It was only last month when I came across a post of an aortic aneurysm in a difficult spot (link) and I couldn’t help chiming in some comments. Reading it now, I sound insufferable, because I wrote,
“Depends on etiology and patient risk stratification. Also assuming aneurysm goes to level of SMA and right renal artery origins and involves side opposite celiac. Options depending on resources of your institute: 1.Open repair with cannulation for left heart bypass and/or circulatory arrest 2.Open debranching of common hepatic, SMA, R. Renal from infrarenal inflow and then TEVAR 3.FEVAR from custom graft from manufacturer on protocol 4.Parallel grafting to CA, SMA, R. RA with TEVAR 5.Surgeon modified FEVAR 6.Open Repair We would have a multidisciplinary huddle around this patient -Vascular, Cardiac Surgery, Cardiology, Anesthesia, and ID (if needed) to help choose. Be ready to refer to a center with more resources -including prepping patient for transfer and imaging -including uploading images to the cloud for transfer with patient’s permission. More info please”
More Info Please, Indeed
The post I commented on was of a saccular aneurysm in the transdiagphragmatic segment of aorta. Ironically, only a few weeks later, I got called from hospital transfer center about a patient with a leaking aortic aneurysm, a type V thoracoabdominal aortic aneurysm as it turned out, from an outside hospital, needing urgent attention, and we accepted in transfer based on the conversation I had with the tranferring physician. And that was the problem -usually in taking these inter-hospital transfers, you have to pray that the precious CT scans come along with the patient burned correctly onto a CD-ROM. You can buy and watch a movie in 4K resolution over the internet -about 4 gigabytes, but a patient’s CT scan which is about 200 megabytes -because of various self imposed limits, overly restrictive interpretations of laws, and lack of computer skills, these life saving images get transferred on CD in 2019. That last time I purchased a CD for anything was over 15 years ago.
An Interested Party
The technical solution –to use the internet to transfer critical life saving information between hospitals – came about when our IT folks took an interest in my quarterly complaint email about using the newfangled internet for sharing files. After mulling various solutions ranging from setting up a server to using commercial cloud solutoins, we came upon the compromise of using our internal cloud with an invitation sent to the outside hospital. I would send this invitation to upload the DICOM folder of the CD-ROM to an outisde email address. It was simple and as yet untried until this night. “Would the patient agree to have his CT scan information transmitted to us electronically?” I asked the other physician. He assured me that the patient was in agreement.
It Takes Two to Tango
Of course, being able to transfer these pictures requires a willing partner on the other side, and the referring physician made it clear he did not have the technical expertise to do so. It took a bit of social engineering to think about who would have that ability. Basically, aside from myself, who spend all their time in dark rooms in the hospital in front of giant computer monitors? The radiologists! I got through to the radiologist who had interpreted the report and explained the simple thing I needed. Gratefully, he agreed, and I sent him a link to our cloud server. I explained to him, “When you receive this, clicking the link opens a browser window and then you open the CD-ROM and find the DICOM folder and drag and drop it on the browser window.” The 200-500 megabytes of data then get sent in electronic form, as it was meant to in 2019.
The Internet Saves a Life
The brutal truth is that in locking down a computer system, many hospitals make it impossible to even load an outside CD-ROM, creating many self imposed barriers to care. Thankfully, at CCAD, we were able to work together to find a secure solution. With the CTA on our servers, I was able to review the study within 15 minutes of accepting the patient, and arrange for the right team to be assembled, and confirm that we had the right material (stent grafts) for treating the patient. When the patient arrived, OR was ready to go, saving hours of time that normally would have been required if the CT scan had to be reviewed from the CD-ROM that came with the patient. Sometimes, the CD-ROM does not come, and in a critical situation, the CTA has to be repeated with some risk to the patient for complications of the contrast and radiation.
What to Do
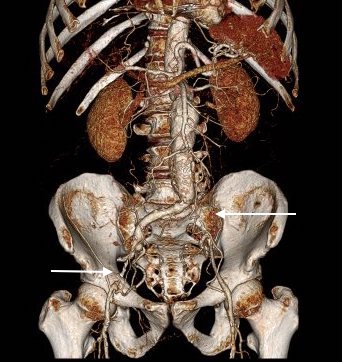
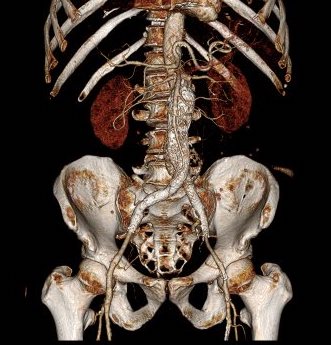
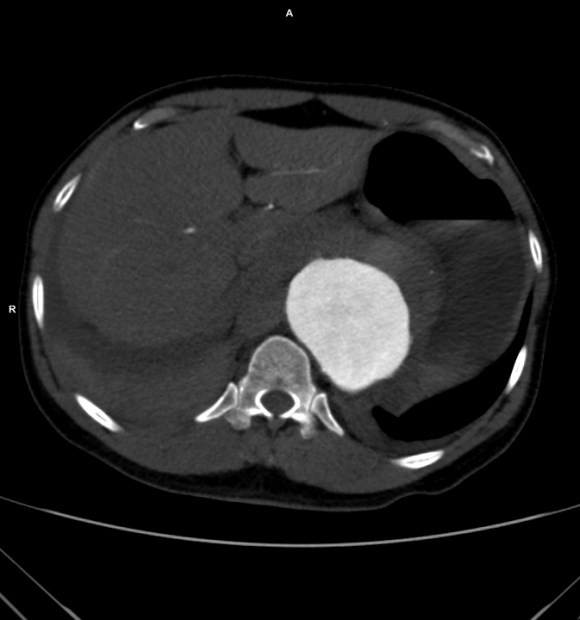
The patient had a 8cm sphere shaped aneurysm arising in the transdiagphragmatic aorta, leaking into the right pleural cavity.

