My partner, Lee Kirksey, Vice-Chair of Vascular Surgery, just got a paper on-line (link) about the curiously increasing volume of open surgical repairs we were experiencing from 2010-2014 at the Cleveland campus of the Cleveland Clinic. When I joined in 2012, my impressions at that time were mostly the paucity of straightforward EVAR cases that I had seen in private practice, and the high prevalence of stent graft explantation, infected aortic grafts, and open aortic aneurysm repairs (OAR) for juxtarenal and thoracoabdominal aortic aneurysms for nominally high risk patients. My TAAA muscles had atrophied during my years out of fellowship and I eagerly took the opportunity to recruit the help of my partners and scrub in on these cases with Pat O’Hara, Jean Kang, Dan Clair, Ezequiel Parodi, and Lee Kirksey. It is without any shame that I sought out not just extra expert hands, but interrogated these experts for different ideas and approaches, and absorbed feedback. At the time, I was ten years removed from graduation, a full-fledged vascular surgeon who thought he could do any operation put before him. I cannot imagine the thoughts churning through the head of a recent graduate faced with the choice of taking on an open aortic operation with only 5 cases under their belt, referring the case on to the regional tertiary center, or trying to McGyver an endovascular solution. I contributed probably about 35-50 cases to this paper, but the outcomes were a collective effort. Even today, I will run cases by Sean Lyden, Christopher Smolock, or Lee, if only for the company and gossip.
