Why There is a Literature on Filter Removal

A long time ago, there was the IVC clip which survives today as a vestigial CPT code. Then in the 1980’s, the Greenfield filter was introduced and changed the management of thromboembolism (reference). The explosive adoption of endovascular technology in the late nineties and early 2000’s drove the growth in implantation of newer generations of IVC filters that were designed to be retrievable. The people requesting the filters -the physicians, surgeons, and even patients looking to stop taking anticoagulation, were basing their decision on common sense –“sometimes, people are vulnerable to pulmonary embolism and are at risk of hemorrhage with anticoagulation, so an IVC filter makes sense.” There was frankly a data gap -a breach into which multiple companies jumped in with their own flavor of filter. Many interventionists saw no need to be selective -these were easy to place, and easy to remove, and if they stayed in there was the excellent long term results of the Greenfield filter to cite, and their referring docs asked for it. There was also the high revenue density (revenue/time) that gave filters a gravitational pull. There were several problems with this endo-enthusiasm (like in so many other cases). In the absence of data and with the aggressive marketing, too many filters went in for weak indications. We now know that most of these filters do not behave like the Greenfield, which itself is not completely innocent. Unless followup is part of a process, many patients neglected to have their filters removed. And finally, the data caught up and failed several filters which are no longer on the market and the indication for these filters is now quite narrow.
In 2009, I was asked to consult on a young man who was hospitalized for upper GI bleeding. The EGD revealed the tines of an IVC filter poking through (the jpg is somewhere I swear). The prior year, he had been in a bad car accident and had a filter placed but never had it removed. The filter had migrated out of the IVC into the duodenum and into the spine and aorta. I removed it operatively, and that was the beginning of a series of cases, about 1-2 annually in my general vascular practice, of filters that had eroded through the IVC and was causing symptoms of bleeding or pain. The pain typically was associated with a tine touching on or eroding into the spine. Biomechanically, the IVC is a collapsible tube and all the viscera on top of it weighs about as much as an equivalent sack of uncooked chitterlings when recumbent and grinds on the filter and any sharp parts. Imagine unbending a paperclip (figure) and putting it in the belly. Over time, that clip will poke a hole in something. Why would we not expect an IVC filter to behave otherwise?

An Iliocaval Thrombosis Below a TrapEase Filter
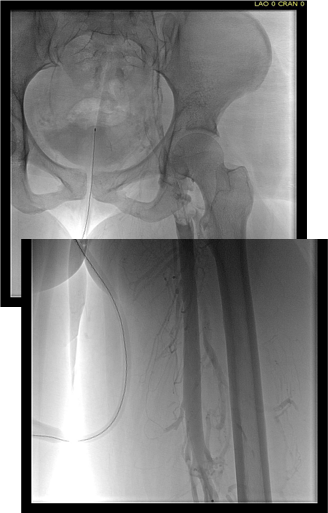
The patient is a younger man who over a decade ago had a TrapEase permanent IVC filter placed when he had a pulmonary embolism while having multiorgan failure. He was on coumadin briefly, but in the 17 years since filter placement, never had another venous thromboembolic event, but did develop venous insufficiency and varicose veins that were successfully treated. Several days prior to admission, he had been working out and developed back pain. After trying to sleep it off, he woke with severely swollen and painful legs. On admission, he was found to have no lower extremity DVTs, but had slow flow suggesting central occlusion. An abdominal x-ray showed the TrapEase filter (image below).

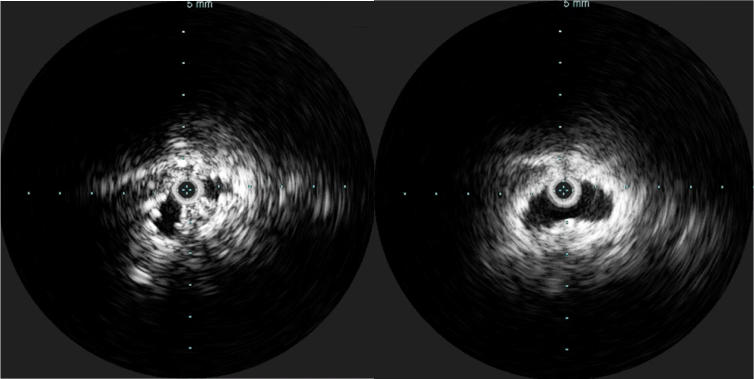
CT scanning and MRV showed the occlusion of the patient’s iliac veins and erosion of the struts of the filter outside the IVC (axial images below).