The patient was otherwise a healthy middle aged man with risk factors of smoking and hypertension. The centerline reconstructions showed the thoracic aorta above the aneurysm to be around 20mm in diameter and same below, with the celiac axis and superior mesenteric artery in the potential seal zone of a stent graft. The only plaque seen was around the level of the renal arteries and was focal and calcified. Looking at the list I had made as a comment to the Linked-In post, I realized that I really only had one viable option.

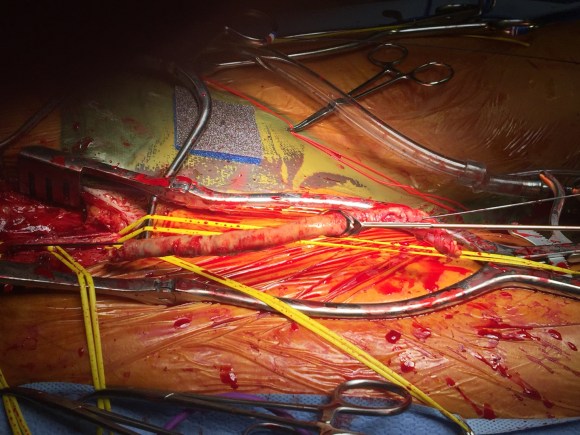
Open repair, usually the most expeditious option, was made challenging by the right hemothorax, making a left thoracotomy hazardous if the lung had to be deflated. Cardiopulmonary bypass would have to be arranged for, and that adds a metabolic hit that greatly raises the stakes. Of the endovascular solutions, the only viable option was TEVAR to exclude the rupture and debranching of the celiac axis and superior mesenteric artery. To those who would advocate for parallel grafts, there was no room in the normal 20mm diameter aorta. And branch systems for rupture are some time in the future. Also, the patient was becoming hypotensive. So the planned operation was first TEVAR to stop the bleeding, and then open surgical debranching. A hybrid repair.

The smallest stent graft we have is a 21mm graft, but it would not be suitable for this aorta. In practice, the normal aorta is quite elastic and will dilate much more than what is captured on a CTA. The next size we have is 28mm graft and I chose this to exclude the rupture, which was done percutaneously.

