If you work long enough, you will not only see everything, but you may end up doing something that you say you would never do. You will be confronted with a scenario that would test not just your skills but also your boundaries. The adage, never say never is a warning that all of us may face a choice -to remain rigidly consistent with some earlier proclamation or to excuse a little hypocrisy for the sake of the patient.
If you work long enough, you will not only see everything, but you may end up doing something that you say you would never do. You will be confronted with a scenario that would test not just your skills but also your boundaries. The adage, never say never is a warning that all of us may face a choice -to remain rigidly consistent with some earlier proclamation or to excuse a little hypocrisy for the sake of the patient.
At one point in recent years, I saw a patient who had an axillary artery to anterior tibial artery bypass with PTFE (figure above). That was a kind of marvel to me, but my initial response was a bit of a sneer.
“Who does this?” I thought.
Giving it some thought, the rationale could have been to spare the patient from a hip disarticulation as the patient had had failed revascularizations and was occluded from the infrarenal aorta to the anterior tibial artery on that side. If you see such a thing, it sparks wonder as it feels both wrong and splendid at the same time because some surgeon had the audacity to pull it off. By the time I saw it, the patient had avulsed the proximal anastomosis, infarcted their leg to their thigh, and was headed for a hip disarticulation, four years after the creation of the bypass. Four years of patency!
It was no wonder I remembered this case when this middle aged man presented to our clinic with a gangrenous right third toe. He had diabetes, hypertension, CAD with prior PTCA, prior acute mesenteric ischemia with bowel resection with an SMA stent, CHF with moderately reduced EF, CKD, and aortoiliac occlusive disease treated in past with aorto-bi-iliac bypass, left to right fem-fem bypass complicated by graft infection requiring resection of the fem-fem bypass, with subsequent development of rest pain on left leg and gangrene on right leg. He had been told at his home institution that he required eventual bilateral hip disarticulations. At the time of consultation, he was minimally ambulatory, limited by severe pain. He had been this way for over a year.
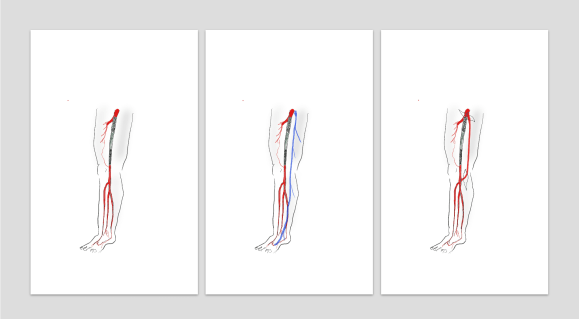
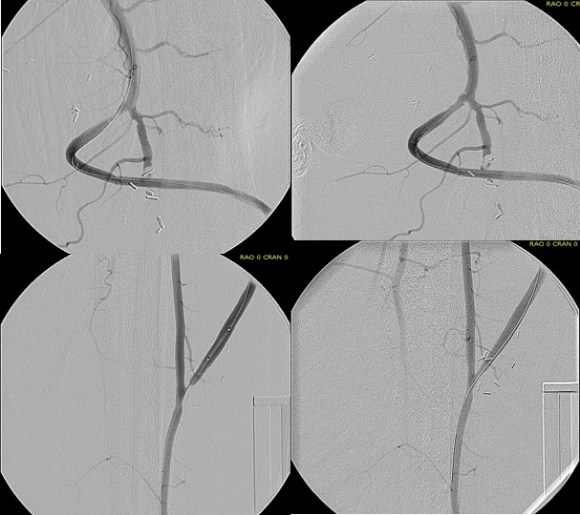
On examination, he had heavy scarring in both groins from prior open incisions healed by secondary intention, a midline laparotomy incision. He had weak bilateral axillary and brachial artery pulses. He had no pulses in either leg. The right foot had gangrene of the distal phalanx of the third toe. The both feet were anemic and painful -the left foot had more dependent rubor. Pulse volume recordings were flat in both legs. TCPO2 was in the 20-40mmHg range at the thighs bilaterally suggesting reduced potential for healing an above knee amputation. Vein mapping showed no suitable saphenous vein in either leg. CTA (figure below) showed both external iliac arteries to be occluded or absent and the common femoral arteries to be occluded or missing bilaterally.

