visceral segment dilatation was concerning. At 6 months post infrarenal AAA repair, he underwent CTA and it showed patent thoracic stent graft and infrarenal abdominal graft. The intervening visceral segment continued to enlarge and was now 46mm. The decision was to wait another interval 9 months to see if this would stabilize. The segment grew some more and was 49mm. He wanted to give it another 6 months and at that time, CTA showed further growth over 5cm, and he had developed some abdominal discomfort. He was taken to the operating room.

A four branch repair of the visceral segment thoracoabdominal aortic aneurysm was performed. The diaphragm was taken down and the stent graft was clamped as was the infrarenal tube graft. A premade Coselli graft was used to bypass to the right renal, SMA, celiac, and left renal in those order. The patient had a CSF drain for the case which was removed on postoperative day 2. He recovered rapidly and went home on postoperative day 6. His estimated risk of paralysis was about 2-5%, mitigated by a protocol centered on CSF drainage and blood pressure control. His risk of death was 5%. Telephone followup reveals the he is pain free at a month out and functional nearly at baseline.
This illustrates the notion that three smaller operations in an aggregate over three years achieved the equivalent of the single big open type II TAAA repair.
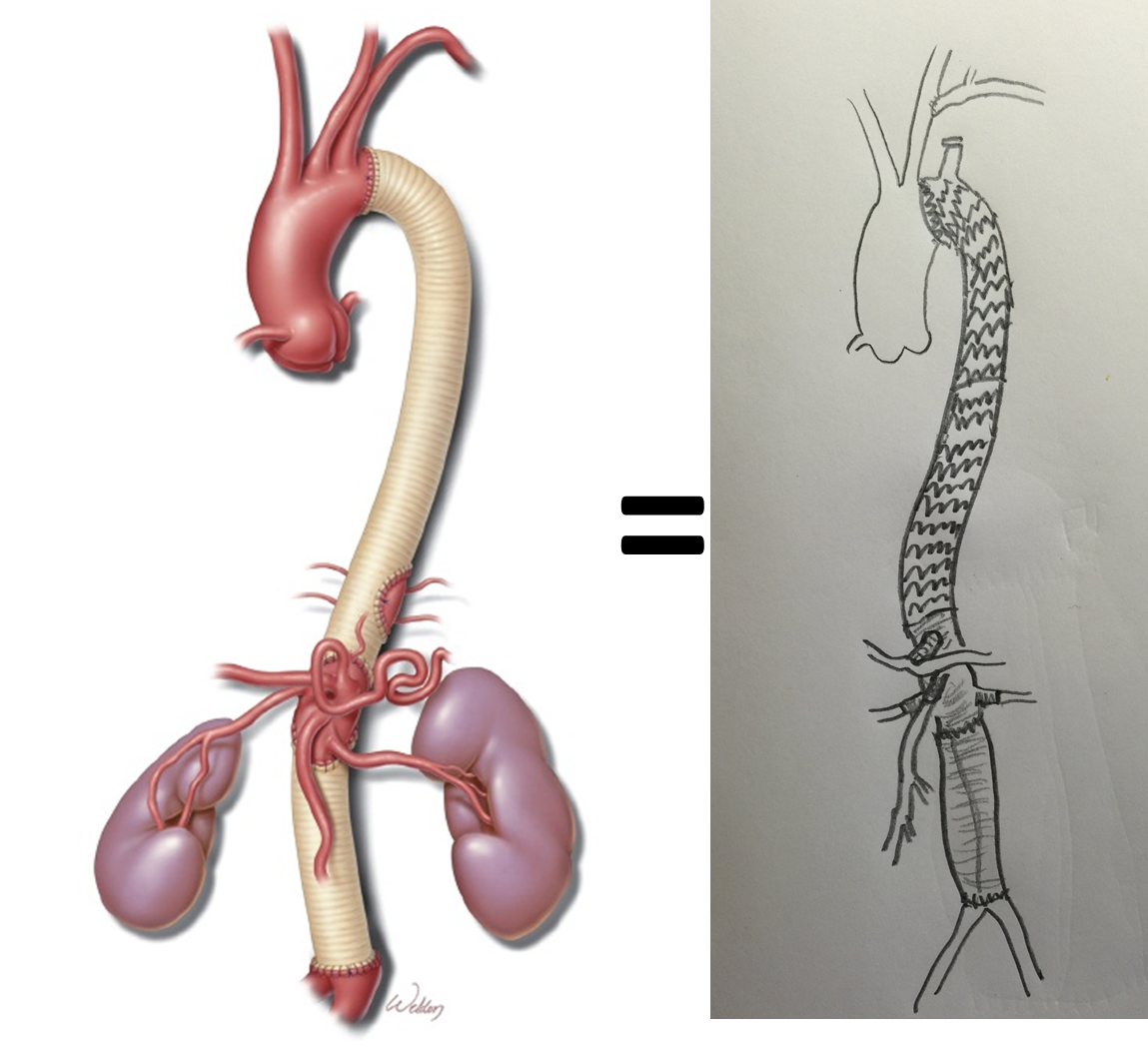
The idea is to make each step achievable -like coming down a mountain taking three days on well marked paths rather than base jumping off the summit.