“We explain this distribution of cases as a function of several factors: a unique, broad regional quaternary referral practice whereby patients with complex aneurysmal disease are referred to our institution; an institutional practice evolution resulting from a critical analysis and understanding of EVAR failure modes that lead to explantation, thus generating a different perspective in the EVAR vs open decision-making process; a parallel high-risk IDE fenestrated graft study; a historical willingness to accept all physician and self-directed patient referrals (ie, a willingness to manage more complex cases); and a published expertise in the area of EVAR device explantation with an annually growing volume of commercial device removals” -from El-Arousy et al.
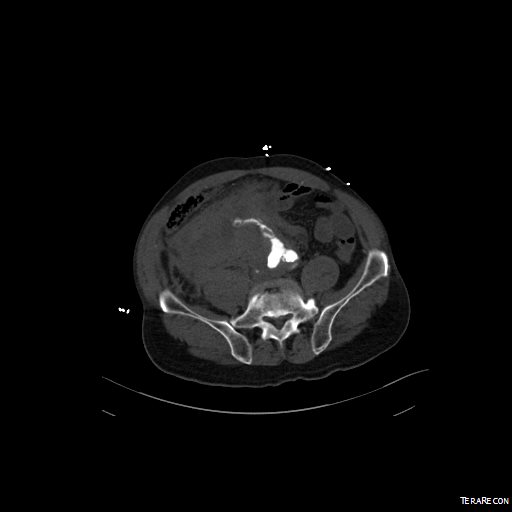
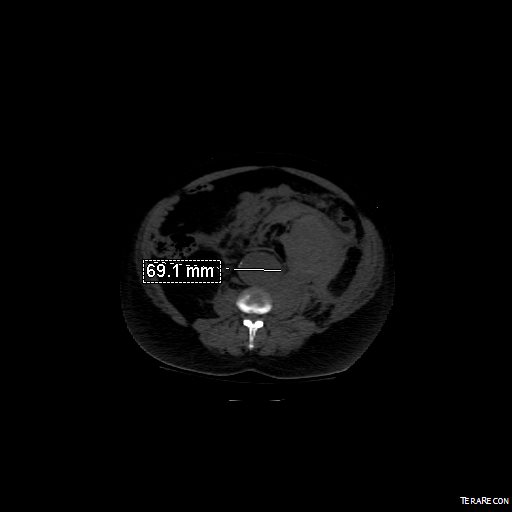
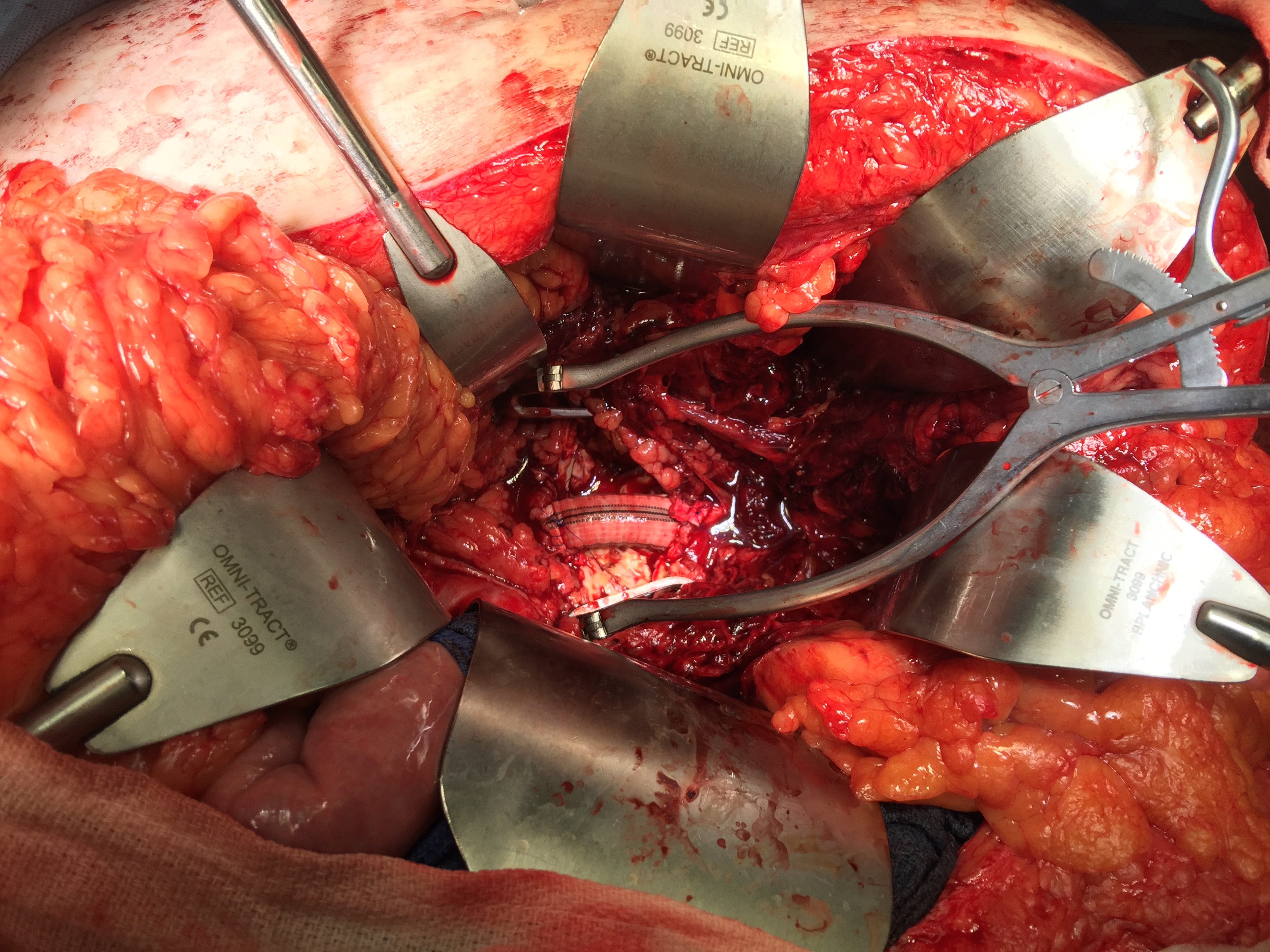
Reading through that paper, I have come to the conclusion that the increasing open aortic volumes at the Cleveland campus has as much to do with the ongoing retirement of experienced surgeons regionally as it does with the ability to attract these cases. Loss of these surgeons has a cascade effect like losing a species in an ecosystem. The operating rooms forget where the OMNI retractor is because nobody asks for it anymore. The ICU’s are no longer familiar with the ebb and flow of the postoperative open aortic operation. The floors lose institutional memory of the care of these vascular patients as the stent grafts and interventions go home within 48 hours, sometimes the same day.