As seen below, the graft excluded the celiac and SMA. Late in the phase of the final aortogram (second panel) there was an endoleak that persisteed despite multiple ballooning. The timing suggested the intercostals and phrenic vessels contributed to a type II endoleak, but it was concerning.
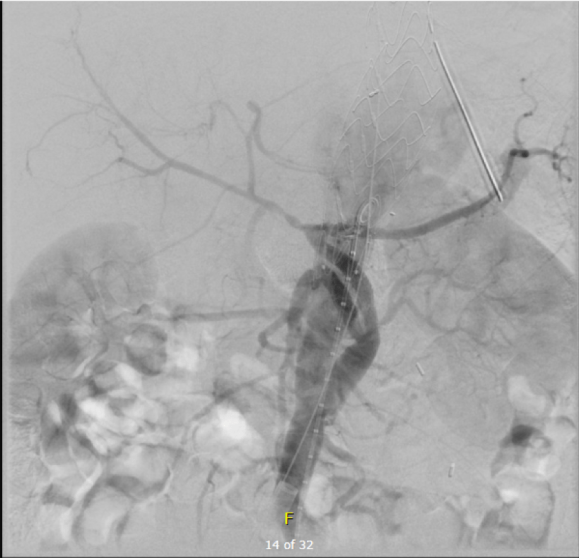
The bypasses were sent from the infrarenal aorta to the common hepatic artery and the SMA close to its origin, and the origins of the celiac axis and SMA were clipped. The bypasses were then done with a 10x8mm bifurcate Dacron graft originally for axillofemoral bypassing. It had spiral rings which I removed at the anastomosis and this resulted in a kink at the closer bypass. Usually, I loop this for iliomesenteric bypass but there was not enough distance from the infrarenal aorta. I have to add a little trick I modified from my pediatric surgery experience as a resident -a Heinecke-Mikulwicz graftoplasty:
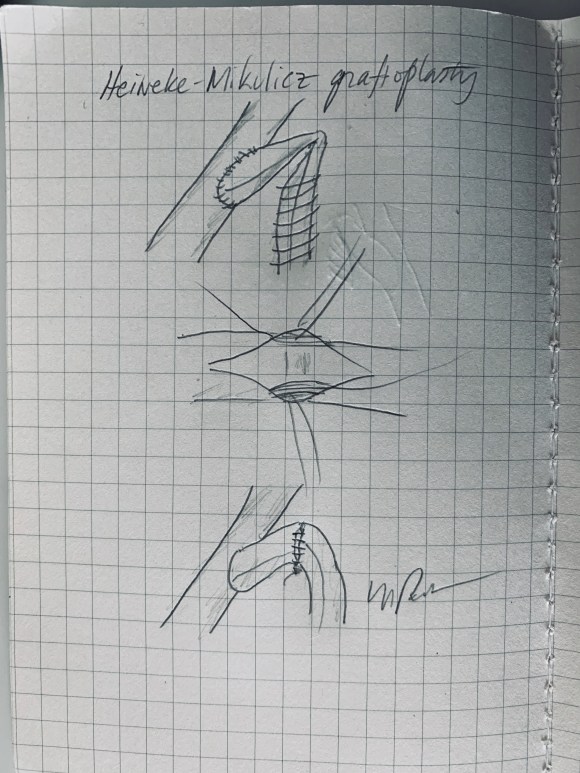
This worked to relieve the kink as evidenced on the aortogram above. After closing the laparotomy, I placed a chest tube in the right chest. The patient had a course prolonged by a classic systemic inflammatory response syndrome, with fevers, chills, and leukocytosis. He bled for a while but stopped with correction of his coagulopathy. All blood cultures were negative, but a CT scan was performed out of concern for the endoleak, and the possibility of continued bleeding.
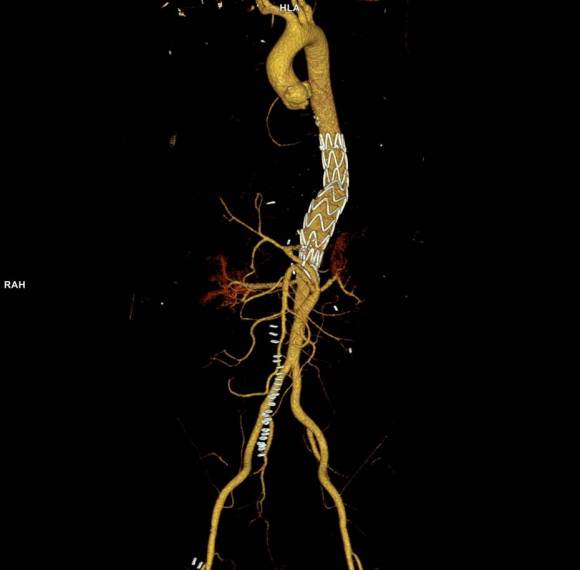
No endoleak was detected as the sac was fully thrombosed. There was a consolidation of the blood in the right chest, but it resolved with fibrinolytic therapy.
Discussion
This case illustrates several points I have been making on this blog.
- Hybrid repairs are not some kind of compromise but the full realization of a complete skill set. When students ask me how to judge a training program, one of the best metrics is how frequently are hybrid operations performed. It means either the endovascularist and open surgical operator are in complete synchrony or there are individual surgeons competent in both open and endovascular surgery. Hybrid operations, rather than being a compromise, are an optimization.
- Time -Laying the stent graft across the celiac and SMA origins starts a warm ischemia clock. The liver and intestines, in my reckoning, should be able to tolerate the 2 hours of work to get the bypasses working. Cardiopulmonary bypass may give you less ichemia but at the metabolic cost of the pump time. These negative factors add up, but were surmounted by the fact that early control of hemorrhage was achieved. Stopping the bleeding and restoring flow are the core functions of vascular surgery.
- Planning and preparation. The ability to see the CT images and prepare the teams and materiel before the patient transferred was lifesaving. This is where our IT gets credit for responding to a critical need and formulating a solution that meets internal policies, external regulations, and saves a life. It illustrates so many opportunities particularly with electronic medical records and their processes which focus more on documentation for billing. A discharge summary should be multimedia like this blog post and it should be normal and easy to generate. And finally, as clinicians, we should mind technology with as much attention as we give to the latest medical devices and techniques.


 which in my experience is highly morbid despite how stable the patient was. Point again to trainees, this is no different from someone having stabbed this patient with a knife at the base of the neck. My options were:
which in my experience is highly morbid despite how stable the patient was. Point again to trainees, this is no different from someone having stabbed this patient with a knife at the base of the neck. My options were: