The patient is a young father in his early thirties who complains of severe headaches, dyspnea, and inability to function or interact with his toddler. He complains of visual disturbancers when bending over and has dilated neck veins when recumbent. He was born with aortic stenosis and has undergone three aortic valve replacements and has had pacemakers since childhood. His most current pacemaker is a DDDR pacemaker in his left subclavian venous position, but he has two dead leads in his right subclavian vein. His symptoms started in his twenties but has worsened sharply over the past year. An effort was made to remove his dead leads at a tertiary referral center, but they couldn’t.
On examination, he had significant findings from dilated neck and supraclavicular fossa veins, a left chest pacemaker, and scars from his sternotomies. Duplex showed a patent bilateral internal jugular veins (IJV) with minimal respirophasic variability.
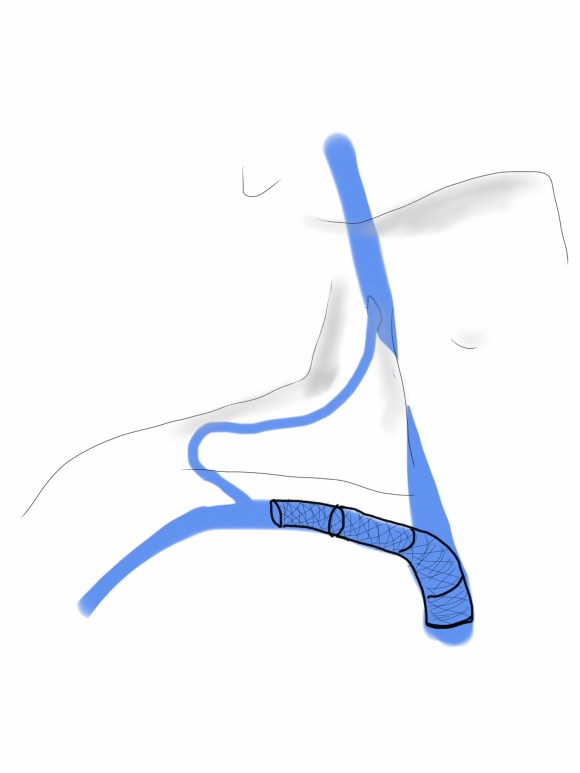
Venography (composite, above) showed superior vena cava occlusion consistent with his diagnosis of SVC Syndrome. I measured venous pressures, which we normally don’t do, because I was curious about them. Right IJ access with ultrasound with a sheath revealed pressures on the monitor in CVP mode of 22mmHg. Access from the groin into the heart revealed a right atrial (RA) pressure of 14mmHg. The SVC was crossed from below into the right IJV, and the SVC was dilated with a balloon -no stenting was possible because of the presence of the leads. His pacemaker did not fail during the procedure. After balloon dilatation, contrast passed easily from his IJV into his RA (below). His pressure in the R. IJV was 17mmHg and in the RA was 16mmHg.

He immediately felt relief and in 1 month followup had sustained symptom relief with patent flows seen in right IJV. He understood it was likely he would need regular reintervention when symptoms returned.
Discussion:
While IVUS is usually the way to check on patency after venous intervention, the presence of permanent pacemaker leads, including a set of nonfunctioning leads from the right SCV, made its use moot -venoplasty to 12mm was done in multiple stations and there was to be no stenting which is the usual next step if IVUS found residual stenosis. The more interesting finding here is the pressures measured in the IJV. The symptoms of SVC syndrome come from venous hypertension, but we rarely if ever measure pressure. Intracranial pressure (ICP) which is used to monitor for critical hypertension in the cranium typically after surgery or trauma, is considered high if over 15mmHg, moderately elevated over 20mmHg, and severe when over 40mmHg. ICP is greater than or equal to intracranial venous pressure, and in patients with SVC occlusion, this pressure is equal to the IJV pressure. With an IJV pressure of 22, this patient’s ICP was likely over 20mmHg, explaining his incapacitation.
IJV pressure is easily obtained and may justify urgent intervention in acute SVC syndrome, although more data needs to be obtained. This patient will benefit most from a more durable solution, but for now, he is very pleased to be able to take part in the care of his young child.