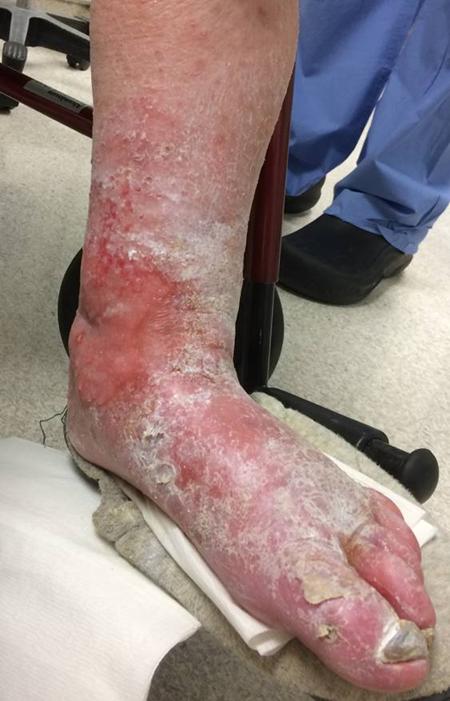
The patient is a very pleasant elderly lady who had a prior EVAR complicated by graft limb thrombosis treated with thrombectomy. She recovered from that but subsequently developed ulceration of her left ankle. She had been sleeping in a chair because it hurt her to sleep flat –her leg and foot would burn with pain. A wound care center had tried an Unna’s boot, but it caused her worse pain, and the ulcer increased in size. At admission, she had an exquisitely tender, edematous leg and ankle with a large ulcer weeping edema fluid. There were no palpable pedal level pulses.
I admitted her for workup and treatment of a mixed etiology arterial and venous ulcer.