On heparin infusion and bedrest, his swelling improved and we had a chance to go over our treatment options. They included
- Catheter directed thrombolysis
- Angiojet thrombectomy
- Large sheath thrombectomy (link)
–with one of the following–
- Surgical resection of filter
- Endovascular retrieval of filter
- Balloon venoplasty and stent exclusion of the filter (link)
Catheter directed thrombolysis of such a large volume of clot in the absence of a good flow channel usually necessitates multiple days of thrombolysis with return for venography and adjustment, with a small but not zero chance of fatal or disabling hemorrhage. It is expensive -multiple ICU days, return trips to the OR angiosuite. Angiojet thrombectomy is useful for clearing smaller vessels and grafts but due to the pulsing of the jet, it has a good chance at creating pulmonary emboli if the filter’s occlusion is not complete. Plus it is expensive and limited by the volume of fluid necessary to create the suction. Large sheath thrombectomy has worked for me in the past (link), but I worry about leaving behind thrombus that would embolize when the filter is removed or pushed aside.
Endovascular removal of the filter is always an option -I have removed a Greenfield filter over two decades in. I have never been able to remove an OptEase or TrapEase filter -there is nothing easy about these. I have a picture somewhere sent to me by a trainee who was consulted on a patient whose IVC was transected during the attempt to remove one of these endovascularly. That said, both my partners Houssam Younes here in Cleveland Clinic Abu Dhabi and Christopher Smolock at Cleveland Clinic Main Campus suggested trying with a two-team approach with a 16F sheath and wire from above and a 12F sheath and wire from below. That would be contingent on being able to clear the thrombus adequately.
Surgery to remove the filter is something I am comfortable with. It sometimes is the only option (link). Several times a year, I scrub in with urologists and oncologic surgeons to remove tumor from the retroperitoneum or IVC and the exposure is straightforward. When I only need control of the IVC, I make a transverse incision slightly above the umbilicus and mobilize the retroperitoneum leaving the kidney down to expose the IVC. For the IVC and iliacs, a midline laparotomy with a Cattell maneuver allows for broad control. Surgical thrombectomy would be great if the thrombus was all fresh, but challenging if there were differing amounts of fresh and chronic occlusion. The CT scan, showing the iliac veins and IVC to be swole with clot, suggesting most of it was fresh. Neither CT nor MRV could tell me if the IVC below the renal confluence was occluded. I had to be certain.
I went over these issues in detail with the patient and we agreed to proceed with diagnostic venography to check out the clot. The verbiage of clot, tofu, and cheese (link) worked well in communicating the information needed to achieve our goal of getting the filter out and the IVC and iliac veins cleared of thrombus. I sent a hypercoagulability study (even on heparin, the genetic component is useful information) which returned negative.
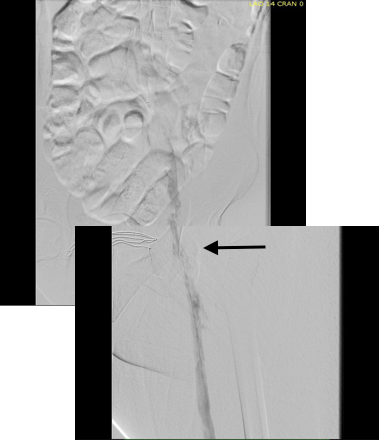
The diagnostic venogram is much more than just the pictures. For me, so much information is transmitted from the tip of a Glidewire as it passes through an obstruction or clot. Venography (image below) showed the thrombus but more importantly, the wire passed effortlessly in either side, got caught up in the bottom of the filter, but I was able to get through and the 5cm of IVC below the renal confluence turned out to be patent. The wire looped easily on both sides to the full extant of the dilated vein suggesting no chronic component.

