The patient is a young woman from overseas who was referred to the clinic for abdominal and chest pain that persisted after diagnosis and treatment of Takayasu’s Arteritis affecting her visceral (middle) aorta. A year prior to presentation, she had been having severe abdominal pain, 30 pound weight loss, and weakness. Laboratory findings included elevated inflammatory markers. CT scan showed inflammation around her celiac axis and superior mesenteric artery. She was treated with prednisone but only responded ultimately to immunosuppressives. Her pain remitted for a while and she regained weight and strength, but eventually in the months prior to consultation, symptoms of postprandial abdominal pain and nausea set in with concomitant constant midsternal chest pain which at times was incapacitating.
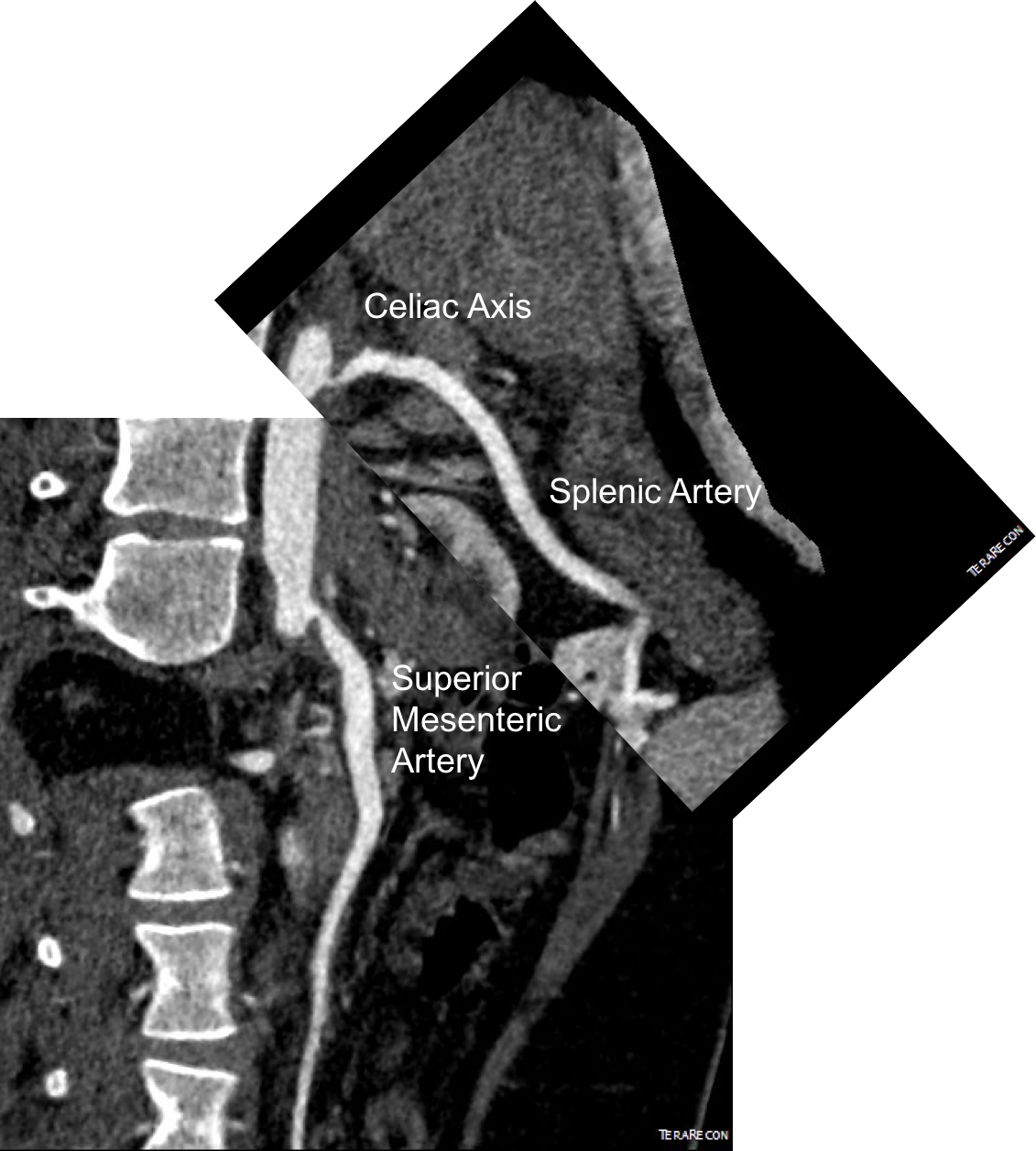
Examination was remarkable for a well nourished young woman in distress with epigastric tenderness. Inflammatory markers were normal and she was on methotrexate and tocilizumab. CTA (above) and duplex showed severe stenoses affecting the origins of the celiac axis and superior mesenteric artery. A composite of the centerlines through these showed the arteries to be critically narrowed at their origins (below), along with a mild to moderate stenosis of the aorta (above). No active inflammation could be seen.