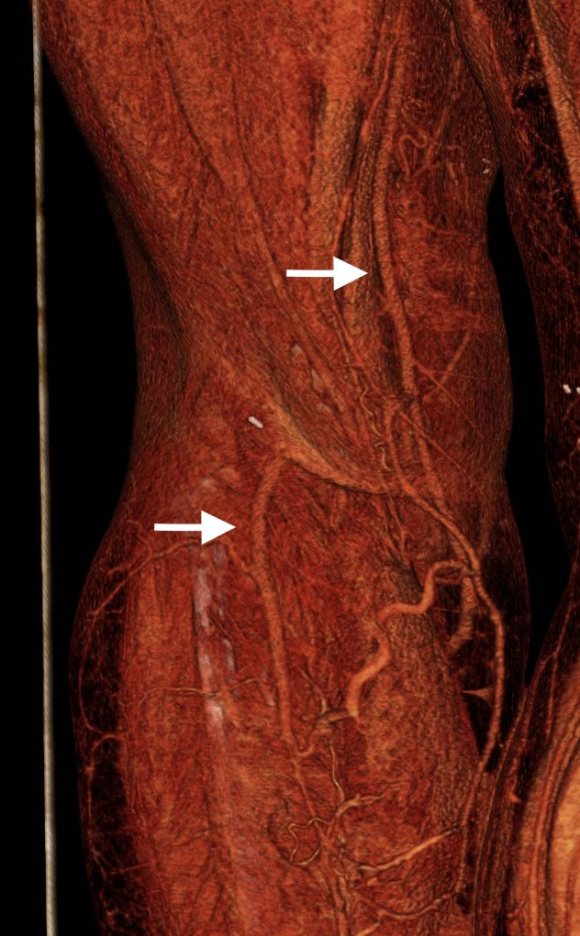
The left femoral bifurcation was preserved and the left SFA was patent into small underfilled tibial vessels. On the right, there was an isolated segment of profunda femoral artery that reconstituted from pelvic collaterals. The right below knee popliteal artery reconstituted and had underfilled but patent three vessel runoff (figure below).

The patient was admitted for workup and treatment. Heparin drip was started. In the setting of rest pain, I find that heparin drip improves circulation and symptoms even though it shouldn’t. I don’t have a great explanation for this, but it does, and I would welcome comments. To better assess how much operation the patient could tolerate, a cardiac risk assessment was performed. He was deemed a moderate to severely elevated risk due to his EF of 35% but had a normal nuclear stress test.
The options I presented to the patient were
1. Hip disarticulations
2. Bilateral above knee amputations with a wait and see approach to hip disrticulation
3. Sympathectomy
4. Axillo-profunda or popliteal bypass on right and Axillo femoral bypass on left
5. Ilio-right popliteal and left femoral bypass.
6. Thoracic or supraceliac aorta to right profunda and left femoral bypass
Hip disarticulation is the bogeyman of leg amputations done for peripheral vascular disease. When done for trauma or cancer in young people, the ability to rehab and walk again is excellent. When done for tissue loss in elderly, non-ambulatory patients, the reported mortality of the operation rises to above 50%. It is usually posed as a lead in to comfort measures. The above knee amputations were not likely to heal despite the neither here nor there findings of the TCPO2 which is only good when the results or normal or dismally low. Sympathectomy is an option for those without options, but this patient still had options, I felt.