Clearly, the patient was younger and a fast healer, and credit must also be given to the anesthesia/critical care team who see high acuity cases in volume every day and not every patient can expect to have such short stays and excellent outcome, but these are far more likely if operations are planned out in such a manner.
Reference
Svensson LG, Crawford ES, Hess KR, Coselli JS, Safi HJ. Experience with 1509 patients undergoing thoracoabdominal aortic operations. J Vasc Surg 1993;17(2):357-36.
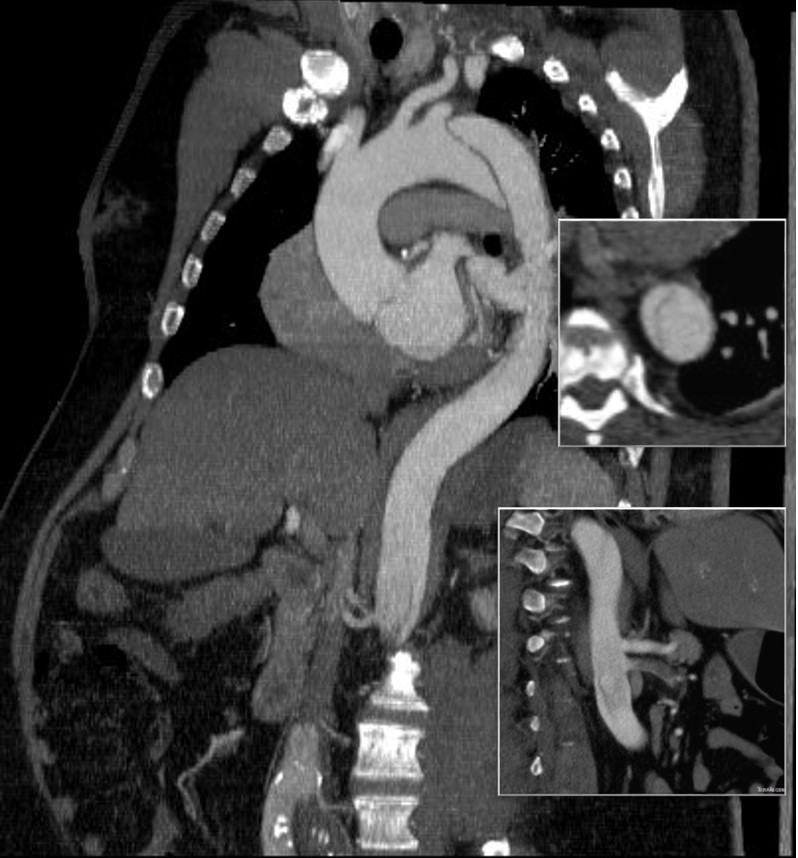
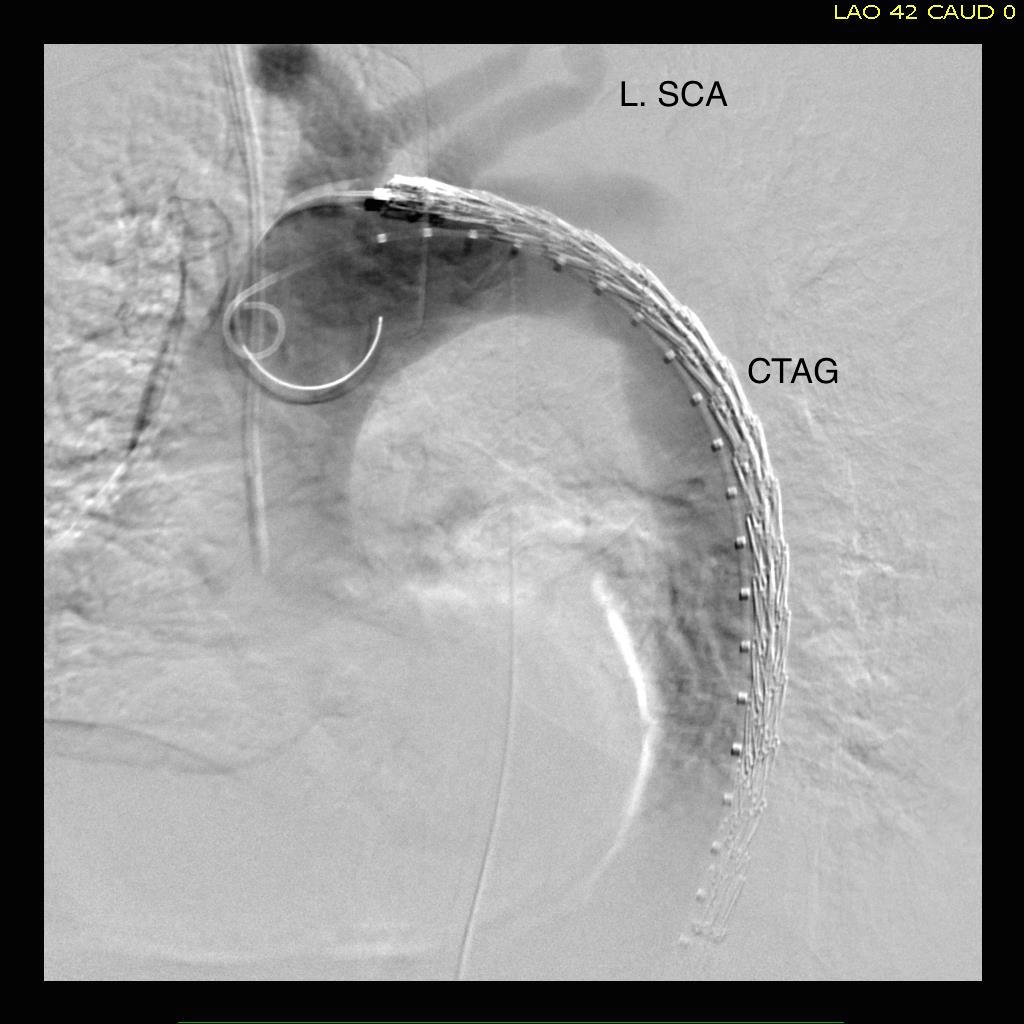
The patient is middle aged and had a type B thoracic aortic dissection (TBAD) as a consequence of recreational substances that acutely raised his blood pressure. At the outside hospital, he had a CTA showing the dissection extending from his left subclavian artery and causing occlusion of his superior mesenteric artery (SMA). He developed abdominal pain and was swiftly transported to our acute aortic syndrome unit. He was taken to the operating room and underwent a TEVAR of the dissection and stenting of his SMA -this is similar to other cases I have discussed in prior posts so I am omitting the technical details. The stent covered the left subclavian artery origin to exclude the origin of the dissection. The stent was extended to the distal thoracic aorta but did not go to the celiac origin.
Post procedure, his lactate never rose and he was maintained on the usual post procedure protocol of keeping MAP’s (mean arterial pressure) above 80mmHg. His left subclavian artery was covered but I do not routinely bypass, especially when the left vertebral artery is at least equal in size to the contralateral one. I don’t often place spinal drains for urgent/emergent cases particularly in patients who have never had infrarenal aortic surgery and patent hypogastric arteries. He was kept sedated overnight and awoke in the morning unable to move his legs to command. He had no pain sensation up to his umbilicus.
A spinal drain was emergently placed and his blood pressure was raised to MAPs of 90+, but these failed to reverse his paralysis. After discussion among my world class partners, I chose to take the patient back for a carotid subclavian bypass which was done through a single incision with a dacron bypass graft.

His paralysis resolved. He was discharged home, ambulating without assistance. Spinal cord complications are reported to occur between 1-5 percent of patients undergoing TEVAR for complicated TBAD. They were seen in 2 of 72 patients in the TEVAR arm of the INSTEAD trial (Circulation, 2009 vol. 120(25) pp. 2519-28), and was permanent in 1. While there are some who routinely place prophylactic drains, it is unclear to me that they have a significant effect if placed unselectively. I will place a Preop drain in the instance of infra renal graft, hypogastric arterial occlusive disease. In the instance of a dominant left vertebral, I will perform concomitant bypass, but just as often not. This is a gratifying and rare outcome of paralysis reversed with a carotid subclavian bypass when spinal drain and permissive hypertension did not.
The figure above shows the summarizes the problem that brought the patient to his local hospital and triggered his transfer to our acute aortic syndrome unit. The concept is that all chest pain of cardiovascular origin gets intake through a vast intensive care unit staffed by cardiovascular intensivists. Stabilization, workup, transfer to operating room or interventional suite all happens in an ICU that encompasses almost a city block.
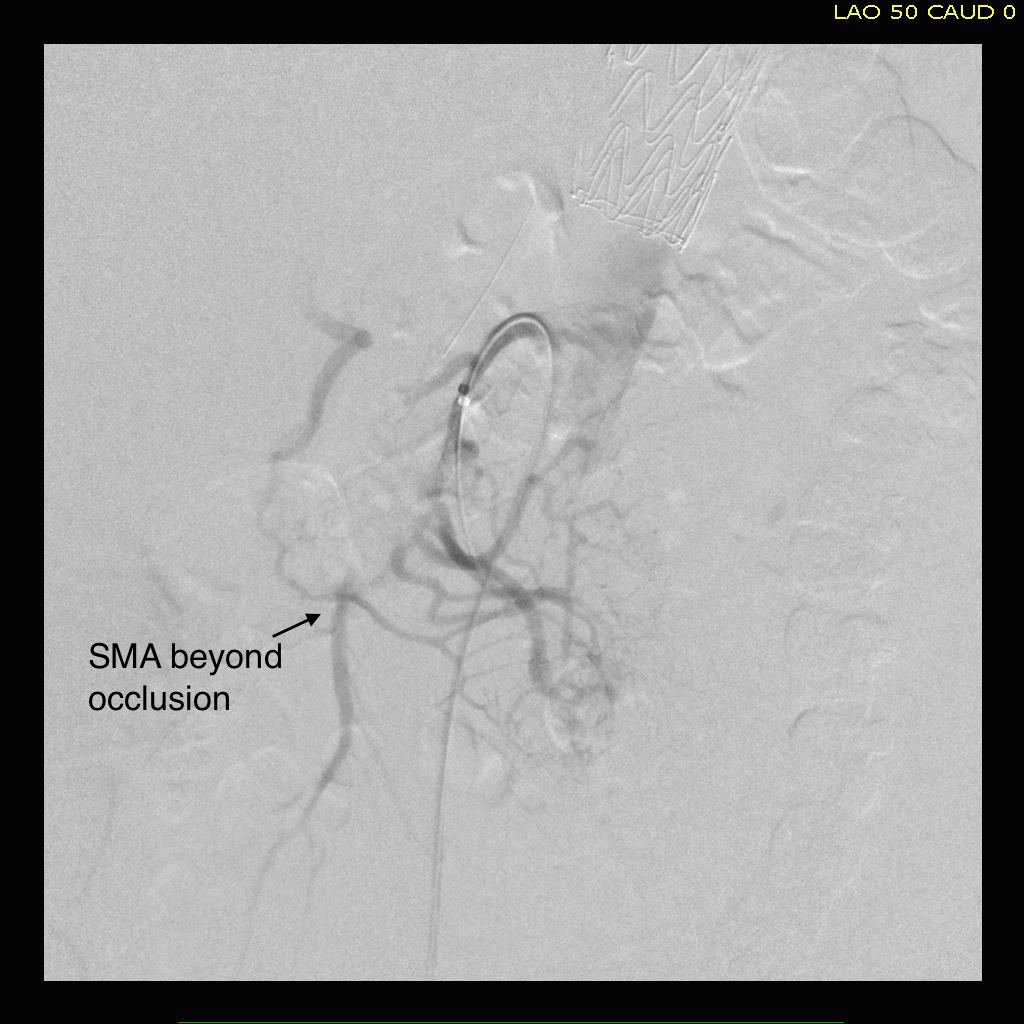
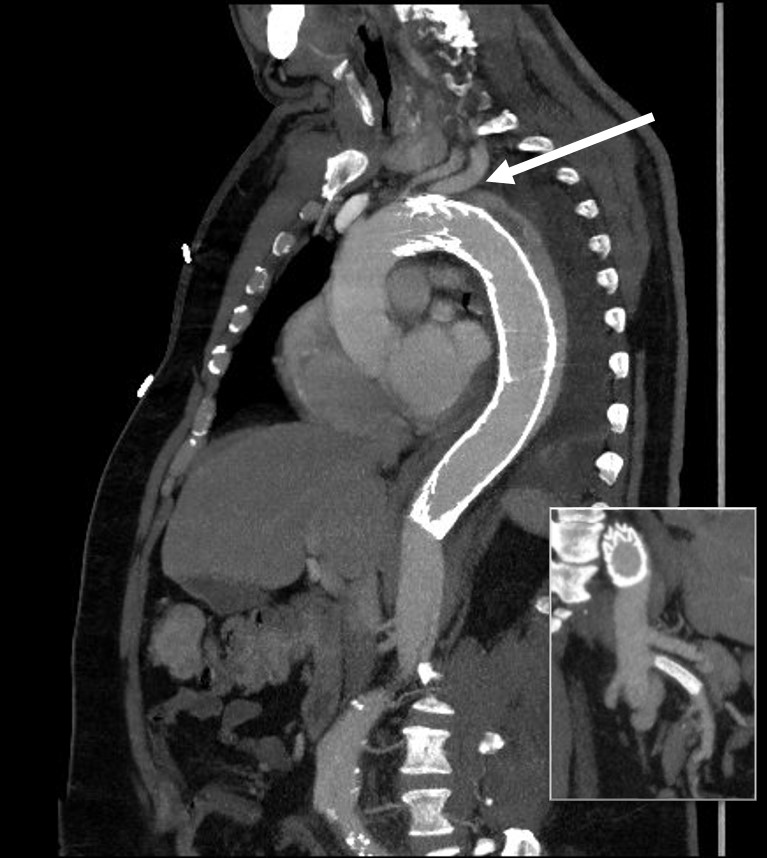
The patient is an older middle aged man with sudden onset of back and abdominal pain. He was on coumadin for a prior SMV thrombosis and treatment for a ruptured appendicitis -antibiotics with plan for staged appendectomy. CT at his local hospital revealed a type B aortic dissection (TBAD) that extended into his superior mesenteric artery.

The dissection started at the left subclavian artery origin. The false lumen compressed the true lumen up at the proximal descending thoracic aorta. This is an important finding because in this configuration with much of the filling of the dissection from the distal reentry sites, the false lumen acts like a pressurized, compressive lesion. With time, the adventitia around the false lumen may become aneurysmal if the false lumen fails to thrombose or obliterate. When the dissection is acute, the flap may cause a direct obstruction to flow or a dynamic one that depends on the pressure difference between true and false lumen.
In this patient, thrombosis occured in the SMA beyond the origin due to dissection and decreased flow. This was consistent with the patient’s complaint of generalized abdominal pain and examination findings of pain out of proportion to the exam, indicating acute mesenteric ischemia.
His laboratory findings were within normal ranges, indicating this was early in the process. It is important to remember that no lab value correlates with acute mesenteric ischemia except very late in the process, and acute mesenteric ischemia remains a clinical diagnosis (reference 1) that is associated with a high mortality rate.
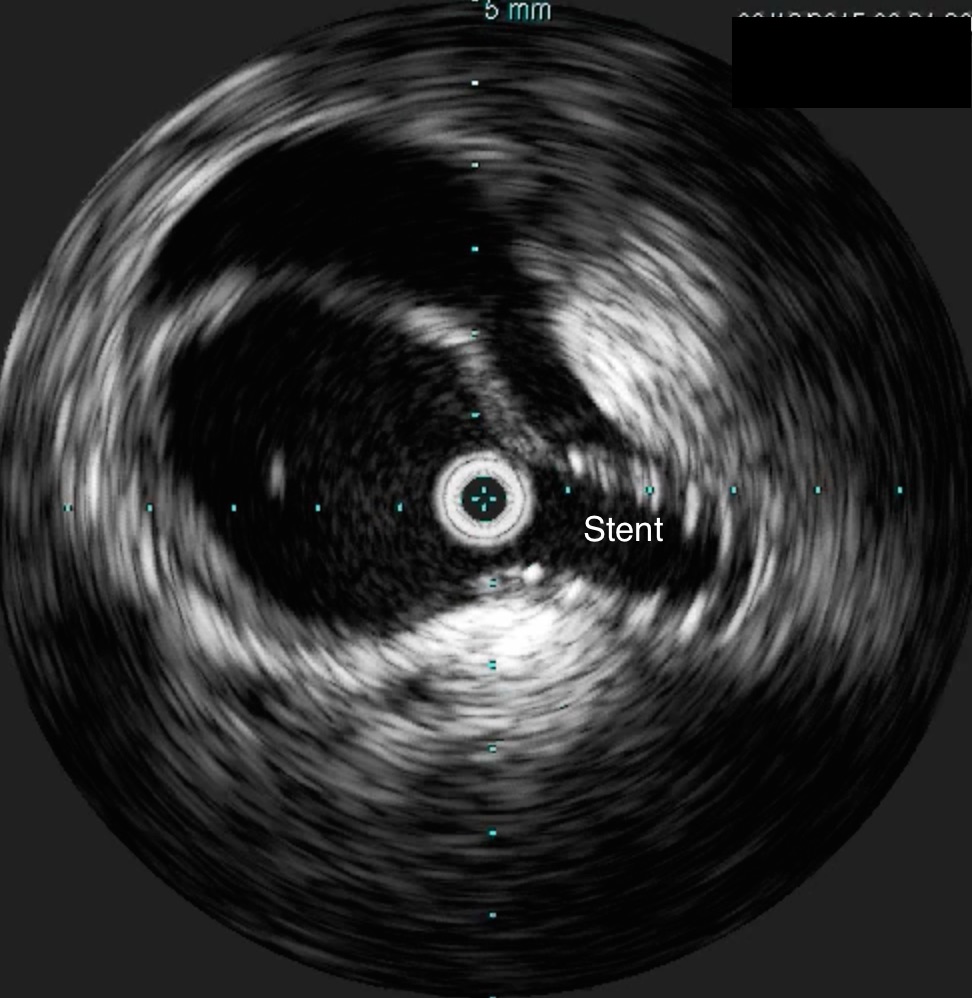
He was taken to the hybrid operating room. Right groin access was achieved and wire access to the arch was achieved. IVUS (Intravascular ultrasound, Volcano) was used to confirm the location of the wire -I believe this is an important adjunct as simply passing the wire doesn’t guarantee travel up the true lumen.