These are patients for whom rest pain is relieved by avoiding recumbency, but with prolonged sitting, as in this lady, edema accumulates and starts to leak, creating an ulcer of the venous type, in the medial ankle (gaiter) region. These don’t resolve without addressing the underlying cause which is the arterial insufficiency. Fixing the arterial insufficiency then allows for leg elevation and compression. For the trainees, venous ulcers almost uniformly heal with Unna’s boot therapy. Elevation should relieve discomfort in venous ulcers. Neither of these occurred and raises the suspicion of arterial insufficiency.
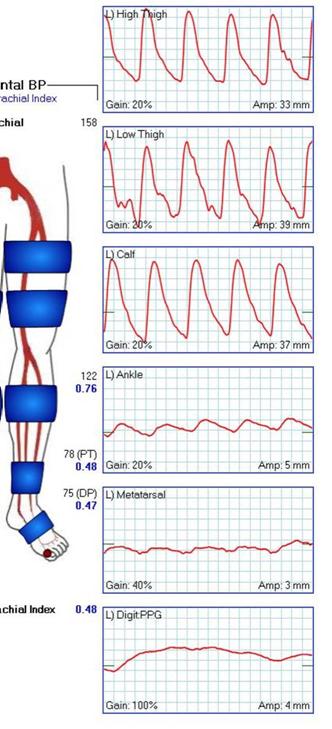
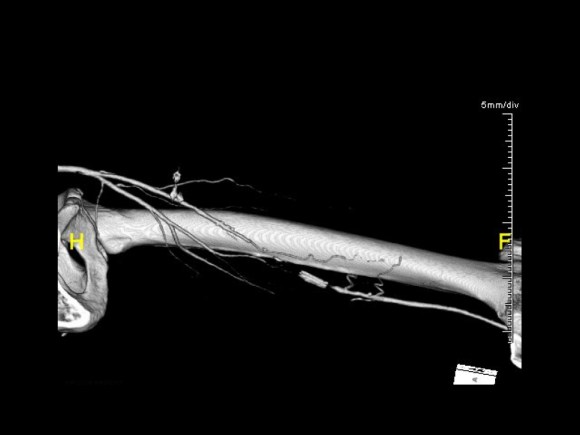
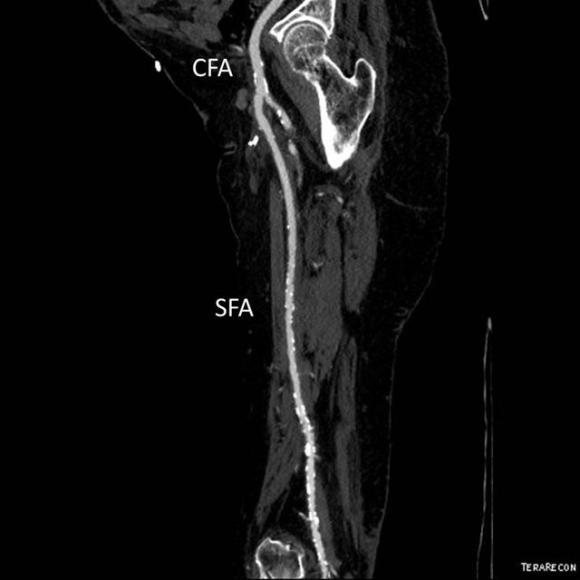
At admission, her PVR’s showed severe popliteal/tibial level occlusive disease. CTA was performed and it showed the common femoral and superficial femoral arteries to be patent but plaque occluded the popliteal artery and origins of the tibial vessels.