Any revascularization relies on the choice of inflow, outlow, and conduit. In endovascular revascularizations, the conduit is the previously occluded vessels, but in this instance, because of the infected grafts, there was neither continuity, nor a good option even if there was as the common femoral artery is a terrible recipient of endovascular therapy. The options then devolve to choosing an inflow. The axillary arteries are technically easiest to access and manage and form the basis of treatment of high risk patients requiring limb salvage who have no endovascular options. The axillofemoral bypass is given a bad reputation of having a poor patency, but the key is the quality of the vessels and the number of potential tension, compression, and kink points. I think the reason why the axillary to anterior tibial bypass lasted for four years in the first patient had to do with his immobility, and the pristine nature of the anterior tibial artery -the only patent vessel below his umbilicus. Here to, the inflow disease appears to have spared his right popliteal artery and his left superficial femoral artery.
The only compromise with an axillary artery inflow is the amount of potential flow. In a patient with a 6-7mm axillary artery, the amount of flow going to both an arm and a leg, and a lower torso, would greatly exceed the flow capacity of that vessel. The infrarenal aorta on this man is graft and is relatively inaccessible due to the prior laparotomy for acute mesenteric ischemia, signalling the high likelihood of adhesions. The supraceliac aorta is an excellent inflow source and I have had good results dissecting it out laparoscopically as it is often deep and narrow an exposure to try to dissect open -While the retroperitoneal tunneling can be tricky, it is not insurmountable and good bypasses can result (link).
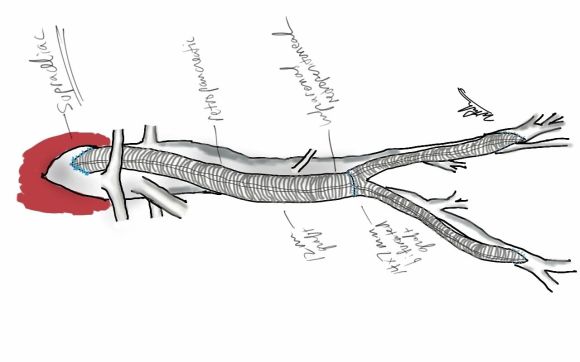
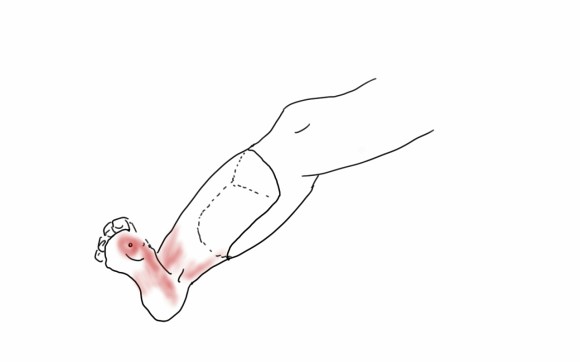
My eye focused on the left iliac graft which perfused the internal iliac artery on that side. The graft was generous, and likely a dilated 8mm graft, and could be exposed via a left lower quadrant retroperitoneal exposure (the transplant exposure). This would allow me to to then tunnel to avoid the terribly scarred groins. On the right side, the obdurator canal could be traversed into the postioer compartment of the thigh -a graft could be sent to the below knee popliteal artery with a side graft to the tiny profunda femoral artery. On the left, the graft could be tunneled laterally near the insertion of the sartorius muscle and onto the superficial femoral artery. All of the incisions would be made in virgin skin, the only redo dissection being digging out the left iliac graft while avoiding injury to the ureter.
So I proposed a ilio-popliteal bypass. Not quite an axillo-tibial bypass, but almost there. There was some karmic balance being restored by my taking decision. It would be with PTFE all around. I quoted a 5-15% risk of major morbidity and mortality, lifelong anticoagulation, and right third toe amputation. The patient agreed.