I recommended surgery in our hybrid angiography suite. It would avoid multiple days of thrombolysis and its attendant risks. It would avoid subtotal clearance of thrombus. It would avoid failed filter retrieval and bailing out by stenting the filter (link), something acceptable in an older higher risk patient but not in an active young man. It would avoid surgery after several days of failed thrombolysis. The ability to perform venography and sonography with a clamp on the IVC ensured the ability to fully clear clot. And we had cell saver. After laying out my thoughts and concerns, the patient agreed.
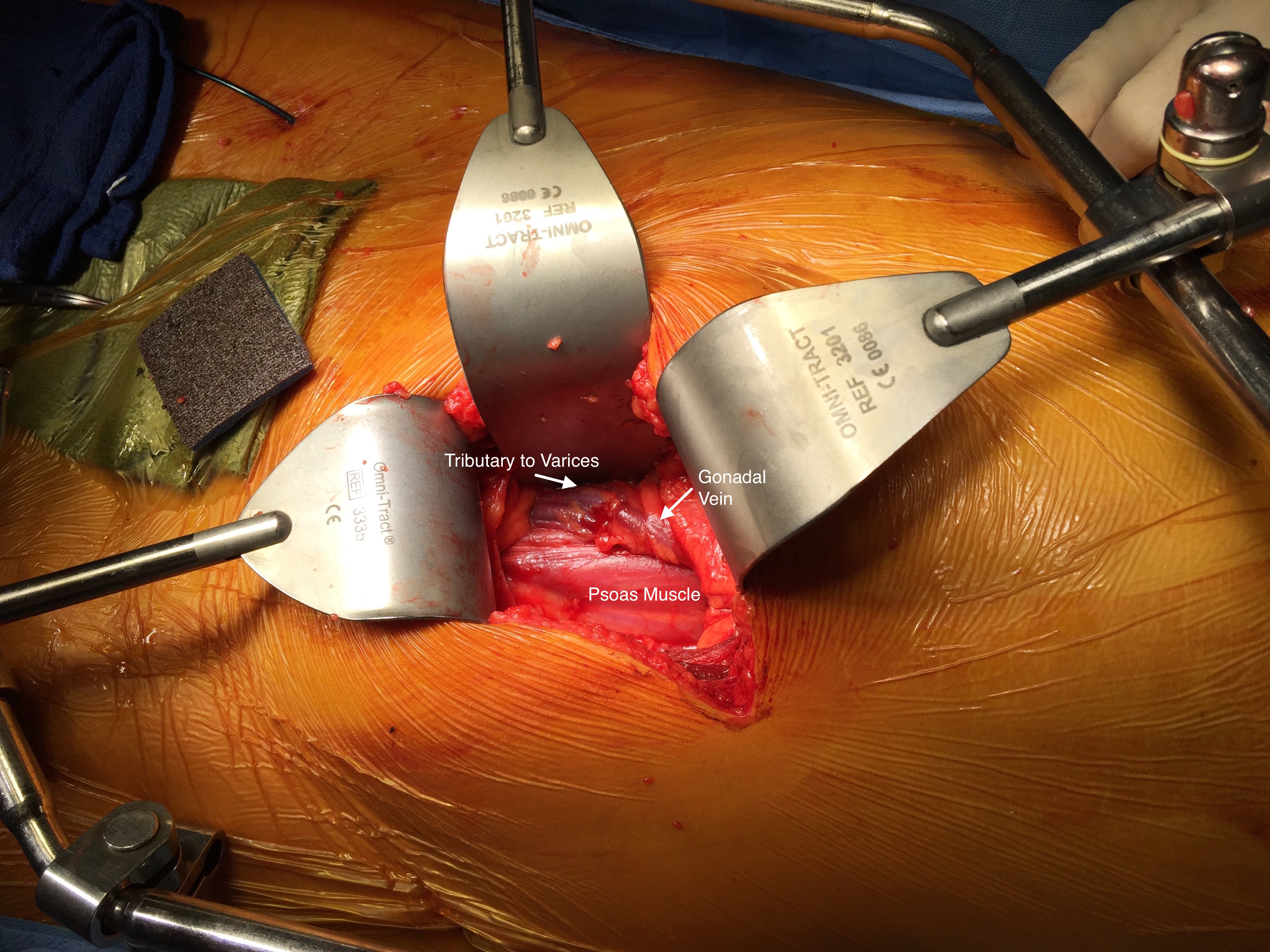
The patient was opened via a generous midline laparotomy. I recruited the assistance of my friend Waleed Hassen, master urologic surgeon here at CCAD, in exposing the IVC. The vessel loop in the picture (below) is around the ureter. Green arrows on the right image show the anterior struts (there are three posterior struts). I had just assisted Waleed recently in removing a metastatic testicular tumor off the IVC, aorta, and mesenteric arteries through the same exposure.

The surprising finding was the anterior hooks of the TrapEase filter had penetrated the serosa of the overlying duodenum. While not perforating, it eventually would have, as the struts had eroded through the wall of the IVC and were outside the adventitial. After heparinizing the patient, the cava was clamped above the filter and I opened the cava lontitudinally along the anterior most strut. I got the sternal wirecutters and removed the anterior three struts along with their hooks. There were large draining lumbar veins which were acting as collaterals that were ligated. The filter was removed internally in pieces leaving the posterior three struts behind as they were outside the IVC lumen. The lower cone can be seen with tissue ingrowth and chronic thrombus. It was dangerous and bloody behind the IVC and I chose to leave these struts behind as they would no longer be pressing into the spine with the filter mostly out. (image below).

The initial thrombectomy was done manually by massaging the vein from either side and up the cava -the thrombus (image below) in the basin on the left expelled as a unit with a spout of blood. Thrombectomy with a #5 Fogarty proved ineffective in the large vessel, and I resorted to using a Foley catheter, directing it left and right, with removal of some more thrombus.

Duplex demonstrated clearance of thrombus from the right iliac vein but adherent thrombus on the left. I placed an 18F sheath into the left femoral vein over a wire I had directed up and over into the right iliac system and advanced the sheath while suctioning -this