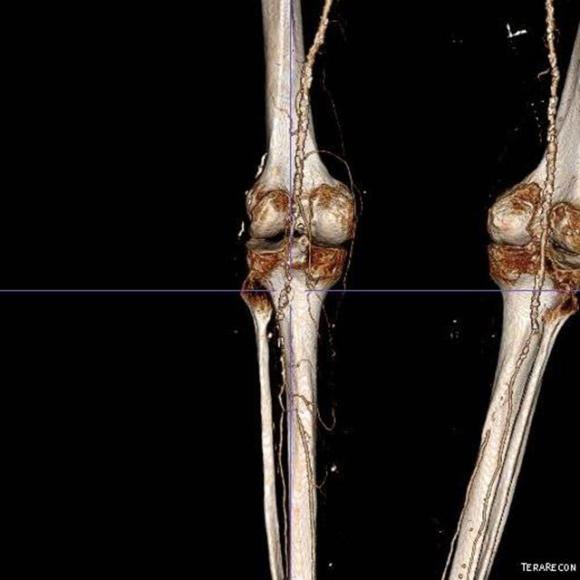
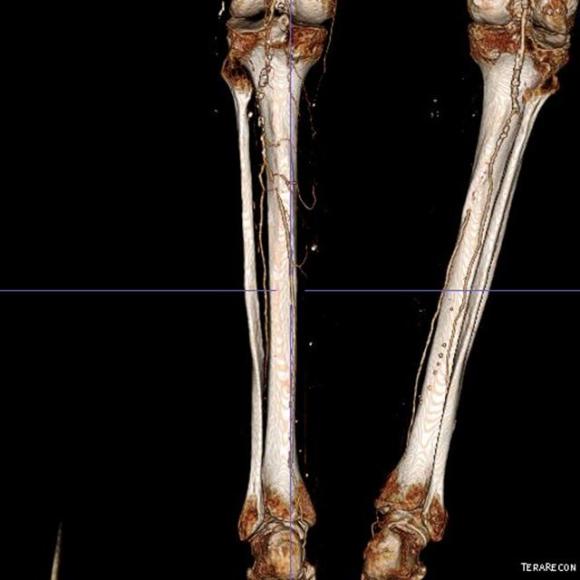
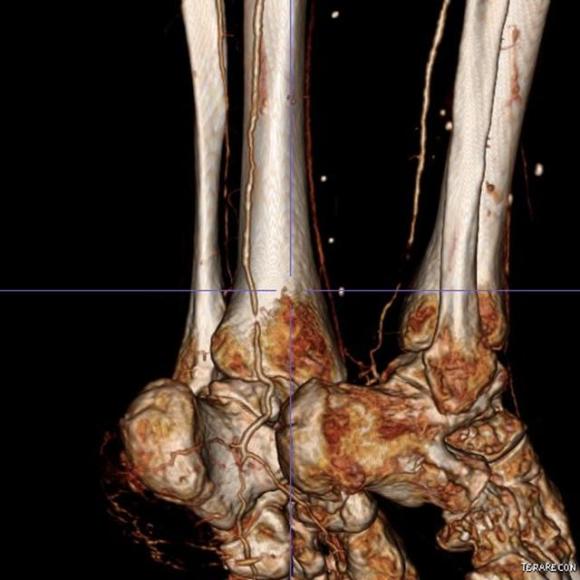
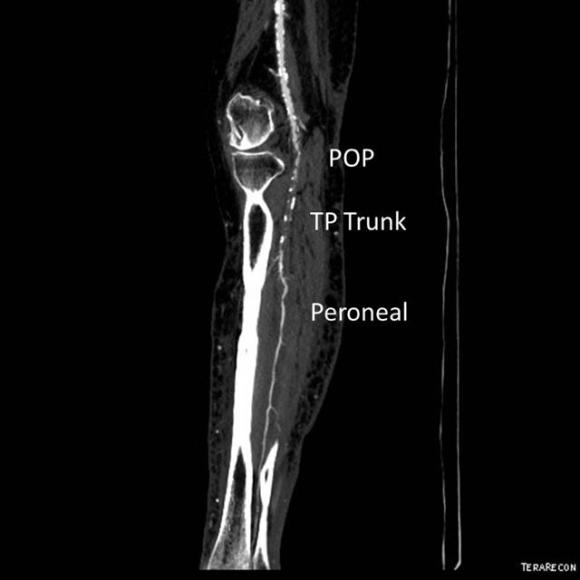
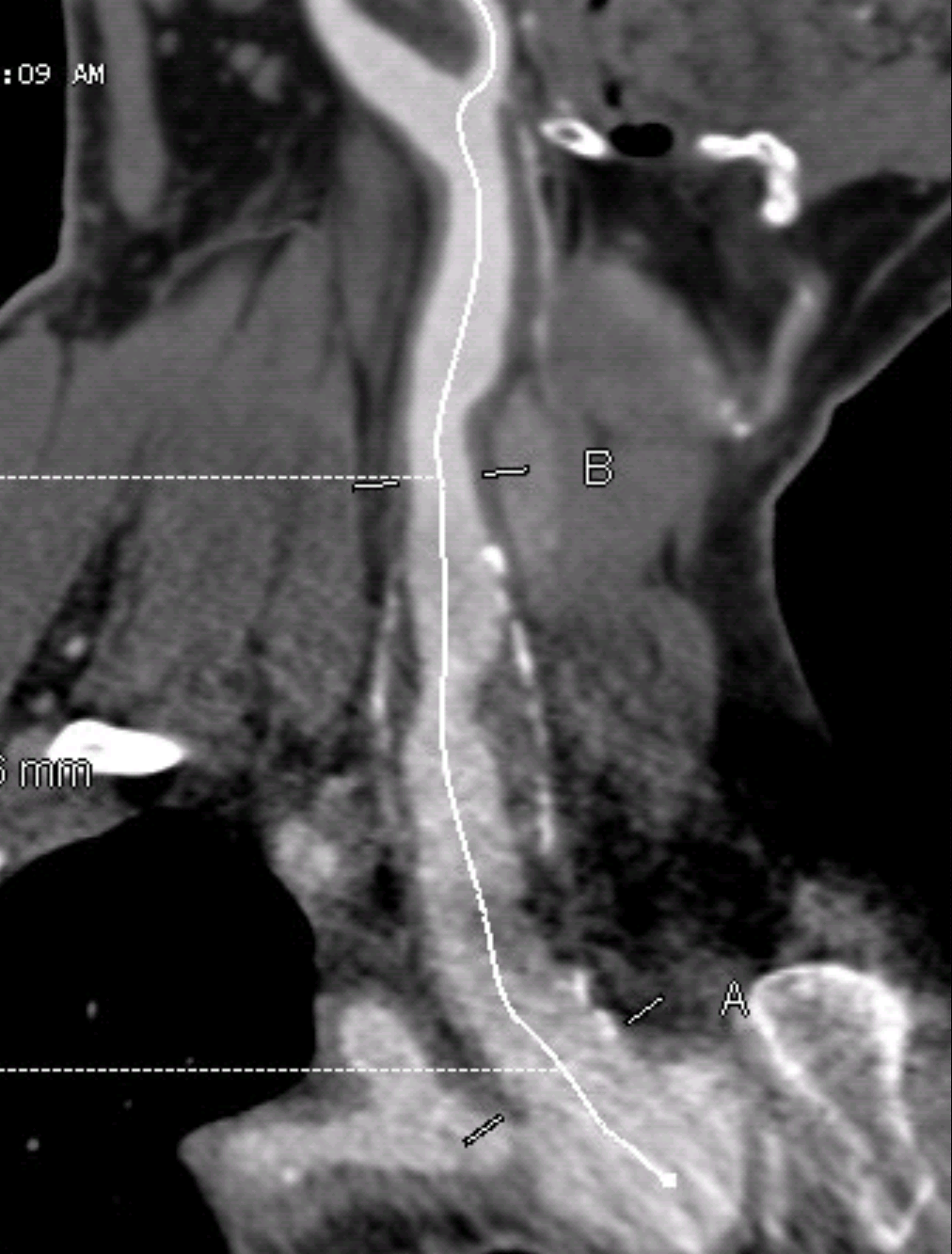
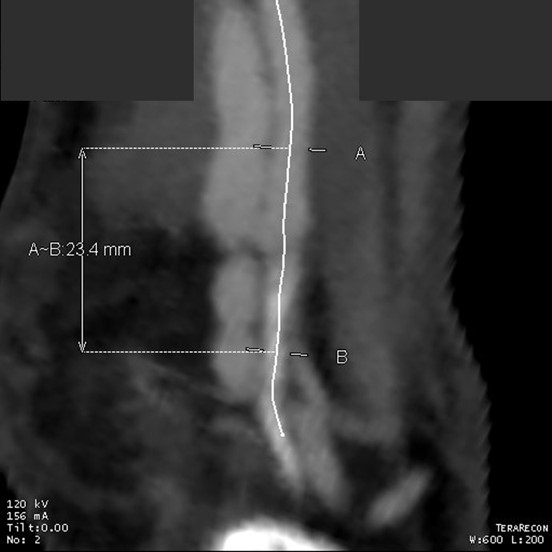
The only patent runoff was via her peroneal artery. Centerline evaluation of the CT scan was performed, with manual centerline created through the occluded segment of popliteal artery. I find this useful for planning endarterectomy and bypasses, and with attention to detail, images that are the equivalent to tibial angiograms come to life. This is a centerline through the femoropopliteal to peroneal system.


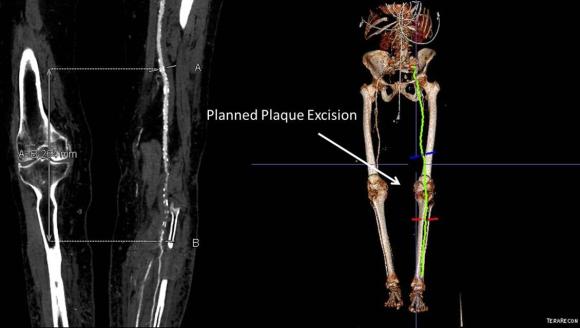
Vein mapping revealed a paucity of good vein –only a short segment in the proximal thigh on the left and for a short segment on the right. Stress testing revealed that she was good to moderate risk. Isolated popliteal occlusive disease with poor tibial runoff, while feasible for intervention, is not likely to be durable. Multisegment vein bypass on the other hand, using at least three segments, meant a long operation for this frail old lady and a prolonged recovery. I felt that popliteal endarterectomy and distal SFA remote endarterectomy offered a good option for revascularization, with either a patch repair or a short bypass to the peroneal artery. The backup plan was composite vein, but it was unlikely to be needed because the plaque was not the calcium pipe type plaque that does not endarterectomize well.