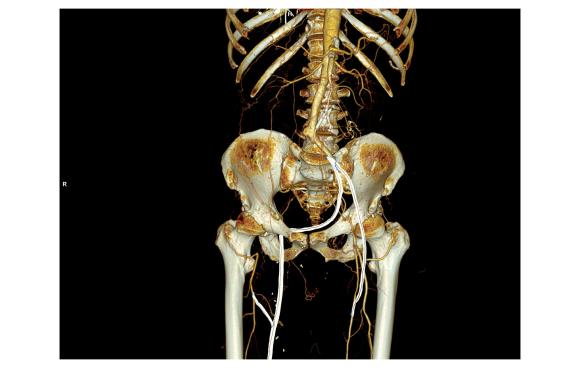
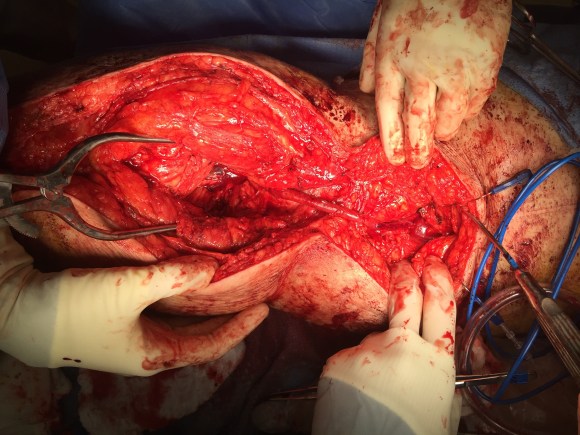
The operation was done in a hybrid suite, as should all limb salvage cases. The retroperitoneal dissection was challenging because of the heavy scar tissue around the well incorporated iliac bypass, but with patience, a clampable 3cm segment was achieved. I am a big fan of Wylie hypogastric clamps because they stay out of the way when placed in a tight narrow spot, and for that same reason, I prefer the Cherry supraceliac aortic clamp. They were designed by my mentor, Ken Cherry, and his mentor, Jack Wylie for this kind of operation. A few venous bleeders were easily handled with my ring compressors (below), and I hope to continue this chain of innovation, but I digress.
The bladder was dissected off the pubis to allow the graft to be tunneled to the right pelvis. A counter incision in the right lower quadrant abdomen and a mid thigh incision mobilizins the anterior compartment muscles to the posterior compartment allowed me to tunnel across the obturator foramen. The obdurator vessels need to be avoided or there will be bleeding. this mid thigh incision allowed exposure of the profunda femoral artery more proximally and allowe the graft to be tunneled anatomically to the below knee popliteal artery for anastomosis. A jump graft was taken off this graft on the thigh to the profunda femoral artery which was small and diseased -no more than 2mm in size. An axillary bypass to this profunda would be doomed to the compromised patency rate published for ax-fem bypasses giving them a bad name. The left superficial femoral artery was exposed and provided outflow to the left iliofemoral bypass which was tunneled far laterally under the inguinal ligament to avoid the scar tissue where the common femoral artery was.
There was immediately multiphasic signal in both feet on release of clamps. After closure of all the incisions and dressings, the right middle toe was amputated.
The patient recently came back for an 8 month followup. His grafts remained patent and he was walking without limitations. Given the high quality of the inflow (large iliac graft), and the amount of decent outflow -the right popliteal and profunda, the left superficial femoral and retrograde to the profunda, gives the patients some surety of longevity for his grafts. These grafts will need lifelong surveillance.
I have since opened my mind a bit about that axillary artery to anterior tibial artery bypass. When given the choice between comfort measures for an otherwise nonambulatory but alert patient and hip disarticulation with its attendant high risk, a bypass from a large axillary artery to a large, relatively disease free anterior tibial artery is not the worst thing that could happen. For that patient, it gave them 4 more years with their family, which in any measure, is priceless. To that patient’s family, that surgeon was a savior.







 This closes the fascia and allows the skin to be closed without tension using only occasional interrupted dermal sutures and a running 3-0 or 4-0 absorbable monofilament.
This closes the fascia and allows the skin to be closed without tension using only occasional interrupted dermal sutures and a running 3-0 or 4-0 absorbable monofilament.