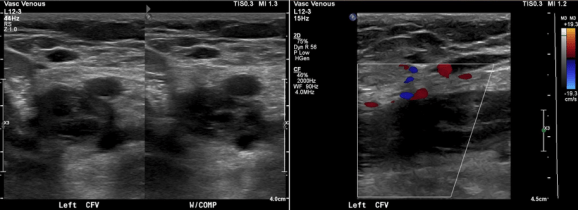
 The patient is a young woman who three weeks prior to presentation developed sudden low back pain and left leg pain while exercising on an elliptical. This pain worsened through the subsequent weeks and she developed fevers, chills, and night sweats, and she came to the emergency department. There, she was found to have left thigh and leg swelling. Duplex revealed a left iliofemoral DVT starting from the iliocaval tributary and extending to her left femoral vein (figure above). A CT scan revealed a pulmonary embolism to the left lung (below). No precipitating factors were present. Vascular surgery was consulted.
The patient is a young woman who three weeks prior to presentation developed sudden low back pain and left leg pain while exercising on an elliptical. This pain worsened through the subsequent weeks and she developed fevers, chills, and night sweats, and she came to the emergency department. There, she was found to have left thigh and leg swelling. Duplex revealed a left iliofemoral DVT starting from the iliocaval tributary and extending to her left femoral vein (figure above). A CT scan revealed a pulmonary embolism to the left lung (below). No precipitating factors were present. Vascular surgery was consulted.

Plan was for catheter directed thrombolysis. Venography from the patient’s popliteal vein via a short saphenous access revealed thrombotic occlusion from the left common femoral confluence to the iliocaval confluence. The thrombus was crossed, and ballooning showed there was chronicity to the occlusion in the pelvis evidenced by waisting of the balloon on inflation. A multihole infusion catheter was placed across the thrombus from the thigh to the inferior vena cava and recombinant tissue plasminogen activator was infused overnight.

The venogram from the popliteal vein showed a patent popliteal and femoral vein and the goal of this procedure became opening the common femoral vein and its confluence of multiple veins from the thigh, to connect it via stents to the vena cava (second image below).
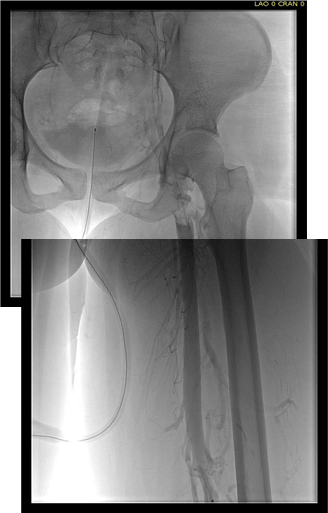

Clinically, there was no change overnight and when the patient was restudied next morning, there was still an occlusion starting at the common femoral vein.

At this point, I had a choice as to what to do next. First, I could stop, and have the patient start anticoagulation and return several months later -often, the common femoral vein returns to drain into pelvic collaterals. As I had discussed in an earlier post, venous interventions are no different from arterial ones in that inflow, draining vein, and outflow have to be considered. In the case of the veins, I like to think of it as connecting major confluences, and for a leg, the common femoral venous confluence is paramount.
Intervening from the popliteal vein to the vena cava is inferior to being able to connect draining veins at the common femoral confluence to the vena cava. So getting the common femoral vein to patency is critical, and can sometimes be achieved with anticoagulation and time. The second option is to break out a thrombectomy catheter and try to remove the thrombus by various machinations, ie. gadgets and novel catheter systems of which there are many. I felt that given the three week time course of the thrombus, the best we could get was some clearance of thrombus, leaving behind a complex network of chronic thrombus and fibrinous scar with the overnight lysis. I didn’t even try this second option and the thrombectomy machine stayed unplugged, the fancy (and expensive) catheters left hanging. The third option, surgery, was not indicated as the patient did not have signs of phlegmasia, and for the same reasons that the lysis didn’t work, opening the common femoral vein for an endovenectomy has uneven outcomes.
The fourth option, mechanical aspiration sheath thrombectomy (MAST), is a technique developed by Dan Clair, our former chair. As a concept, it is very simple. A large sheath (>12F) is introduced and the thrombus is aspirated while the bare sheath is advanced over a wire. The sheath is then removed and the contents emptied. For this case, an 18 F sheath was introduced into the femoral vein in mid thigh.

The blood is ejected into a basin and a cell saver (in non-malignant cases) is used to salvage the whole blood.

This reopened the common femoral vein. This was for me a very important step as without achieving this, I would have had to stent into the femoral vein, excluding many smaller veins draining into the common femoral vein, and effectively basing my revascularization off the popliteal vein confluence, an inferior inflow source for venous revascularization.

With the common femoral vein open, placing stents from the vena cava to the common femoral vein was straightforward and described elsewhere (reference). The iliac vein remained closed due to the chronic thrombosis, which was clinically May Thurner’s Syndrome, and was stented.

Three things deserve comment: the vena cava and iliac veins need to be dilated up to 18mm, and larger for the cava. This is disconcerting, but size does matter. Second, IVUS is critical in confirming that everything is open. Third, the 14mm nitinol stent placed into the common femoral vein will stay open, unlike a stent placed into the artery across the inguinal ligament. It likely has to do with the deeper position of the vein in relation to the artery which protects the vein from the ligament. We don’t have the large diameter nitinol stents designed for iliocaval venous revascularization yet, but the available stents do a good job.

The patient was discharged on anticoagulation with resolving edema in the left leg and thigh. At one month followup, duplex confirmed wide patency of the stents and IVC and no new DVT.
Discussion: MAST illustrates a critical issue for all innovation in the current setting of resource limitation. Innovations must be made with not just a consideration to efficacy and potential market, but also cost. The large sheaths used in MAST are commonly available and cheaper by multiples of tens compared to the thrombectomy systems and catheters. Unpublished data reviewing 13 patients undergoing MAST with a mean followup showed 69% with complete thrombus removal, 31% with subsegmental removal, no operative mortality, and 92% primary patency at an average of 79 days of followup, all with symptom improvement (Clair, correspondence). Other groups have reported similiar results using “large catheters” (reference 2), but nothing can compare to an 18F sheath in clearing the iliocaval system.
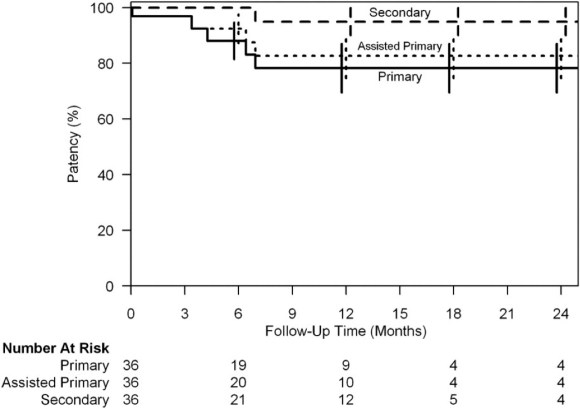
The patient can expect to have excellent patency in the short to mid term (reference 1).
References
- Titus JM et al. J Vasc Surg. 2011 Mar;53(3):706-12.
- Chung HH et al. Vasc Endovascular Surg. 2016 Jul;50(5):321-7