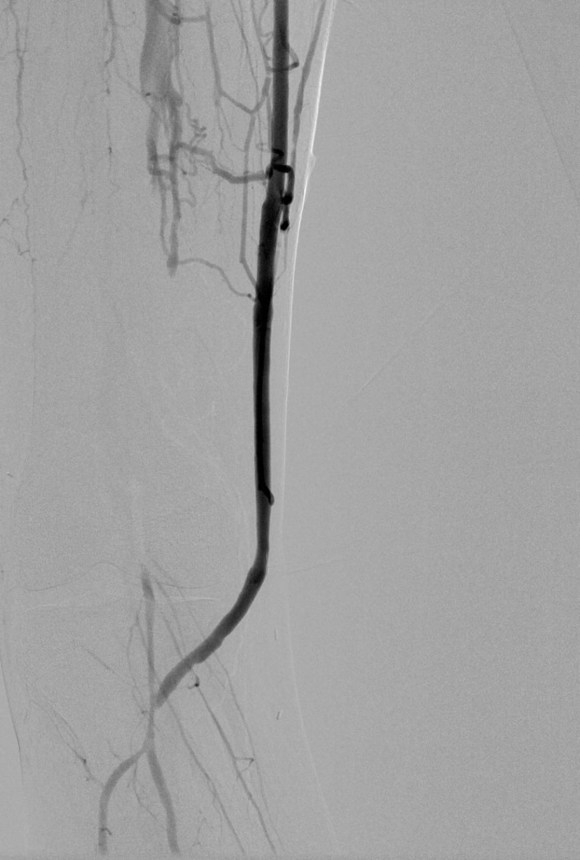
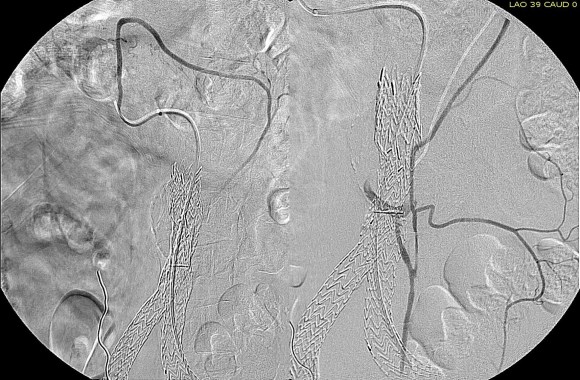
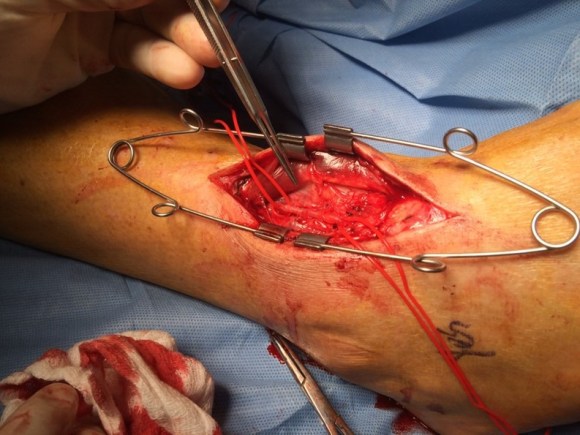
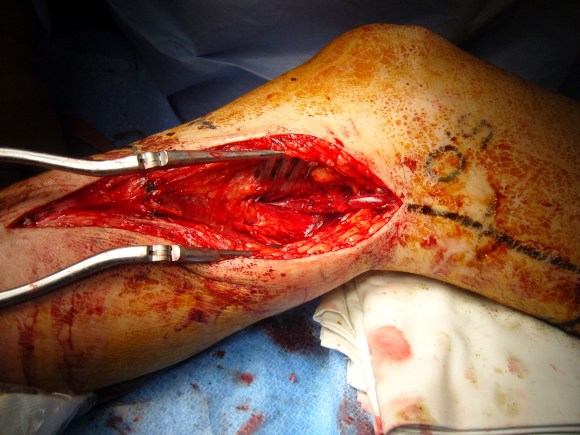
The patient was positioned on the table supine. The short segment of proximal greater saphenous vein was harvested –it was of suitable caliber, but below its first major tributary point, the veins was thick walled and small. The total length was about 10 cm. The below knee popliteal space was opened and the popliteal through tibioperoneal trunk bifurcation was exposed. Antegrade puncture of the common femoral artery allowed for arteriography and it showed the occlusion at the knee with reconstitution of the peroneal artery.

