From Dr. Dmitirios Virvilis, formerly my trainee, now my colleague, who had this interesting case of end-stage iatrogenic pseudoaneurysm.
History
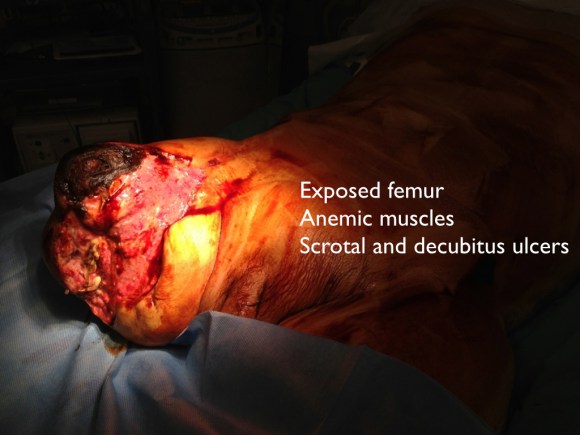
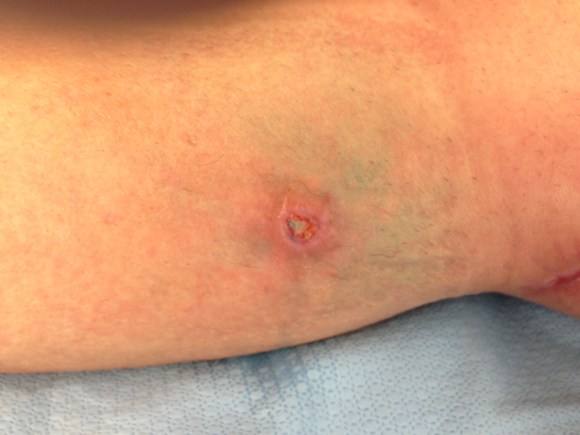
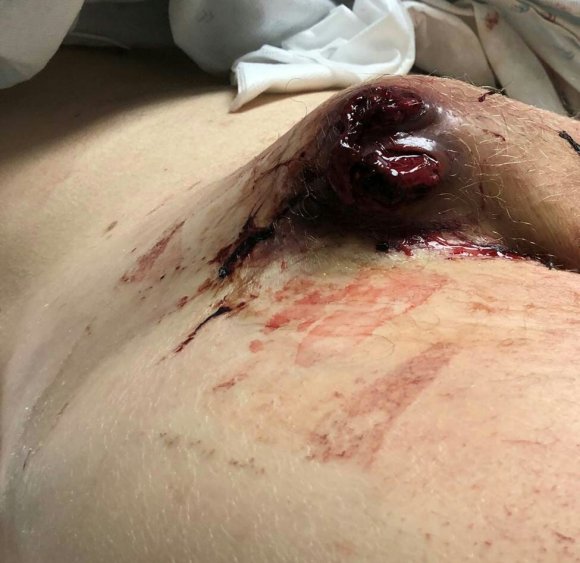
85-year-old gentleman with history of atrial fibrillation on anticoagulation, mechanical valve on Coumadin presented to our emergency department with significant hemorrhaging from the right groin (figure above). The patient had a coronary angiogram 2 months earlier done elsewhere complicated by a pseudoaneurysm which was managed with ultrasound guided thrombin injection that was not successful. Patient was sent to a vascular surgeon at another facility and the decision was made to intervene with the placement of a covered stent on the common femoral artery (possibly due to high patient risk?) compromising the junction of the SFA and profunda artery. The patient developed a large hematoma following placement of the covered stent and was managed solely with antibiotics for over a month prior to presentation.
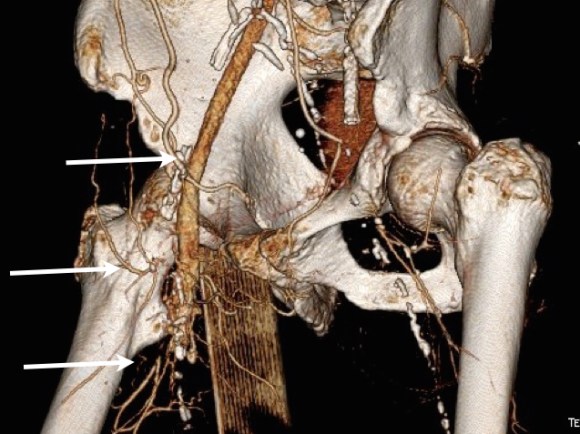
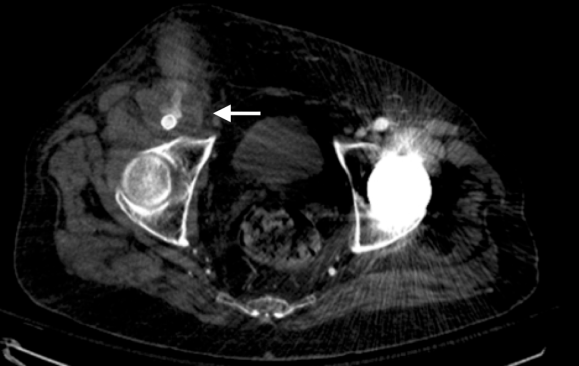
On physical examination, the patient was septic, lethargic with grossly infected groin (image above). A CT scan with contrast was performed which showed active extravasation with stranding around the femoral vessels (below).
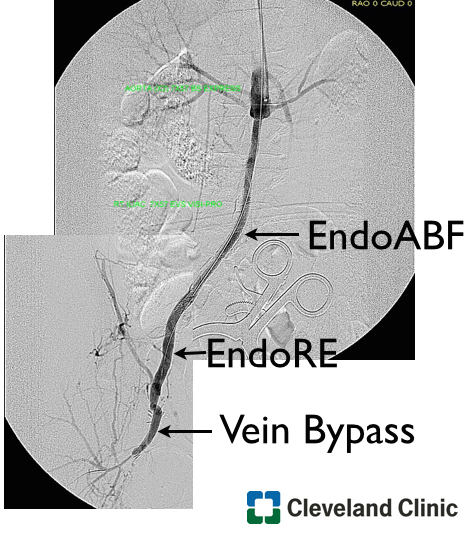
The patient was taken emergently into the operating room. Due to the extent of the infection under the inguinal ligament I obtained proximal control by performing a retroperitoneal flank incision. The iliac vessels were controlled and then I proceed with exploration of the hematoma. The junction of the superficial femoral artery and the profunda artery was separated and the common femoral artery was liquefied. There was only a very thin posterior layer of the common femoral artery and the cover stent was floating in the hematoma. The wound was thoroughly debrided. The distal external iliac artery was transected and the stent was removed. The junction of the superficial femoral artery and profunda artery were separated. A Rifampin soaked graft was used (picture below), which was an 8mm gelatin impregnated Dacron graft soaked in Rifampin (600mg in 200mL of saline) for 20 minutes.