collapsed the vein and allowed the sheath to scrape the walls of the vein, retrieving the final clot material in the right basin. Duplex confirmed the absence of clot in the left iliac vein. The venotomy was then closed primarily and a completion venogram was performed (below).

The patient recovered and was sent home after a duplex confirmed patency of his leg and iliac veins and IVC. He will be on 3 months of a NOAC for provoked DVT.
Discussion:
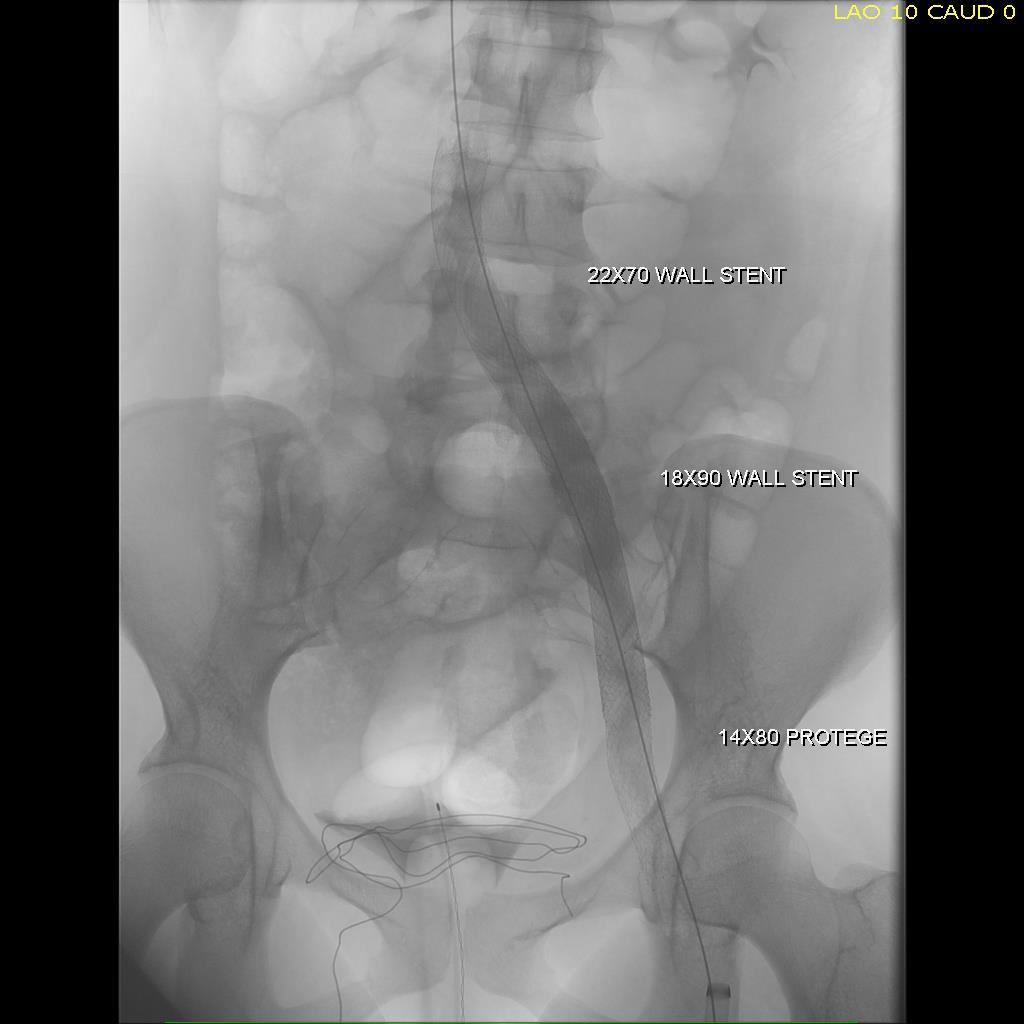
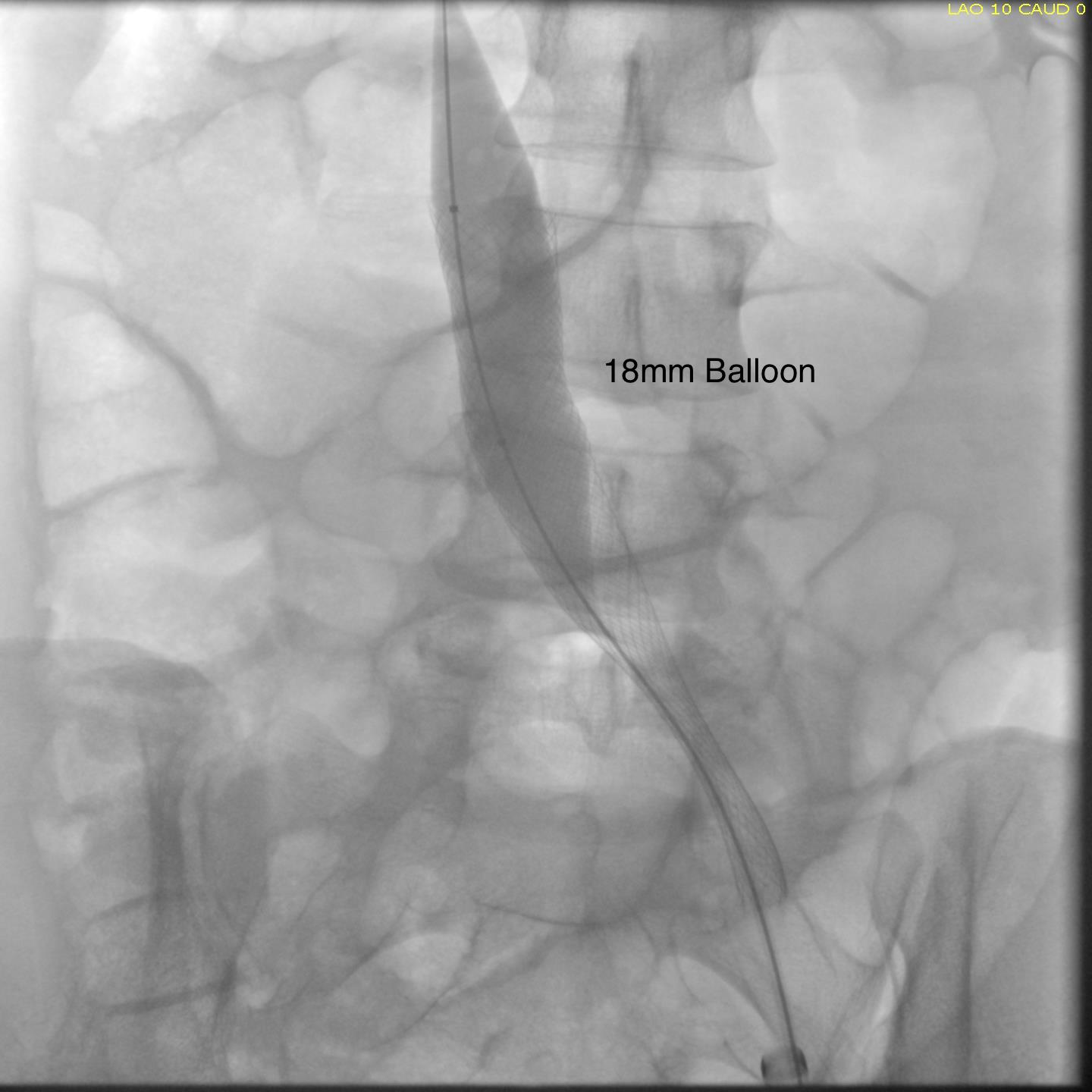
Most of these filters can be retrieved with endovascular techniques. The principle is of gaining control of the top of the cone and collapsing it like an umbrella. For the Cordis TrapEase and OptEase filters, both cones have to be collapsed, and the struts which will have grown into the walls of the IVC have to be be stripped away from the IVC. I had attempted removal of an OptEase with control from above when I was in Ohio, but like in this case, the filter had tissue ingrowth on the lower cone making looping and control of the filter difficult as the hook was encased. While I was able to collapse the filter into a 16F sheath supported through an 18F sheath, it was clear the IVC was invaginating into the sheath and with enough force, I would tear the IVC. It is not the worst thing, perforating the IVC, as it is a low pressure system, and a small perforation is tolerated, but a large one needs operative repair. This can be avoided with surgical removal of the filter.
