A body floating in space, a fetus in the womb, a dad lounging in his favorite chair, share the feature of weightlessness and represent the absolute neutral position (figure above) of the human which is the position of a relaxed supine quadruped -a dead mouse. Anything else is a stress position, including standing. Repeating motions outside of this relaxed pose or holding those positions away from this absolute neutral for long periods of time is a nidus for injury and pain. That is why most land animals sleep flat on the ground.
The Pain Operations
Operations to relieve pain are often the most gratifying to both patient and surgeon to perform successfully. This circumstance applies to the commonly performed procedures such as spine surgery, endometrial ablations, and varicose vein resections. When the pain is due to a rare set of circumstances, things are not so easy. Typically for rarer pain syndromes, two things need to coincide for the successful operation to happen. First is the patient must suffer while more common and potentially life threatening diseases are ruled out and even treated if these are found. This may take months or years. The second necessary condition is finding a physician who has seen the particular pain syndrome before and understand how to test for it and treat it. That meant the majority of people never get treated, or are shunted into the circle of shame as malingering, drug seeking, and mentally unstable. The opioid epidemic creates double jeopardy for these patients -they can become addicts as their pain is never successfully diagnosed and treated and they get labeled as drug seeking.

All pain syndromes that can be successfully treated share common features that give you a degree of surety about the diagnosis, but at the end, there is a leap of faith on the part of both patient and practitioner because many of these operations have a failure rate ranging from 5-20 percent. First, the symptoms must be associated with sensory nerves, somatic or visceral. Second, there is a physical mechanism for that nerve to be inflamed from compression, swelling, or irritation that can be accounted for through history, physical examination, and imaging studies. Third, though not a constant, a major nerve trunk will be associated with a blood vessel, typically and artery, that is also affected by compression. Fourth, when swollen veins are the cause of pain, it has to be recognized that at an end stage the organ that the veins drain can also be affected.
The Pain Must Have a Testable Anatomic Basis
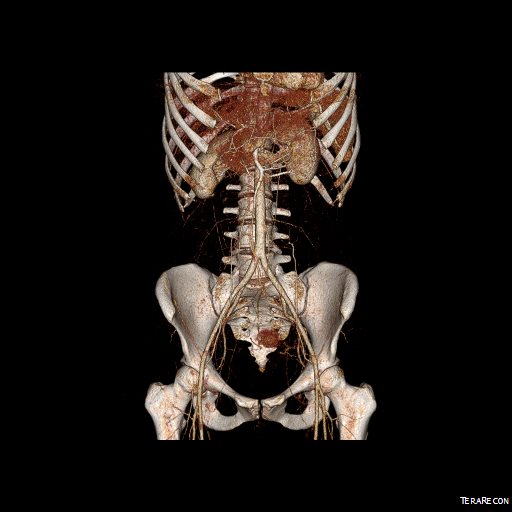
The somatic sensory nerves in the periphery are well mapped out and known since even classical times. The described pain should be consistent with a nerve. The best and easiest example is a neuroma that forms in an amputation stump. It triggers pain in its former distribution. It is palpable as a nodular mass. It is visible under ultrasound or cross sectional imaging. And it is easy to turn off temporarily with an injection of lidocaine, either under palpation or image guidance. If you can turn off the nerve and relieve the pain, it is likely that ablating or relieving the nerve of irritation will also relieve the pain. Such is the case in median arcuate ligament syndrome (figure below).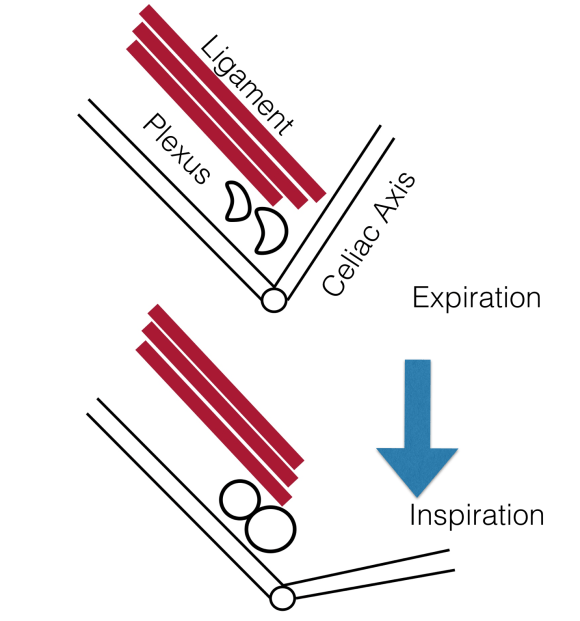 The celiac plexus is caught under the median arcuate ligament and compressed. It causes a neuropathy that is felt in its visceral sensory distribution and the brain interprets these signals in the typical ways irritation of the stomach is interpreted -as pain, burning, nausea, sensations of bloating, and general malaise. These nerves can be turned off with a celiac plexus block and the effects tested by giving the patient a sandwich. When it works, the patient will say they will have had relief for the first time in years and operation to relieve the ligament compression and ablate the nerve can proceed. Same for many of the diseases listed.