If you were a vascular surgeon born before 1970, your approach to the scenario of the ruptured abdominal aortic aneurysm may differ substantially from that of surgeons born after the Carter administration. Most of my cohort, Gen-X and older, feel comfortable applying some betadine, opening the belly and placing a clamp. Those younger than us have told me they feel more comfortable putting up a large balloon and deploying a stent graft. In this generation, it is normal to call a general surgeon to decompress the abdominal compartment syndrome and manage the abdominal vacuum dressing. For our generation, the giant industrial robot arms and 80 inch monitors creates a barrier to the problem at hand, and gathering all the extra staff after hours and on weekends requires the logistical skills of a wedding planner.
We prefer an operating table, a willing anesthesiologist, a cooler full of O-neg blood, Prolene and a graft, strong suction, and an extra set of hands. The data suggests either method is equivalent in outcome, but I would argue that depending on the circumstance, there is an optimal method for that patient and you have to have the ability to do either open or endovascular or some hybrid combination. Unfortunately, it is clear that open vascular surgery is year over year diminishing, and and it might not be so great when we start rupturing our aneurysms.
The open approach is preferred because we got good at it by doing a lot of these cases. Your hands -it becomes natural to change the course of the disease and the fate of the patient with your hands. One of the things you lose with a wire based approach is the tactile feedback from the organ that you are treating. Yes, there is a subtle feedback from the flexible tip of a Glidewire, but that’s missing the point. The thing that is rarely considered with open surgery is the tactile aspects of operating.
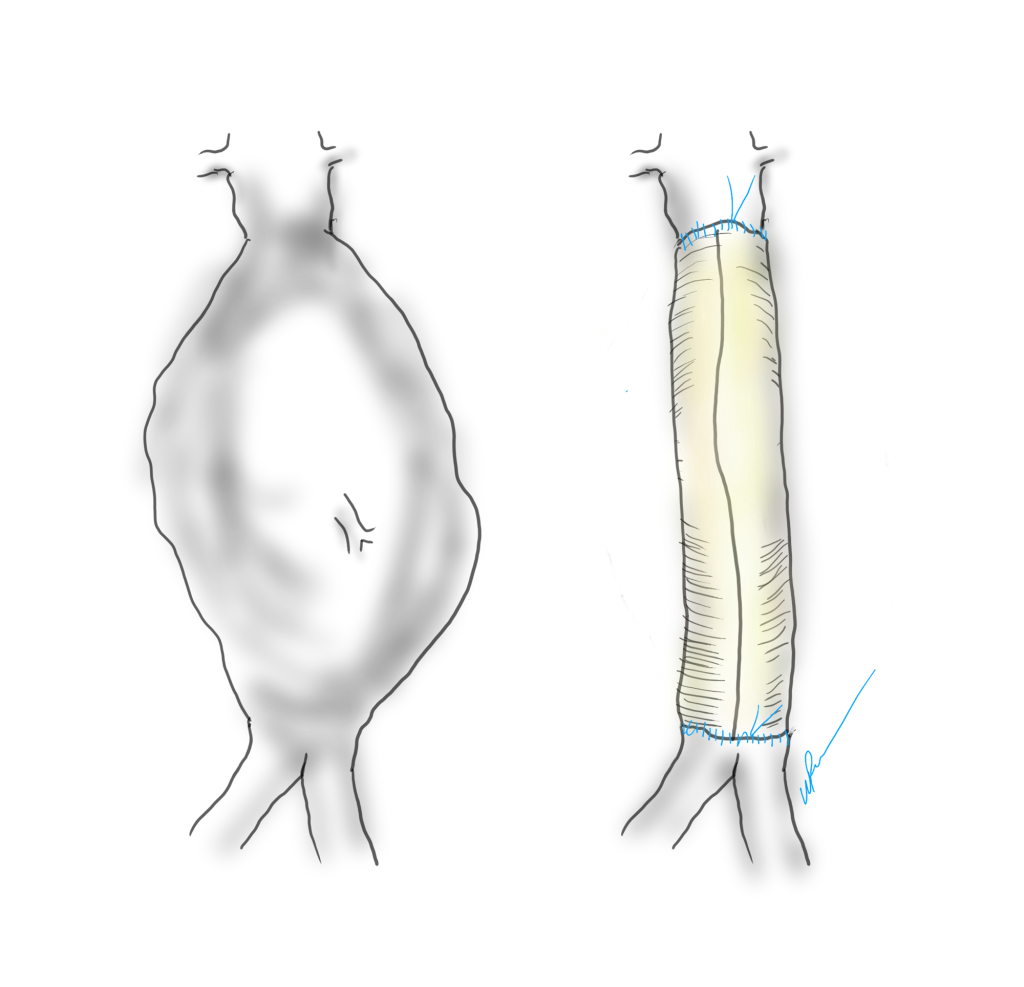
Your fingers are your point of care ultrasound. As an intern, one of my earliest tasks was reaching in through a 2cm incision with my index finger, feeling for what I would describe as a rotten shrimp, and delivering it out by hooking my distal phalanx around its base. Adhesions were rubbed like money between finger and thumb to judge if you could bovie through it. If you felt a sliding sensation, it was mucosa to mucosa and you looked for another spot to cut. Into my fellowship which could be called the triple-redo, no-one else wants to do-, difficult vascular operations fellowship, the pulse or the plaque felt under the finger would guide me to carve away scar tissue from blood vessel, visualizing the feedback from the fingers. In a rupture, with the belly under a dark mire of blood, there is no seeing, only feeling. Your hands reach into the lesser sac or transverse mesocolon and strangle the aorta -your fingers while clamping, feel and avoid the caudate lobe, the NG tube in the esophagus, and split the crura of the diaphragm like a pick pocket. Once the pulse returns as anesthesia refills the tank, you scratch free the aorta with your thumb and forefinger, then slide the jaws of the aortic cross clamp over your fingers and against the spine and clamp. This takes about 60 to 90 seconds (link).
When a patient is bleeding out, this is the way to control the bleeding. In practice, no amount of rehearsing for getting a patient into a endovascular suite, getting airway and access, swinging in the industrial robot arm, and getting everyone into lead aprons, sending up wire, placing a 12F sheath and an aortic occlusion balloon, will be satisfactorily smoothly and efficiently as a STEMI or STROKE alert. The rAAA is for most hospitals, unless you are in Seattle, a once in a while occurence. Many more people can find a scalpel and an aortic clamp than they can find a 32 inch aortic balloon, a stiff exchange length wire, and a 12F sheath.
When a patient presents with a slowly bleeding, contained rupture, there is time to assemble the teams required for an endovascular repair, and for opening and decompressing the abdomen, for anesthesia to get IV’s, central lines, arterial lines, and order crossmatched blood. One has the time to get and review CT scans and choose grafts. One can even do things backwards, debranching after securing the leak (link) with a stent graft. The luxury of time should signal to you that the endovascular option is the preferable route, as all the advantages of minimally invasive repair are possible. Rural hospitals sending patients two hours by ambulance or arranging for a helicopter -this is the great filter through which those likely to survive make it into the endovascular suite. These patients do great with EVAR, because everything moves more or less like a routine elective EVAR.
The setting up the operating room for tackling rAAA is quite simple. Keep everything nearby. Nothing should ever be stored out of sight, retrievable only by arcane codes whispered in the ears of people down in the basement or across the street. Amazon gives itself a day to get that gadget to you, but the rupturing patient does not have the time to have a 28mm stent graft ordered by looking up a Lawson number, finding the materials person in the faraway room to find it, running it over a city block. The stuff has to be next to the OR. Every scenario is unique, and the best planning is assuming no one person knows where everything is but everything is close at hand -major vascular sets, retractors, C-arm, cell saver, stent grafts, open grafts, balloons, cardiopulmonary bypass -every gewgaw is few steps away. The inventory is what you see, because if you can’t grab it, it does not help the hemorrhaging patient.
If you are a vascular surgeon born after 1980, it is likely that you may have trained in a 0-5 residency and all the old people harumphing about the old ways seem biased. Rather than being rational about their awful upbringing, the old people seem to suffer from Stockholm Syndrome, turning from victims of a harsh and brutal system inherited from the original, Halsted, a cocaine addict, to willing collaborators now mooning about the good old days of every other day call and 120 hour work weeks.
There might be a growing suspicion about advocating for open surgery when fewer people can perform it. One of the truisms of surgery is if only one surgeon claims to be able to do a rare operation with great results when everyone else abandons it, like venous valve surgery or robot assisted mastectomies, it can mean that surgeon might be uniquely talented or shamelessly selling something. It is a shame that open vascular surgery is devolving into that category of arcana, like the Jedi. I have no doubt that the last open vascular surgeon will be a reclusive, bitter, wild eyed hermit like Luke Skywalker was in episode VIII, if we let it get that far.
Bald eagles were saved from extinction. The methods of species reclamation may be what is needed to save open vascular surgery. Financial metabolism drives behavior, and there must be recognition of the time and dedication required to perform good open vascular surgery in the form of increased RVUs and reimbursement. The surgeons retiring in their mid 60’s with straight backs and steady hands need to be incentivized to stay around and coach the next generation in the ways of the Jedi. Call it the master surgeon designation. Every 0-5 graduate needs to focus on getting 100 leg bypasses, 50 carotid emdarterectomy, and 25 open aortas within the first five years of practice with a master surgeon if they did not get this experience during training. Like dead Jedi, it would help even if they were just virtually present, shimmering on Facetime in their (bath)robes to go over planning and approaches, but being physically present and reimbursed for it would make the most sense.
There is always self service in any human activity. One mildly prominent vascular surgeon that I have come across is famous for saying he did not have a vascular fellowship because he did not want to train his competition. It is easy for the fifty somethings to sit and proffer their open skills and profit from its scarcity but it goes against decency to not pass on this collective body of hard won knowledge and skills. There must be stewardship of this great thing we do, this honorable and treasured endowment.
Of the concrete ways we are trying is creating a network of advanced open surgery capable surgeons regionally organized by Martin Maresch, capitalizing on social media and electronic communications. Here at CCAD we are in the organizing phase of a vascular residency, and I very fortunate to have Houssam Younes join us as he shares my interest in surgical education and open vascular surgery. We are contemplating a non-accredited fellowship. We have general surgery residents coming through our service as well as medical students.
One of my mentors told me, “I can train a monkey to do cardiac surgery,” as he was training me to do cardiac surgery. And he was right. The final comment I have is you have to demystify surgery, take away the Instagram perfection, the romance, and list in practical terms the toolkit of maneuvers that form the component parts of all operations and propagate it. Let us not kid ourselves. The technical skills of surgery can be taught to anyone. The Mayo brothers were performing surgery as teenagers before medical school. The knowledge and experience and judgement -that varies as much as people vary and we have a curriculum for that, but the physical acts of surgery need to be taught starting at the medical school level. Standardized drills and exercises need to be created so that proficiency can be metered.
“The individual per trainee OAR volume did not decrease during this period. In the training program, the use of “component separation” (separation of each
operation into discrete, instructionable steps that facilitates trainee mastery) is integral to instruction of open aortic aneurysm repair techniques and permits the
trainee to master all of the technical exposure and repair skills necessary to address and to manage both straightforward and complex aneurysm anatomy. Component
separation is essential to maximize trainee experience across all levels” –from reference 1
Here is my list of things a trainee must accomplish by the time they graduate from a vascular residency or fellowship.