Performing this in the hybrid suite allowed for complete clearance of thrombus. That said, the thrombus in the internal iliac veins likely did not come out, nor did I seek to clear them. Rather, I will rely on systemic anticoagulation to do this for me.
I use duplex sonography intraoperatively liberally during my procedures. During EVAR, transabdominal ultrasound is sufficient in ruling out or specifying endoleaks. During complex kidney transplantation which I sometimes participate in, duplex is a critical tool for evaluating flow. In this case, images showing a cleared vein (will post, currently stuck in portable ultrasound memory) assured me that I could open the clamps with confidence that pulmonary embolism could be avoided. Gratifyingly, the patient had immediate reduction of leg swelling and can be expected to avoid problems as nothing (such as stents) was left behind in the vena cava lumen.
References
- J-P Galanaud, J-P Laroche, M Righini. J Thromb Haemost 2013;11(3):402-11. doi: 10.1111/jth.12127.



 The celiac plexus is caught under the median arcuate ligament and compressed. It causes a neuropathy that is felt in its visceral sensory distribution and the brain interprets these signals in the typical ways irritation of the stomach is interpreted -as pain, burning, nausea, sensations of bloating, and general malaise. These nerves can be turned off with a celiac plexus block and the effects tested by giving the patient a sandwich. When it works, the patient will say they will have had relief for the first time in years and operation to relieve the ligament compression and ablate the nerve can proceed. Same for many of the diseases listed.
