During our daily morning huddles, peopled by cardiologists and cardiac surgeons, one thing impresses me more than anything else. The assembled interventional cardiologists, world class and renown, they who can place a stent in any part of the body, will defer to the unassailable superiority of the LIMA to LAD bypass over any existing intervention. I am always a little sad that the analog for this, the vein bypass in the leg does not get the same love. The open surgical bypass of the leg is the great straw man at international symposia. It is fast becoming a diminishing and curious habit of a fading generation.
The acknowledged superior hemodynamics and patency of the bypass is diminished in the literature by pooling patency loss with other factors such as amputation, heart attacks, and death. Some vascular surgeons dogmatically cling to habits learned in training that favor complications, making themselves their own worst enemies both in the literature and in the marketplace. These bad habits involve long incision length, closure techniques that do not anticipate edema, and wound orientation that makes failure more likely.
Operations require far more support and resources to succeed than do interventions that soon go home. Brilliant operations alone will not heal the patient. It is pathways and postoperative care infrastructure and staff that prevent these secondary complications -the very complications that keep the leg vein bypass from being as respected, if not loved, as the LIMA to LAD.
The postoperative care of these patients devolves to management of leg edema. No medical or nursing school adequately teaches the basic science nor management of edema, which is the most common vascular condition
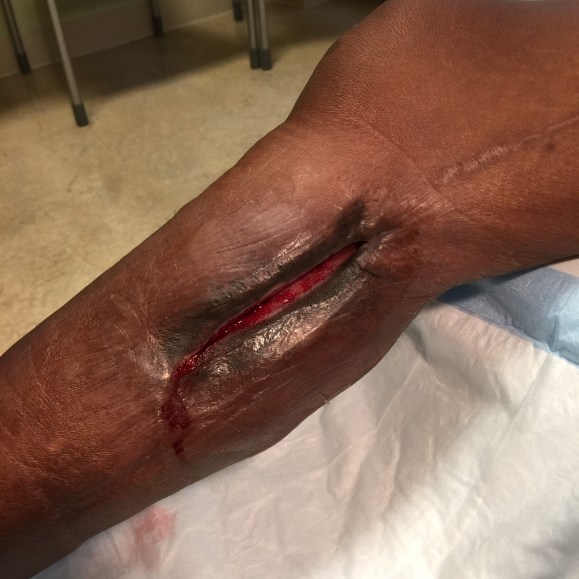
The incisions are too long in the classic vein bypass. When you create and then close an incision, the inflammation drives the accumulation of fluid in the extracellular space – creating edema. This postoperative edema, poorly managed, results in complications that leave the patient hobbled with time lost to healing wound complications, pain, and excess limb weight. Additionally, vein bypasses usually involve groin exposure and the delicate lymphatics that coalesce there are perturbed or destroyed during exposure. Postop, this damage and the inflammation rapidly overcomes the capacity of a lymphatic system. The traditional vein harvest also involves cutting through deep layers of fat. The fat is typically closed by broad sutures that create areas of fat necrosis -potential fodder for bacteria. The best ways the complications of long and deep incisions is to avoid them altogether. The calculus of the operative moment – “I must see the vein,” must include the vision of a patient losing months to wound therapies to heal a gaping, necrotic, infected wound. I recommend skip incisions or adopting in-situ bypass technique with endovascular management of fistulae. Or corral your cardiac PA to harvest the vein segment in the thigh after mobilizing the vein in the leg with the endoscope.
The incisions are often closed with Nylon sutures and skin clips which can become potential foci of infection. With edema, they create zones of ischemia around them, killing skin and creating entry points for skin flora as the skin expands under an unyielding clip or suture. Placed under a pannus, these sutures or clips fester in an anaerobic environment. Closure should adhere to anatomy. The body relies on connective tissue planes to keep itself together. In the groin, these are Scarpa’s layer and the dermis. They should be closed with absorbable monofilament in a buried interrupted fashion at the dermis with a final running subcuticular layer of 4-0 absorbable monofilament. Steristrips or glue at the skin finishes the job. If you use sutures, particularly at the distal anastomotic site, take care to realize that you have about 12 hours before the skin dies in the best of circumstances, and less with microangiopathy of diabetes and ESRD. Squeezing out the edema before closure with a sterile Esmarch or short counterincisions or even a large one to allow for tension free closure over an anastomosis will prevent wound complications over your graft.
The classic longitudinal groin incisions that cut across the inguinal crease divides a tension point -that crease is like a cord that supports the pannus that is slung over it and when divided and then closed with a stitch, that stitch then bears the weight of that pannus every time the patient sits up or stands. If you are observant, wound necrosis typically starts at the groin crease under a surgical clip or suture. Incisions in the groin should be obqlique and parallel to this crease, or if you can, even inside this crease. When these wounds are closed, the natural lines of tension are in line with the incision rather than orthogonal to it. The natural forces keep the wound shut.
This is only the first step. The next is keeping the wound clean and dry for at least 5 days. At the Mayo Clinic, where I trained, the nurses up on 5 Mary Brigh were trained to blow dry the groin wounds every few hours on cool setting and redressing the wound with dry gauze. You can get something close to it by ensuring the wounds painted with betadine, allowed to air dry, and dressed with dry gauze. If there is a constant leak of fluid, you have a serious problem as there is too much edema in the leg, or the wound isn’t closed, or there is a lymph leak. It needs to be actively dried out or you get a wet, macerated, infected wound like a grenade went off in the groin.
They don’t teach compression wrap techniques in medical or nursing school
The simplest thing to avoid lymph leaks is to not make them. Cutting near lymph nodes is hazardous, and once below Scarpa’s you have to orient your dissection directly over the femoral artery. Stray horizontally and you will undoubtedly cut one of the 4 to 10 invisible lymph channels.
They are invisible but detectable -after you break them, you will see a constant wetness in the wound. Think about injecting a cc or two of Lymphazurin (Isosulfan Blue, for those not allergic to Sulfa) into the intertriginous space on the same foot and you will see the lymph channel in bright blue, or stare carefully at the likely spots for a lymph leak and clip it, burn it, Ligasure or Harmonic scalpel it.


So how did we get to a rather dry discussion about edema? Wound complications are tremendously debilitating and offset any benefit from vein bypass operations. These long incisions become terrible big wounds if not prevented. It takes the concerted effort of a team and particularly nursing in actively managing edema. And at the end, the patient too must be included in this discussion. For the vein bypass of the leg to get the same respect and love as the LIMA to LAD bypass, surgical wound complications must become never events.