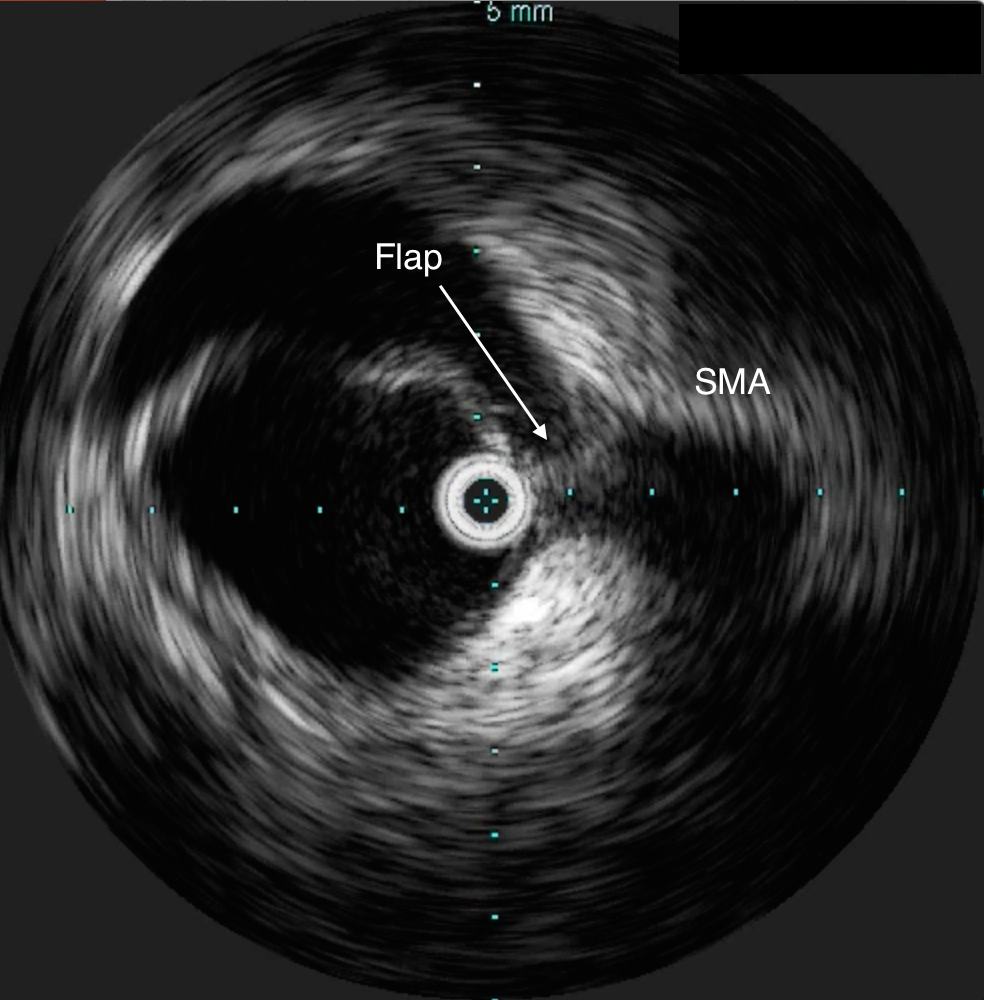



Wire access was achieved across the thrombus. At this point, I had a range of options for thrombectomy including simply aspirating which retracting a catheter. This was not optimal as I could lose subsequent wire access or reenter the false lumen. The other option was an open thrombectomy and patch angioplasty -the thighs were prepped in case we had to harvest vein. Again, in the setting of dissection and going into the mesentery, an open revascularization while feasible, is challenging.
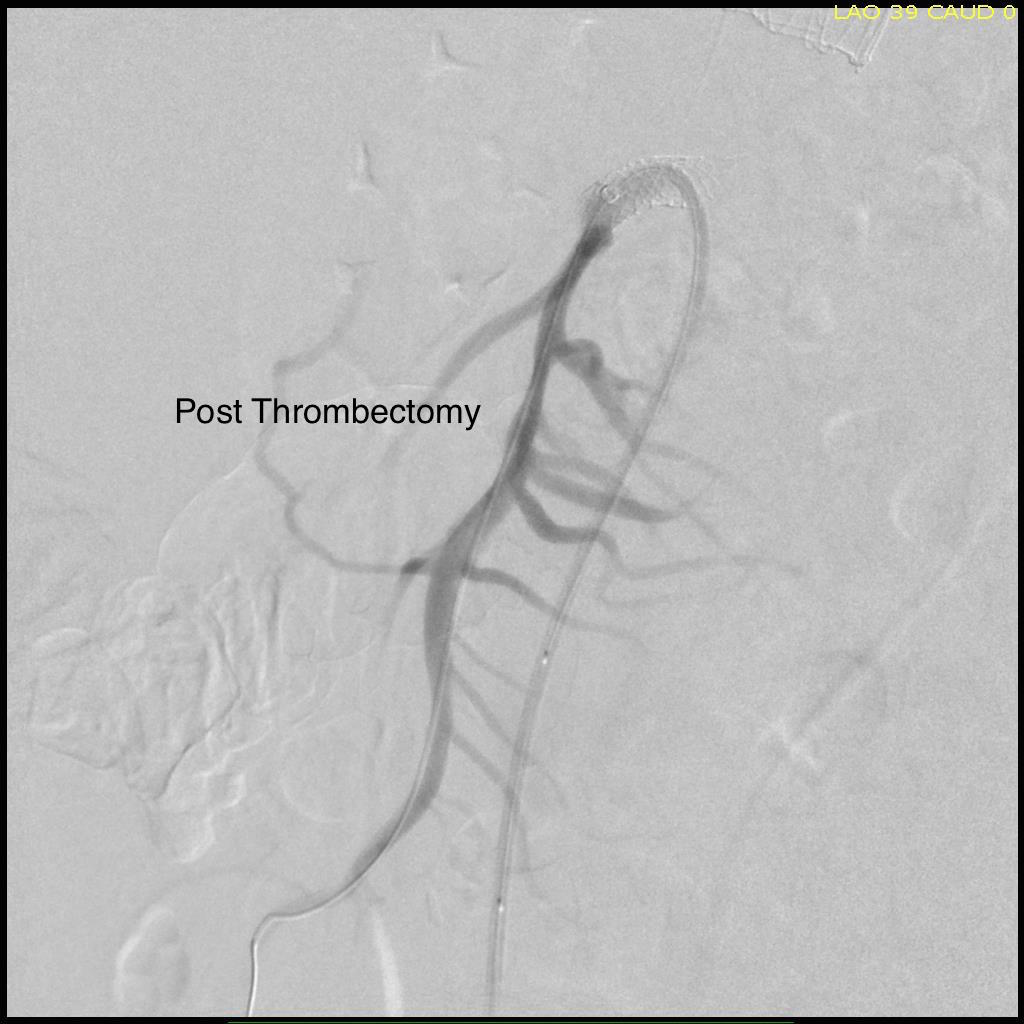
Thrombectomy catheters like Angiojet were available, but I chose to try the Export aspiration catheter (Medtronic). It is simple to set up and goes over a 0.14 wire. It is designed for the coronaries which have a similar lumenal diameter as the SMA. It worked well in this setting in retrieving thrombus which had a pale element that may have indicated some chronicity.
The completion arteriogram was satisfying.
The SMA completely filled as did the celiac axis and both renal arteries. I opted not to treat the right renal artery as we had given 250mL of contrast, and it was filling well without intervention. The patient was making excellent urine and his blood pressure had been maintained with mean arterial pressures above 70mmHg. At this point, IVUS confirmed good deployment of the stent.
The sheath was removed and the access site repaired. The general surgeons explored the patient and found all the bowel to be well perfused with pulsatile flows seen in the mesenteric arcade. The appendix was removed.