The graft was anastomosed to the common iliac artery to fully exclude the external iliac artery which was debrided and ligated. The profunda artery was reimplanted on the side of the graft in an end-to-side fashion. After the wound was thoroughly irrigated and the graft was covered . The muscle flap was created in the usual fashion with lateral mobilization preserving the medial vascular pedicle. The wound was partially approximated and the top of the wound was left open for an application of wound VAC (below). The fact that I am practicing in a remote area has made me to be more efficient and incorporate in my practice many procedures picked up through training that fall under general surgery, plastic surgery, and advanced vascular surgery.

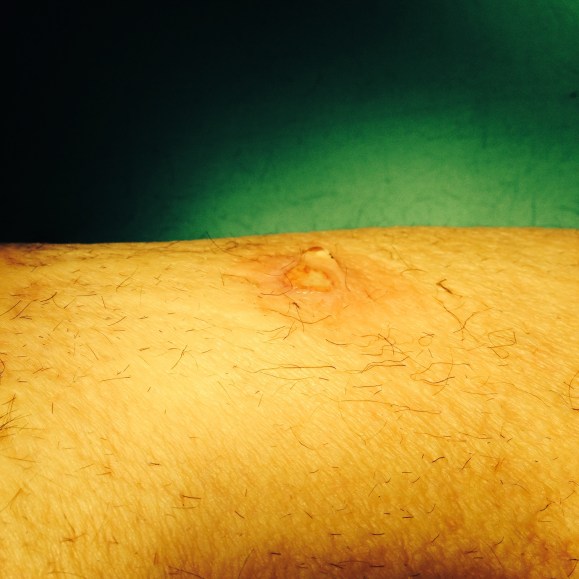
Patient remained hemodynamically stable and recovered in the ICU for 48 hours. After hospital course of approximately 5 days was finally discharged to rehabilitation center. Patient was seen 1 month and 2 months after the first operation and has had completely healed incision (below). There is a strong femoral pulse on the right groin with multiphasic pedal signals. Patient has completed 8 weeks of IV antibiotics. I am planning to perform a CT angio with runoff in the next few months in order to evaluate my distal anastomosis because I am concerned about the Pseudomonas that grew from the OR cultures.

From technical standpoint there is 2 things that I would like to address:
#1: The textbook answer would be to perform a bypass with a native conduit with deep femoral vein or great saphenous vein, but this is difficult to perform efficiently solo. I do not have CryoVein on the shelf and the cost of such a conduit is prohibitive in my institution. And extra-anatomic bypass also on the lateral approach, an obturator bypass, adds time and complexity especially with the separation of profunda and superficialis.
#2 Next time I will have to perform a bypass like that I will probably perform the bypass first to the profunda artery and then reimplant the SFA which is more mobile.
Discussion by W. Michael Park, MD
I have to congratulate Dr. Virvilis on this nice outcome. I agree on remaining vigilant for late re-emergence of infection, but it is very unlikely to occur as time passes without signs and symptoms. I have three things to add. First is that Rifampin soaked graft is effective in revascularizing within a decontaminated field while sewing to uninfected artery. Taking the graft to the common iliac bifurcation which was exposed to gain proximal control was wise as anastomoses to the external iliac under the inguinal ligament are challenging and there was an unknown degree of infection here. Second is the sartorius muscle flap must be in the armamentarium of every vascular surgeon and is an easy jumpoff point to learning gracilis and rectus femoris flaps as well. The lateral mobilization and medial rotation is done preserving the medial arteries that feed the graft -dividing too many of these to mobilize the flap kills the flap. The flap delivers the immune cells and vascularity to clear the remnants of infection and forms a better granulation source bed than debrided, infected wound.
Finally, Dr. Virvilis did reach out to me from around the planet (I work in Abu Dhabi, he in Mississippi) for a run through of his plan. I cannot tell you how valuable this is to be able to bounce ideas off of someone you trust. When I graduated from my fellowship, Tom Bower put his hand on my shoulder and said, “Do not hesitate to call me if you have a difficult situation.” I have since grown a long short-list of mentors, friends, and partners on speed dial. It taps me into cumulative millennia of surgeon-years of experience. With social media and increasing acceptance of its use to share ideas, there is absolutely no reason to work in isolation.