1. Tying knots with gloves on with 6-0 Prolene inside a pickle jar without lifting or moving a 12 ounce lead fishing weight to which the suture is being tied, fast, one handed, two handed, left and right handed.
2. Holding forceps, needle holders, and clamps
3. Correct operation of the OMNI retractor, Weitlander retractor, Balfour retractor, Thompson retractor
4. Incise skin through dermis through correct depth and length with both #15 and #10 blade
5. Open the abdomen through midline and flank incisions and close these incisions
6. Harvest saphenous vein
7. Vascular anastomosis on a table, inside a pickle jar, inside a short Pringle’s can
8. Dissection of adhesions and scar tissue around blood vessels and organs
9. Dissect and expose the common femoral artery via vertical and oblique incisions and close these incisions
10. Dissect and expose the carotid bifurcation, left and right side, and close these incisions
11. Dissect and expose the tibial vessels in various parts of the leg and foot
12. Dissect out the brachial artery at the elbow
13. Dissect out the axillary artery and vein below the clavicle
14. Dissect out the axillary artery and vein from the axilla
15. Dissect out the subclavian artery, vein, and brachial plexus above the clavicle
16. Dissect out the arm veins
17. Dissect out the iliac artery via a lower quadrant pelvic retroperitoneal exposure
18. Dissect out the abdominal aorta via midline laparotomy
19. Dissect out the abdominal aorta via retroperitoneal approach
20. Dissect out the thoracoabdominal aorta via a thoracoabdominal exposure
21. Dissect out the popliteal artery via suprageniculate, infrageniculate incisions and prone position
22. Dissect out the inferior vena cava
23. Dissect out the iliac veins
24. Harvest deep femoral vein
25. Temporal artery biopsy
26. Endarterectomy of carotid, femoral artery, any artery with patch angioplasty
27. Exposure and control of supraceliac aorta, suprarenal aorta for clamping
28. Exposure and control of thoracic aorta
29. Exposure and control of the great vessels via sternotomy and supraclavicular incisions
30. Exposure and control of the vertebral artery
31. Safe removal of vascularized tumors
32. Amputations of digits, legs and arms up to pelvis and shoulder
33. Exposure and control of radial and ulnar arteries
34. Hand surgical techniques of exposing arteries, tendons, and nerves in forearm and hand
35. Plastic surgical techniques of skin grafting and basic rotational flaps
36. Fasciotomy of arms and legs, hands and feet.
37. Exposure and control of celiac axis
38. Exposure and control of superior mesenteric artery
39. Exposure and control of left renal vein
40. Exposure and control of hepatic veins, portal vein
41. Exposure and control of renal arteries
42. Exposure and control of profunda femoral arteries
43. Safe removal of spleen
44. Transabdominal retroperitoneal exposures of the abdominal aorta and inferior vena cava
45. All of the above in a reoperative field
46. All of the above with limited visualization and by sense of feel only
47. Laparoscopic and thoracoscopic techniques
48. Orthopaedic surgical techniques of myodesis, bone grafting, precision osteotomies, infection control, external fixation, spinal exposure
49. Safe resection and anastomosis of bowel
50. Drainage of infection
51. Intensive care of SIRS, MOFS, CHF, Septic shock, postoperative fluid shifts
52. Nonsurgical and surgical management of lymphedema, seromas, and edema
53. First rib resection
54. Spinal exposure
55. Organ harvest and transplantation
56. Planning of complex open, hybrid, and endovascular procedures
Every year, it is apparent that endovascular options suffer from some flaw when outcomes are studied beyond 2 years, but progress will march on in that sphere. It has to. The loss of open capable surgeons to early retirement is accompanied by overapplication of endovascular techniques at least partly due to the lack of knowledge of these open surgical options and achievable good results and partly due to financial incentives. The solution lies in redistribution of reimbursement to open procedures and creation of open surgical fellowships and identifying and empowering mentors who still walk among us.