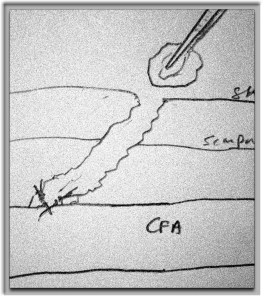
The popliteal artery was opened and endarterectomy of the occlusive plaque was performed. Retrograde remote endarterectomy (EndoRE) with Vollmer rings was performed to the mid superficial femoral artery where on the CTA the calcified plaque ended. The technical point about retrograde EndoRE is that the ring catches as the plaque gets larger more proximally, and has to be swapped out for a larger ring. Ultimately a 7mm Moll Ring Cutter was used to cut the plaque (picture below, arrow to more proximal SFA plaque) .
.
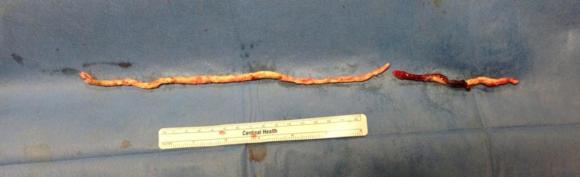
The plaque, because it is larger the more proximal you go, came out with some difficulty via the below knee popliteal artery. This is not a great concern if it won’t come out –you merely have to cut down on the SFA in the thigh to fish out the plaque. In this case, it was not necessary, and it came out in several pieces, facilitated by the cutter which was used to graft the plaque in segments to retrieve it. Unfortunately, I don’t have a picture from this case of the plaque, but I have inserted a popliteal endarterectomy plaque image below from an prior case of popliteal endarterectomy.
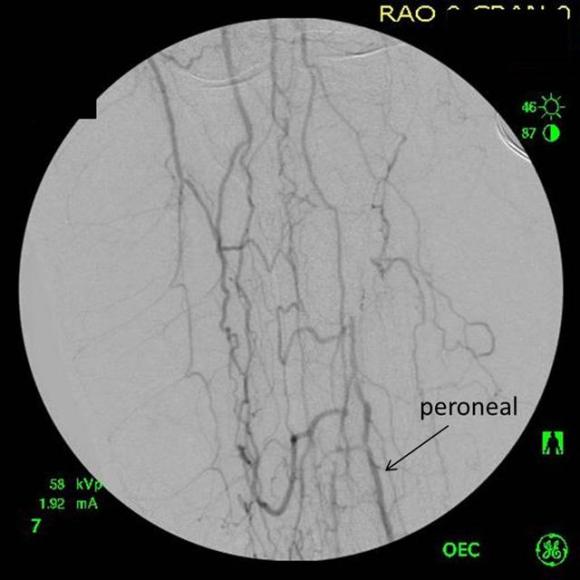
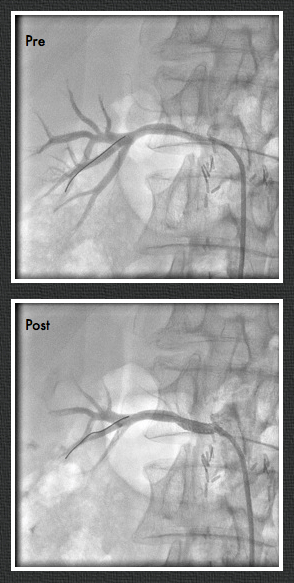
This restored pulsatile flow to the below knee popliteal artery. Opening the artery down to the tibioperoneal artery revealed the artery to occluded and I took the endarterectomy to the peroneal artery origin and everted a short segment of posterior tibial plaque. The peroneal artery was large and would accept flow readily, so I chose to bypass to it using the short segment of saphenous vein that I had harvested for a possible patch or short bypass. The vein was reversed and anastomosed in the usual manner. Arteriograms are below.

