A day in my life
Today, I got up at 630, made some coffee, and Zoomed in on our morning report at our main campus hospital. I have a patient there who I will be operating on tomorrow and wanted to know the current status of the patient. Once the report was over, I brushed my teeth and drove into my hospital which is a regional community hospital. I had an angiogram for a patient who was having a problem with blood flow to the leg. The cath lab was ready to go at 0800, and I was done by 0900, where I quickly ran over to to my office for clinic. My fellow who was doing her community rotation helped me with the angiogram, and then came over to clinic where I saw 27 patients from 0900 to 1600hrs, two of them virtually. At 1600hrs, I had a hospital committee meeting where I am the chief of surgery for my community hospital, and at 1700, was done. I ate a snack as I finished up some paperwork, and got on another Zoom meeting of my institute with over a hundred people to have an update meeting. I then drive to my golf club, and got on the range and hit golf balls for 30 minutes, then got on the putting green and practiced for another 30 minutes. Then I drove home and had dinner with my family. I watched highlights of a football (American) game I recorded over the weekend, while reading email, then sat down to write this before I showered and went to bed.
This week I have 9 cases scheduled -several angiograms and interventions, a leg bypass, a few fistula creations, and a laparoscopic procedure (I’m one of the few vascular surgeons who do laparoscopic surgery). As I sit in bed, I listen to a journal article read to me by the voice of Gwyneth Paltrow (it’s AI) -I find it easier than actually reading the thing, and then I watch a few TikToks, read Reddit, and then go to sleep around 2300h. Cycle starts again in the morning, but will wake at 0530 to get to the main campus hospital to perform an operation. Arriving at main campus on a Wednesday, we have a combined grand rounds with the whole Surgery Department prior to operating.
Lifestyle
On weekends, when I am not on call, I still catch up on my patients from a report from my trainees or my nurse practitioner who makes rounds. I even do this sometimes when I’m out of town. Usually, I play competitive golf with members at my club -the more pressure the better. I find competition to be relaxing. Afterwords, I come home and write, read a little, and watch sports depending on the season or golf. My writing is sometimes work-related, sometimes in my journal. I kept a personal blog for over ten years on golfism.org. I am working on a novel -have been for a decade but not making much progress. I read mostly nonfiction but will listen to audiobooks of science fiction -currently marching through all the Dune prequels written by Brian Herbert, the son of Frank Herbert, the author of Dune and its original sequels. I am working on a grand unifying theory of circulation.
Procedures
As a vascular surgeon, I perform operations in the traditional open fashion, and endovascular procedures which are a done with imaging from x-ray. Occasionally, I do laparoscopic surgery. The open surgical procedures include operations on the aorta and its branches, and on arteries in the legs, arms, and neck. I also work on veins throughout the body. The patient arrives with a set of conditions, a prior history, and an examination, and given a problem, you evaluate it with various tests which can be blood tests, vascular tests, imaging studies like X-ray, Ultrasound, Vascular Lab Studies, CT scans and MRI’s. This is called the workup -getting data to plan a procedure. Knowledge of anatomy and physiology and biomechanics of flow are crucial to put together a plan that will be successful in treating the disease with low complication rate and good durability. The procedures require a great deal of planning and often I include my colleagues within my department and those in other specialties to get their insights for making a plan that accounts for the reason for operation, plan for operation, contingency plans, and recovery in the hospital, and healing outside the hospital. You can see some of these cases on my blog, vascsurg.me.