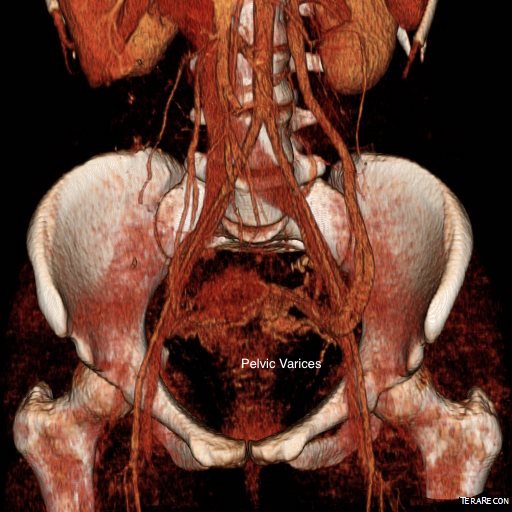
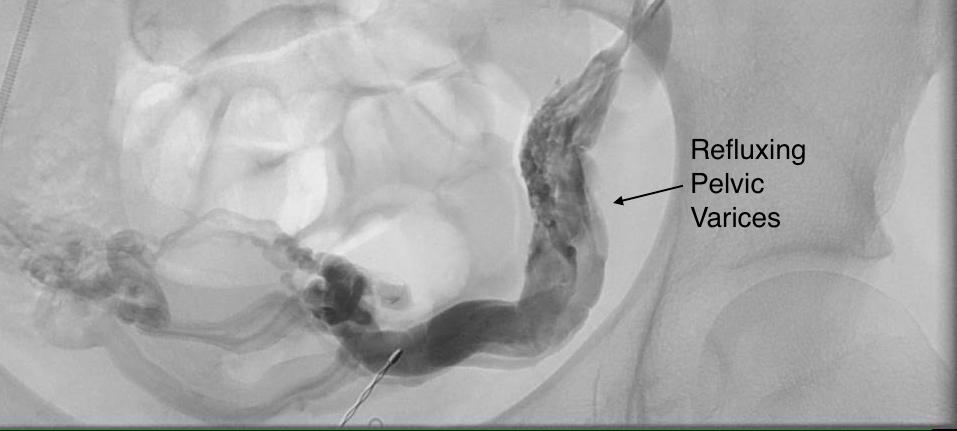
The celiac plexus is caught under the median arcuate ligament and compressed. It causes a neuropathy that is felt in its visceral sensory distribution and the brain interprets these signals in the typical ways irritation of the stomach is interpreted -as pain, burning, nausea, sensations of bloating, and general malaise. These nerves can be turned off with a celiac plexus block and the effects tested by giving the patient a sandwich. When it works, the patient will say they will have had relief for the first time in years and operation to relieve the ligament compression and ablate the nerve can proceed. Same for many of the diseases listed. The swelling from varicoceles which I have also had feel nothing less than feeling the aftereffects of getting a kick in the balls -not the immediate sharp pain but imagine about 5 minutes after with the mild nausea, abdominal discomfort and desire not to move too much, and even a little flank pain. Imagine this occurring low in the pelvis with ovarian vein varices in pelvic congestion syndrome. This kind of swollen gonad pain afflicts many women whose pain is so frequently dismissed by male physicians because they have no context -well imagine getting kicked in the balls hard, wait about 5 minutes and that moment stretch it out to whenever you stand for a long period of time (below).
The swelling from varicoceles which I have also had feel nothing less than feeling the aftereffects of getting a kick in the balls -not the immediate sharp pain but imagine about 5 minutes after with the mild nausea, abdominal discomfort and desire not to move too much, and even a little flank pain. Imagine this occurring