The flows were multiphasic. I attempted to cross the posterior tibial occlusion but ended up with contrast extravasation, therefore stopped with this repair. The patient’s wounds were closed and ulcer cleansed and compressed. In the week postop, she healed her ulcer and her two short incisions, and felt good enough to go home with homecare. Her noninvasive studies and duplex confirmed the patency of her revascularization, and there was a multiphasic signal in her posterior tibial artery as well as peroneal.
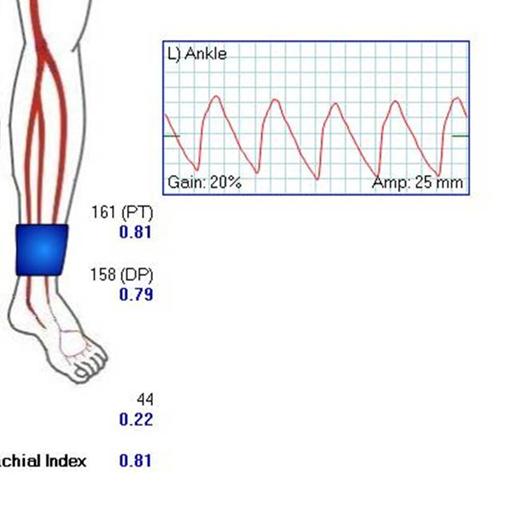
In the handful of patients I have managed this way, either with popliteal endarterectomy and patch or short (micro) bypass, they have stayed patent past a year, but do require surveillance. Because of her frailty and unsteadiness of gait, I chose not to anticoagulate with Coumadin which is my usual practice, but have her on Plavix and aspirin.




















 Completion angiography fits into the range of things that many of us were taught to do because it might help avoid the problem of early graft failure. I remember a time in the nineties when vascular surgery was synonymous with terrifyingly long bypass operations that sometimes worked. Back in that preinternet era, all day bypass operations were capped at the end with a flat plate arteriogram. As with all things archaic and historic, I firmly believe that our trainees should feel comfortable with this type of on-table arteriography because not every place will have a corridor of rooms with robotic c-arms. I feel that each trainee should feel comfortable wheeling in a portable c-arm, assembling it, turning it on, put in patient information, and perform a study. But I digress. The completion arteriogram clearly has a role in bypass surgery, but I question its usage as a “I do it all the time” routine. When anything is written in stone, it immediately takes on a hallowed, sanctified aura, usually taken on during M&M’s when the person at the podium intones beatifically looking skyward, “the completion arteriogram showed no abnormalities.” Science is about questioning the status quo and backing up practice with evidence.
Completion angiography fits into the range of things that many of us were taught to do because it might help avoid the problem of early graft failure. I remember a time in the nineties when vascular surgery was synonymous with terrifyingly long bypass operations that sometimes worked. Back in that preinternet era, all day bypass operations were capped at the end with a flat plate arteriogram. As with all things archaic and historic, I firmly believe that our trainees should feel comfortable with this type of on-table arteriography because not every place will have a corridor of rooms with robotic c-arms. I feel that each trainee should feel comfortable wheeling in a portable c-arm, assembling it, turning it on, put in patient information, and perform a study. But I digress. The completion arteriogram clearly has a role in bypass surgery, but I question its usage as a “I do it all the time” routine. When anything is written in stone, it immediately takes on a hallowed, sanctified aura, usually taken on during M&M’s when the person at the podium intones beatifically looking skyward, “the completion arteriogram showed no abnormalities.” Science is about questioning the status quo and backing up practice with evidence.