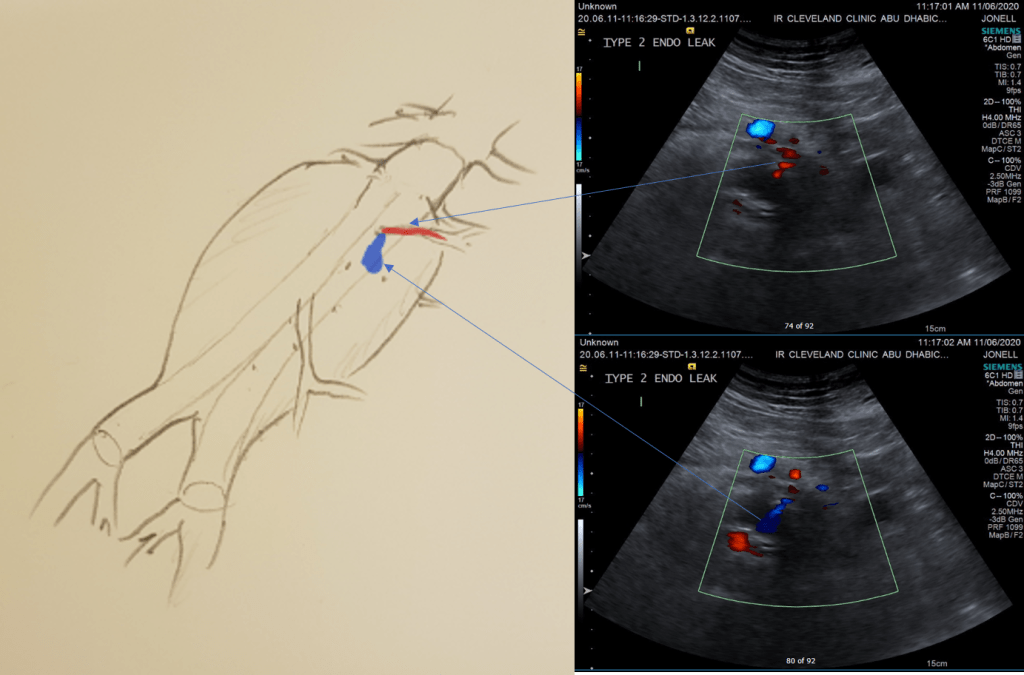
The image above shows a common femoral artery aneurysm presenting as a pulsatile mass in the right groin. The first image on left is an arteriogram (a sketch of one) that I would get prior to surgery. The patient is also suffering from pain in the right leg due to a lack of blood flow because his superficial femoral artery (SFA) is occluded and his profunda femoral artery (PFA) is open but has a blockage at its origin where the aneurysm ends. I plan the surgery and execute it. During surgery, things may pop up -good things like finding an otherwise pristine SFA filled with plaque. Removing the plaque, it becomes a great conduit for replacing the aneurysm and avoids using an expensive graft which can become infected -your own tissues fight off infection better than graft.
In the F1 Movie, Brad Pitt’s character describes a sense of pure driving, being in the flow, being completely at peace on the road. The best moments in surgery, I reach a flow state where actions follow one after the other. It’s a form of spiritual ecstasy, to be completely focused and present. Even better is having the patient do well -to be able to walk without pain and the fear of possibly losing a leg or dying.
Who should not do vascular surgery. By definition, anyone not trained in vascular surgery. Successful vascular surgeons come in all shapes and sizes, but they share common traits -grit, focus, some intelligence, and hand-eye coordination. That would mean those who give up easily, have trouble with focus, are unintelligent, and have poor dexterity should not go into vascular surgery. The saddest cases are when the desire to be something does not match up with the reality. It is possible for non-vascular surgeonsto make a living doing a focused practice around varicose veins for example, but a good vascular surgeon is hard to create. Also, you should not do this for money or prestige, there are easier ways to get money or prestige.
Who should go into vascular surgery. Anyone who thinks they might like it should certainly look into it. The best way is to directly observe a vascular surgeon at work. That is the whole purpose of the rotations in medical school. Sadly, many medical schools do not offer much time in a surgery rotation and vascular surgery exposure is inconsistent. Our society has been working hard for over a decade to improve this and we are seeing it in the excellent applicants to our training programs. The best candidates are driven people with a track record of academic excellence, but the qualities that make a good surgeon are harder to define. Desire alone is insufficient and sadly academic excellence, while it will get you into the door, doesn’t predict who will be a great surgeon. There has to be grit -an ability to persist despite hardship. There has to be a nimble mind that can solve problems quickly. And there has to be the physical hand skills that define surgery but somehow have been dropped from the initial evaluation of candidates for surgery.
Who should not go into surgery. Based on my answers above, those quick to give up, are unintelligent, and poorly coordinated should not go into surgery. I would add to this lazy, dishonest, and sociopathic. No criminals please.
There is no perfect answer to this. I knew a fellow who did not score well on tests and was rejected from medical school five years in a row, but eventually got in and completed a residency in a surgical subspecialty and has a very successful practice. While he was being rejected from medical school, he spent five years in the lab, and he could do open heart surgery on dogs very well, was coauthor on numerous papers, and his surgical skill was excellent -like if you were stuck taking tennis lessons from a professional for five years but never playing an actual game. There are also many examples of people who were told too late that they were no good for surgery.
What you should not do is listen to just a single person who has a poor opinion of you. You should examine the situation and decide if there is some truth to the issue, but you need at least three opinions. For example, I would like to be a professional golfer. I can get at least three people to tell me honestly that this is a bad idea. I would like to be a writer. I can get at least three people to tell me honestly this is a good idea. You get the picture. In medical school you will rotate and work with many people and you will have grades and feedback. You need to get honest opinions as you move forward. You need to study hard and get great grades because no matter what you do, your patients will be depending on you.