low in the pelvis with ovarian vein varices in pelvic congestion syndrome. This kind of swollen gonad pain afflicts many women whose pain is so frequently dismissed by male physicians because they have no context -well imagine getting kicked in the balls hard, wait about 5 minutes and that moment stretch it out to whenever you stand for a long period of time (below).






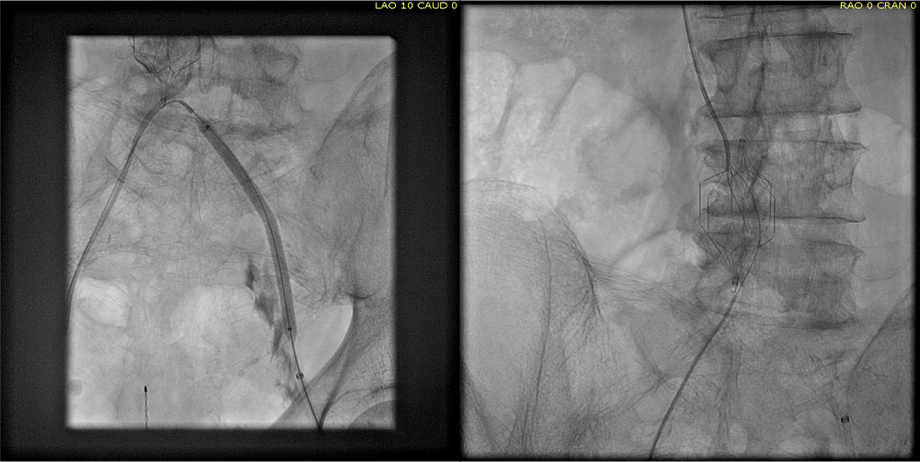


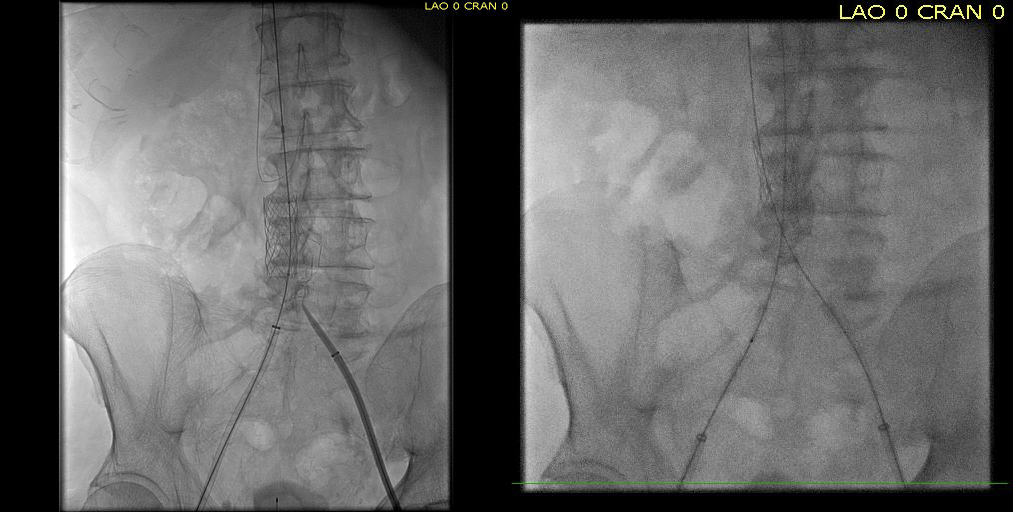
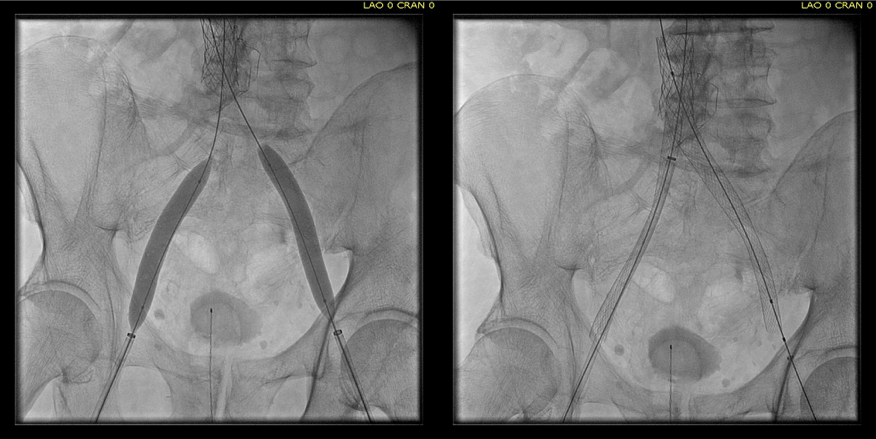













 The patient is a young woman who three weeks prior to presentation developed sudden low back pain and left leg pain while exercising on an elliptical. This pain worsened through the subsequent weeks and she developed fevers, chills, and night sweats, and she came to the emergency department. There, she was found to have left thigh and leg swelling. Duplex revealed a left iliofemoral DVT starting from the iliocaval tributary and extending to her left femoral vein (figure above). A CT scan revealed a pulmonary embolism to the left lung (below). No precipitating factors were present. Vascular surgery was consulted.
The patient is a young woman who three weeks prior to presentation developed sudden low back pain and left leg pain while exercising on an elliptical. This pain worsened through the subsequent weeks and she developed fevers, chills, and night sweats, and she came to the emergency department. There, she was found to have left thigh and leg swelling. Duplex revealed a left iliofemoral DVT starting from the iliocaval tributary and extending to her left femoral vein (figure above). A CT scan revealed a pulmonary embolism to the left lung (below). No precipitating factors were present. Vascular surgery was consulted.