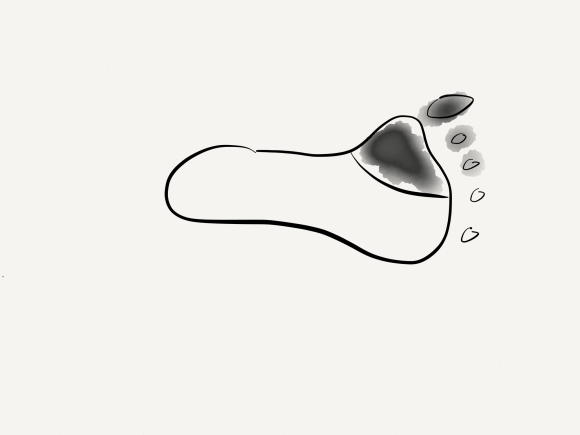
The basics of this air rocket pictured above is the projectile, attached by tube to a large bladder which when compressed by external force, ejects the projectile upwards. These are the same features of a popliteal venous aneurysm. First, the large chamber predisposes to stasis and thrombus formation. This thrombus will form on the flaccid walls which are areas where stasis occurs. During activity, it likely dislodge but catch at the outflow, obstructing it. Pressure builds up in the calf veins below, and flexing the knee and pressing the venous aneurysm ejects the thrombus towards the heart and lungs. Clot Gun.

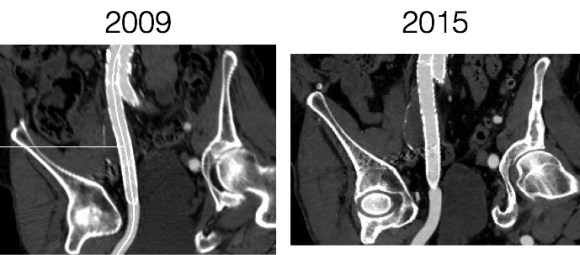
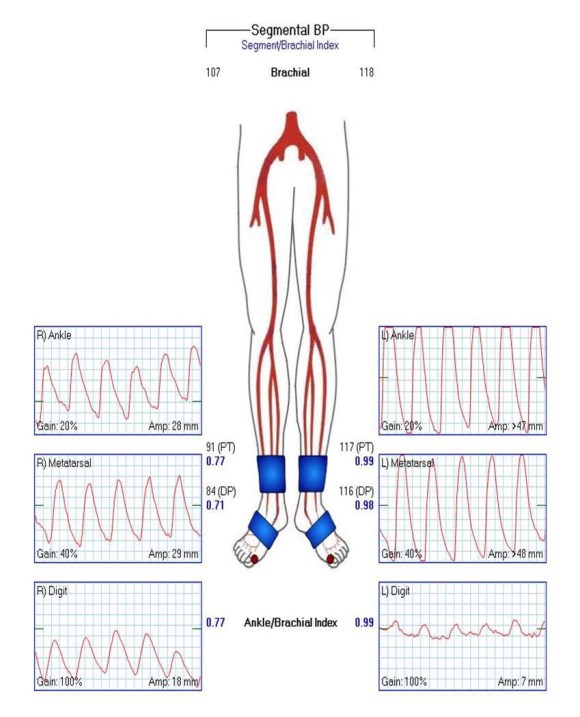
The patient is a young woman who was an active college athlete. She had her first pulmonary embolism occur during practice several years prior to presentation. A duplex noted residual thrombus in her right popliteal vein. Over the next several years, she had two more episodes of pulmonary embolism whenever her anticoagulation was stopped. No thrombophilia was detected on workup. She was referred to the Clinic and Dr. Jerry Bartholomew in the Department of Cardiovascular Medicine noted in her records a mention of a dilated popliteal vein. On examination, she had no historical or physical examination findings to suggest a predisposition to pulmonary embolism. A duplex was ordered.