On waking, the patient was noted to not move his legs. A spinal drain was expertly placed by our cardiac anesthesia staff and his blood pressure was raised to MAP’s above 80. He recovered motor function in his legs soon after. I usually don’t place preop CSF drains in this scenario in the presence of good pelvic circulation, no history of infrarenal aortic interventions, and patency of the left subclavian artery. That said, with TEVAR of TBAD, there is a small incidence of paraplegia (1-5%) which I emphasize in my preoperative discussion.
He was started on heparin anticoagulation postop because of his history of SMV and now SMA thrombosis, interrupting it briefly to remove the CSF drain. A CTA was obtained to confirm absence of bleeding showing obliteration of the dissection in the aorta and good patency through the true lumen of the SMA.
Most importantly, he had complete relief of his abdominal pain.
The treatment of acute mesenteric ischemia has greatly evolved since I presented my paper in 2002. While open revascularization remains a gold standard, it is becoming increasingly apparent that good to better results may be obtained with an endovascular approach. Dan Clair, our chair, has made the comment that early revascularization with endovascular technique is analogous to emergent PTCA in occlusions of the coronary system and that re-establishing flow is a critical first step.
Open exploration still is the mainstay of managing acute mesenteric ischemia, but laparoscopic exploration remains feasible. This patient underwent open conversion after an initial laparoscopic exploration to remove a ruptured retrocecal appendix that had been treated for over a month on antibiotics. Without bowel necrosis, a second look is usually unnecessary, but is critical when threatened bowel is left behind.
Reference
- Park WM et al. J Vasc Surg. 2002 Mar;35(3):445-52.