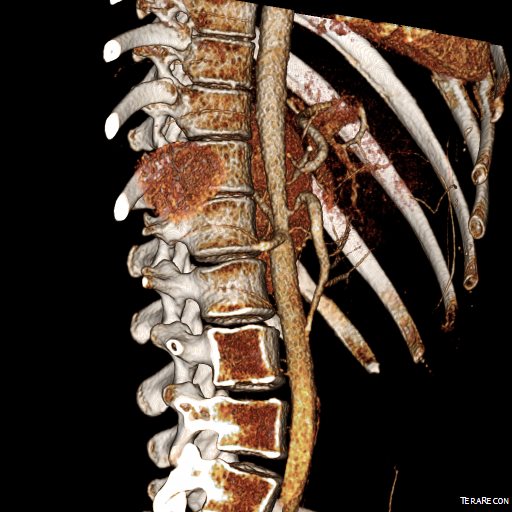
The anterior view of the 3DVR images of her CT showed an absence of collateralization via the inferior mesenteric artery and Arc of Riolan.

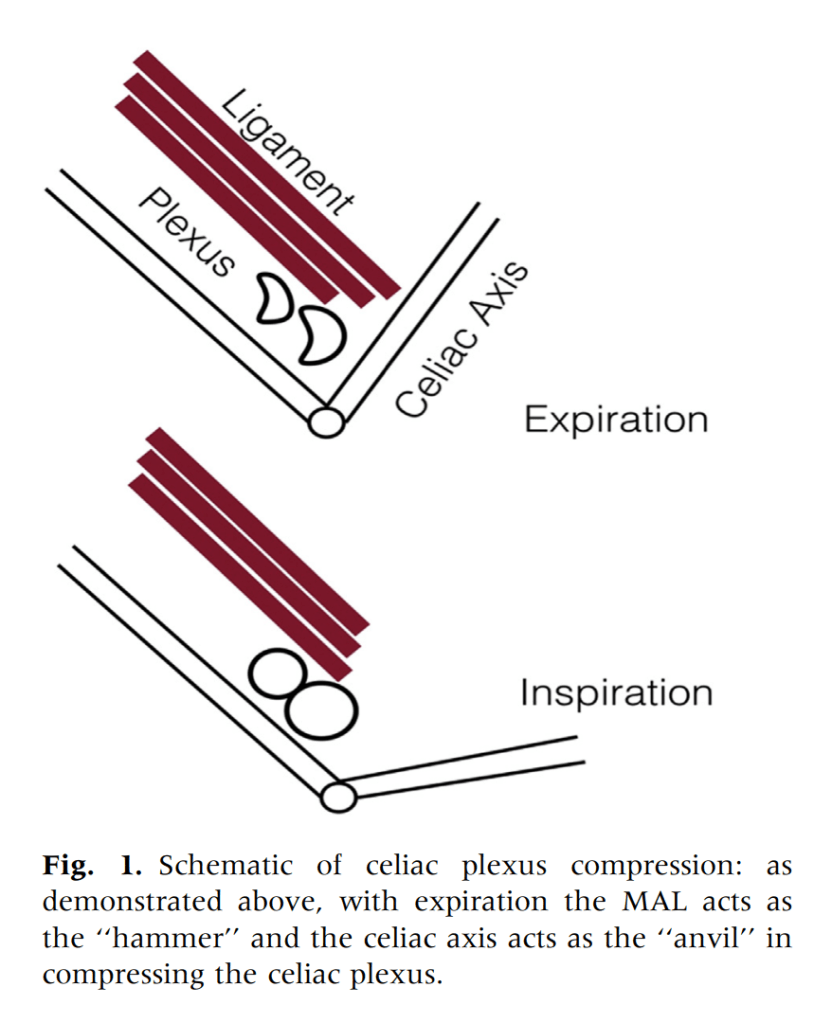
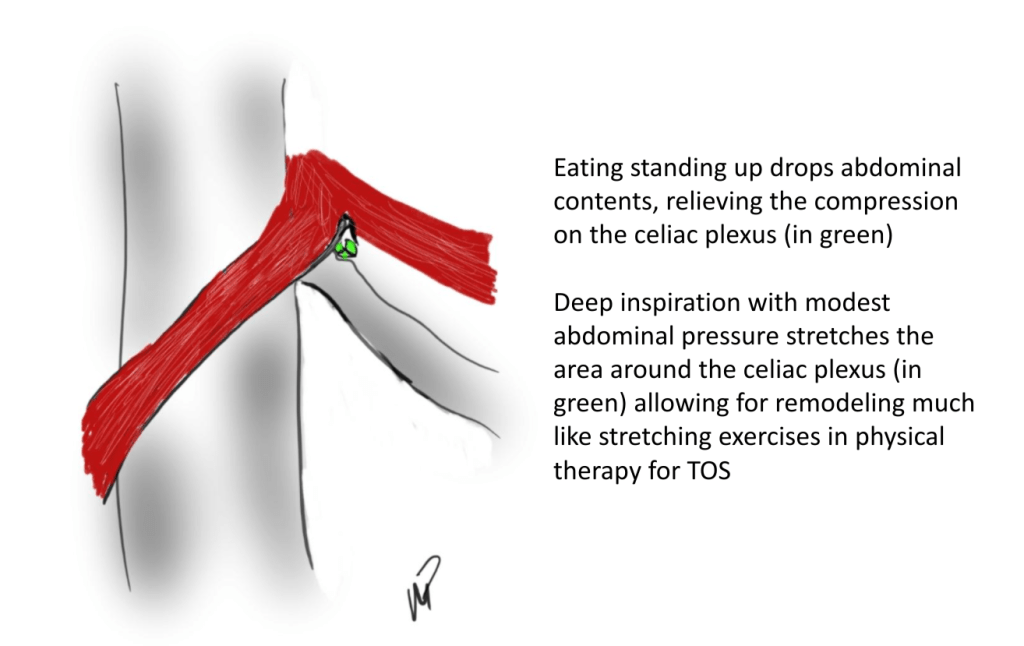
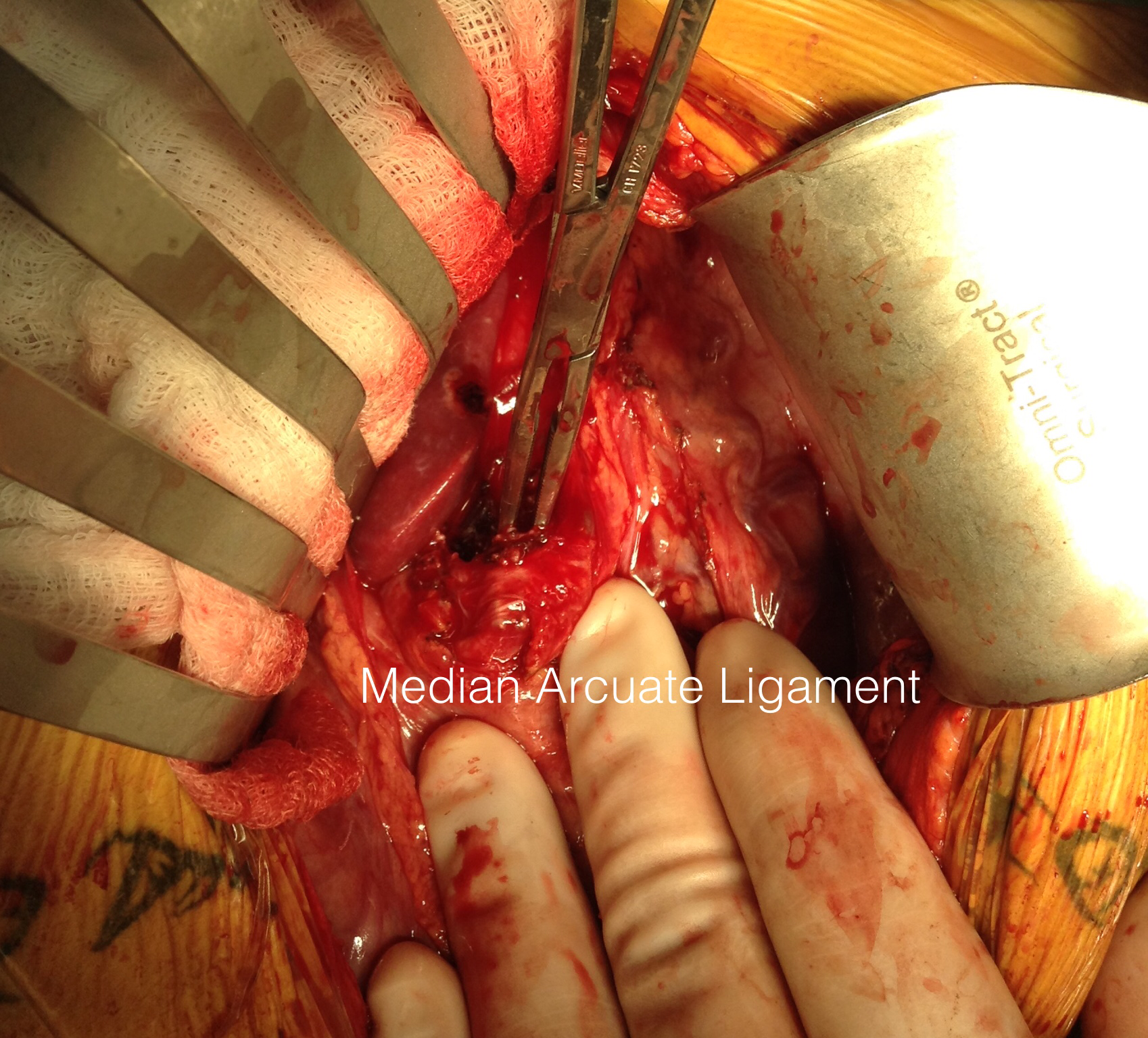
This could account for her postprandial abdominal pain which was midabdominal, but the midsternal chest pain was difficult to explain. A cardiac cause had been ruled out at her home institution. It was on the lower part of the sternum and bordered the epigastrium. Through my work with median arcuate ligament syndrome, it was not unusual to have chest pain be part of the pain syndrome which comes about through the compression of the celiac plexus by the median arcuate ligament (reference), and treated effectively with the division of the median arcuate ligament and celiac plexus neurolysis.
I did feel that revascularizing the SMA was likely to improve her postprandial symptoms, given the paucity of circulation to the gut. I had a discussion about her chest pain and the thought that this was a celiac plexus neuropathy as the result of compression of the celiac plexus by her Takayasu’s disease. Typically, for younger people, I perform a bifurcated graft to the celiac axis and SMA from the distal descending thoracic aorta for younger patients, but I had no intention of replacing her aorta at this time, and wanted to reserve any definitive revascularization of her visceral vessels for a later time if it became necessary. Her pulses were full in the legs and she had no hypertension, renal insufficiency, or claudication. I therefore planned a ilio-mesenteric bypass, as it would preserve planes for a later more definitive operation if necessary, and would address her mesenteric ischemia.