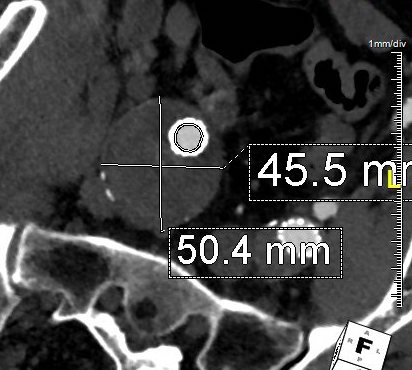
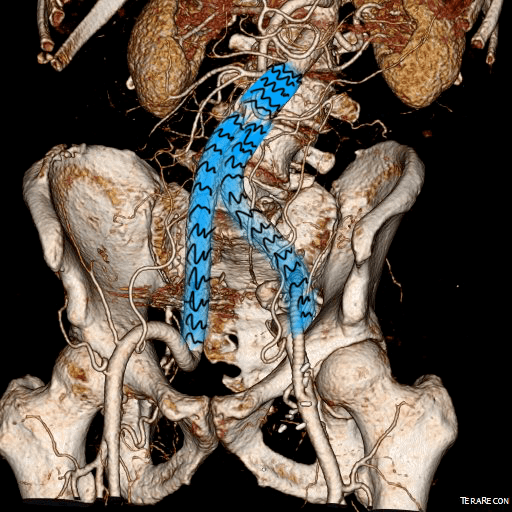
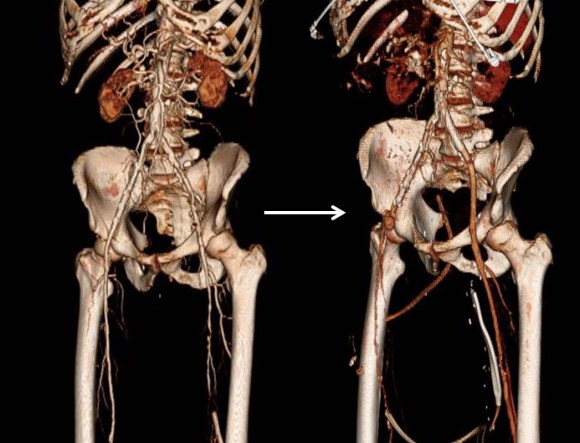
The duplex showed a 2.8cm popliteal venous aneurysm of the right leg. No acute DVT was seen but swirling rouleaux could be seen on the B-mode video. A CT venogram was ordered.

No other defect was detected. Operation was planned. Mapping showed no suitable superficial venous conduit, and venorrhaphy was planned. The patient was kept anticoagulated to the day of operation.
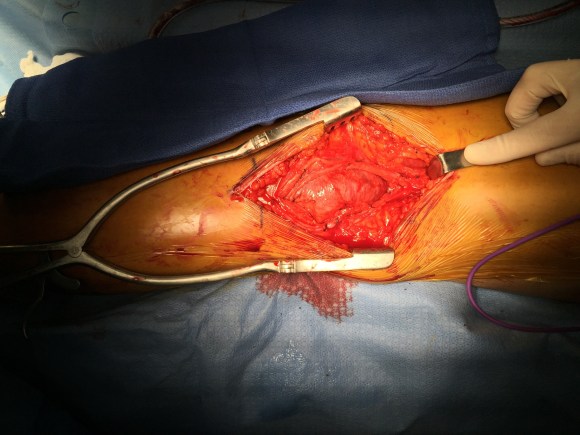
A curvilinear incision (lazy S) was made across the popliteal fossa and careful dissection revealed the aneurysm. It was soft and the vein was normal below in the calf. Above it, there was a tight fibrous band that was contricting it -a popliteal venous entrapment. I released this band. Using a 24 French Foley catheter inserted through a transverse venotomy on the popliteal vein below, the aneurysm was plicated to approximately 1cm diameter, and the catheter removed and the venotomy repaired.

The nerves were restored to their original position and the wound closed in layers. She recovered well and returned to followup about a month later. Duplex showed a patent vein and she had no symptoms of dyspnea.

The plan is to have her come off of her anticoagulation after a visit with Dr.Bartholomew. Reviewing the literature, my confreres at Mayo published their 15 year experience with popliteal venous aneurysms and found that 5 of their 8 patients presented with pulmonary embolism, and that most of their complications occurred with bypass repair while aneurysmorrhaphy fared well (reference). Because of their rarity, about 200 cases in the literature, it may be assumed that many are not found until complications occur or never found because pulmonary embolism, the most common complication, results in death. Also, it would be easy for unknowing physicians to assume that popliteal venous aneurysm falls under the umbrella of varicose vein which this is not. They should be treated when found, and in most cases, such as this, venorrhaphy is preferred.
Reference
Johnstone JK et al. Surgical treatment of popliteal venous aneurysms. Ann Vasc Surg 2015;29:1084-1089.