The celiac plexus is caught under the median arcuate ligament and compressed. It causes a neuropathy that is felt in its visceral sensory distribution and the brain interprets these signals in the typical ways irritation of the stomach is interpreted -as pain, burning, nausea, sensations of bloating, and general malaise. These nerves can be turned off with a celiac plexus block and the effects tested by giving the patient a sandwich. When it works, the patient will say they will have had relief for the first time in years and operation to relieve the ligament compression and ablate the nerve can proceed. Same for many of the diseases listed.
Tight Spaces Impinging Nerves, Arteries, and Veins
Many of the tight spaces involving the nerves have accompanying arteries that are compressed. This results in injury to the artery in the form of intimal hyperplasia, post stenotic dilatation, aneurysm formation, and thromboembolism. Shared tight spaces that cause problems for nerves and arteries have the common features of fixed ligaments, adjacent bones and muscles, inflammation, and motion. These include the thoracic outlet, antecubital fossa, cubital canal, diaphragmatic hiatus at median arcuate ligament, inguinal ligament, popliteal fossa, carpal tunnel, obturator canal, mediastinum, retroperitoneum -basically anywhere nerve, compression, and motion occur. In some instances of median arcuate ligament syndrome, postures and breathing trigger the pain. Holding a child in an arm may trigger pain in neurogenic thoracic outlet. Or sitting while wearing tight jeans may trigger a burning pain in meralgia paresthetica. It is not uncommon to find damaged arteries in median arcuate ligament syndrome, thoracic outlet syndrome, and popliteal entrapment or thrombosed veins in nutcracker syndrome, May-Thurner Syndrome, and Paget-von Schroetter Syndrome. Because nerves are typically difficult to visualize, their compression may only be inferred by testing for compression in their adjacent arteries.
Dilated Veins and Swollen Organs and Visceral Pain
Venous hypertension is most commonly conceived of as varicose and spider veins of the legs and offer a model of pain when applied to other pain caused by venous dissension. The visceral sensory fibers veins and arteries trigger a very intense pain that localizes to the trigger. I have often witnessed this when I manipulate a blood vessel during local anesthesia cases. Visceral pain from swelling has a dull achiness that is localizable to my spider veins after a long day standing like a bruise (below). The swelling from varicoceles which I have also had feel nothing less than feeling the aftereffects of getting a kick in the balls -not the immediate sharp pain but imagine about 5 minutes after with the mild nausea, abdominal discomfort and desire not to move too much, and even a little flank pain. Imagine this occurring low in the pelvis with ovarian vein varices in pelvic congestion syndrome. This kind of swollen gonad pain afflicts many women whose pain is so frequently dismissed by male physicians because they have no context -well imagine getting kicked in the balls hard, wait about 5 minutes and that moment stretch it out to whenever you stand for a long period of time (below).
The swelling from varicoceles which I have also had feel nothing less than feeling the aftereffects of getting a kick in the balls -not the immediate sharp pain but imagine about 5 minutes after with the mild nausea, abdominal discomfort and desire not to move too much, and even a little flank pain. Imagine this occurring low in the pelvis with ovarian vein varices in pelvic congestion syndrome. This kind of swollen gonad pain afflicts many women whose pain is so frequently dismissed by male physicians because they have no context -well imagine getting kicked in the balls hard, wait about 5 minutes and that moment stretch it out to whenever you stand for a long period of time (below).

When a limb is swollen from a thrombosis, the veins hurt and is similar to a bone pain from a fracture or a pulled muscle -that is how the brain processes the pain, but when the muscles and skin get tight from edema, the pain is sharp and dire. This is the same kind for pain from a distended left kidney from nutcracker syndrome or a spleen from a splenic vein thrombosis. These conditions can be modeled and predicted based on history and correct differential and confirmed with proper imaging -always.
Build a theory of the pain based on a testable proposition and set of nerves
That is the final message. These pain syndrome require some imagination and empathy to map and model. Predictive tests then can be performed on physical examination, functional testing, or imaging. Often, the adjacent artery is the only thing that can be reliably visualized and tested, knowing that it is the nerve that is compressed. Turning off the offending nerve with a block and relieving the pain is the most powerful argument for operating. It is building the argument for an operation that requires these objective data, but at the end, it does require some experience and faith. You have to believe in your patient and the science and when they coincide, you have to act.