I also proposed taking down the median arcuate ligament and lysing her celiac plexus to treat her chest pain symptoms. While I knew this would be fraught with some hazard because of the inflammation that had been there, it would be the best chance at treating this symptom that was debilitating her. I did not think the constant chest/epigastric pain had a vascular etiology. Normally, I would test this with a celiac plexus block, but given the likely inflammation involved, a failed block would not be helpful, and a positive one achieved with some difficulty.
The patient thought this was reasonable and agreed. She underwent a midline laparotomy and I exposed both the SMA and right common iliac artery in the retroperitoneum and tunneled a PTFE graft in a C-pattern in the retroperitoneum. The SMA was diseased proximally under the pancreas and affected by inflammatory scar tissue, and the vessel was thickened as well. The iliac pulse was normal and full and provided excellent inflow.
Going into the lesser sac, the crurae of the diaphragm were heavily scarred near the celiac axis. Division of these fiber was taken slowly and with the aid of both loupe magnification and a hook cautery (borrowed from laparoscopic surgery). The aorta was cleared of tissue first -it was also encased in scar tissue. The dissection going distally was made difficult by inflammatory scar tissue as well, but several large nerve trunks were encountered and divided. The celiac axis was atretic and small within this scar tissue, but was released down to the trifurcation.
The recovery was notable for remission of her chest pain and her ability to resume eating without pain or nausea. A CTA done before discharge showed a patent bypass.

Followup communication has revealed continued remission of her presenting symptoms months after her operation.
We are working on presenting a followup to our recent paper on MALS. Increasingly, it is apparent that consideration of celiac plexus compression as an etiology of epigastric abdominal pain unexplained by more common gastrointestinal causes provides solutions for patients given no explanation for their debilitating pain. This pain can also affect the chest, and flanks and back in the distal thoracic dermatomes, and mimic the some of the symptoms of mesenteric ischemia. In those with recurrent MALS after a successful ligament release and interval of symptom remission, even in the absence of celiac axis compression, a positive response to a celiac plexus block points to the presence of either remnant plexus fibers or compression by scar tissue of the cut nerve endings (neuromata) and reoperation has been successful. Extrapolating this experience to this patient with inflammatory compression of the celiac plexus secondary to Takayasu’s Arteritis made sense and celiac neurolysis appears effective.
Reference
Weber JM, Boules M, Fong K, Abraham B, Bena J, El-Hayek K, Kroh M, Park WM. Median arcuate ligament syndrome is not a vascular disease. Ann Vasc Surg. 2016 Jan;30:22-7.