
Every once in a while, I will make an exception to the SVS guidelines on AAA repair with regard to size at time of repair (link). I have a bunch of excuses. When I trained in 2000-2002 with several giants of vascular surgery, there was some controversy when the first guidelines came out in 2003 (link). The board answer became 5.5cm that year, but where I trained, it was a minority opinion held by Dr. Jeb Hallett. The majority was in the belief that as long as operative mortality was low, even high risk AAA repair could be undertaken (link). The published risk for Mayo was low, and that came from both technical excellence and high volume (more on that later). The criteria during my fellowship was 4.5cm in good risk patients for open repair based on data generated in the 1980’s and 90’s during Dr. Hollier’s tenure.
Then as now, the debate centered around the balance of risk. At specialty centers that achieved less than 1% mortality rate for elective open AAA repair, 4.5cm in good risk patients would seem perfectly reasonable. But given the 5-10% mortality seen in the Medicare database at that time for community practice, the 5.5 cm criteria was not only good science, it was prudent. The first set of guidelines held off the contentious volume recommendations that was the nidus of conflict within other surgical societies.
The advent of endovascular was a game changer -the mortality rate in the Medicare databases was 1-2% for EVAR in the community setting, meaning more surgeons in most hospitals could achieve tertiary center levels of mortality with this new technology. The issue was never really settled in my mind through the 2000’s, even with the PIVOTAL Study. I enrolled patients into the PIVOTAL Study (link) at that 4.5cm threshold during my time in Iowa. Eventually I lost equipoise and I stopped enrolling after a handful of patients. It had to do with graft durability.
Around that time, I took two patients in a row to the operating room for sac expansion without identifiable endoleak. They were Dacron and stent-based endografts placed about 5-7 years before by another surgeon and aortography failed to show type I or III endoleak. Sac growth was over a centimeter in 6 months and the aneurysm size was over 6cm in both. I chose to marsupialize the sac and oversew any leaks, with the plan to replace the graft if there was a significant leak. On opening the sac, no significant lumbar or IMA leaks were encountered but in these patients a stream of blood could be seen coming from the sutures securing the stents. It was the same graft that was in the trial, the AneuRx, and that was when I realized that these grafts have the potential to fail in the same way that patio umbrellas leak after years of use -cloth sewn to rigid metal with movement wears open the cloth wherever there is stitching. This did not happen with open repair. I lost enthusiasm for the trial as I lost faith in this graft which was retired from the market. I placed pledgetted sutures to close the leaks on both patients, and closed the aneurysm sac tightly around the graft in one patient who was higher risk, and replaced the stent graft in the other.
There are some exceptions to justify repair of 4.5-5.5cm AAA. During my time in practice, there were patients who lived far away from major medical centers who would not survive a ruptured AAA even if the rupture rate was low and who confessed they only came into town every five years or so. There were patients who suffered from clinical anxiety whose AAA was documented by a psychiatrist to amplify their anxiety. There were patients with vague abdominal pain for whom thorough workup have ruled out gastrointestinal causes and every visit to the ER triggered a CT scan to rule out AAA rupture. And there seemed to be some patients who seemed to have such perfect anatomy for EVAR, whose risks were low, and whose growth rates were so consistent that their repairs could be timed on the calendar. Some combination of these factors and lobbying on the part of the patient got them their repair in the 5cm range. And they still do.
The patient is a man in his sixties with hypertension who presented with a 4.7cm AAA which in various reports he came with described 5.2×4.7cm. After review of his images, it was clear it was 4.7cm. If measured on a typical axial cut CT scan or a horizontally oriented ultrasound probe, a cylindrical aortic aneurysm will be seen as an ellipse in cross section. A radiology report will typically report an aneurysms length and the anteroposterior and lateral dimensions. If you cut a sausage at an angle, the ovals you cut can be quite wide but the smaller length of the oval reflects the diameter of the sausage.
Looking back at his records, for three years he had multiple CT scans for abdominal pain showing the AAA and a well documented record of growth of about 2-3mm annually -the normal growth rate. He asked me to prognosticate and so I relayed that 4.7cm in 2017 with a 3mm growth rate, we would be operating in 2020. The anatomy was favorable with a long infrarenal neck and good iliac arteries for distal seal and access. He was quite anxious as whenever he had abdominal pain, his local doctors would discuss the AAA and its risks or order a CT. After a long discussion and considerable lobbying by the patient and family, I agreed to repair his 4.7cm AAA.
The EVAR was performed percutaneously. No endoleak was detected by completion arteriography (figure). He was soon discharged and was grateful. In followup, CT scan showed excellent coverage of the proximal and distal zones and absence of type III endoleaks. There was increased density to suggest a type II leak, but his inferior mesenteric artery was not the source of it. over a three year period, his aneurysm sac continued its 2-3mm of annual growth despite the presence of the the stent graft.
While CT failed to locate this endoleak, abdominal duplex ultrasound did showing flow from a small surface vessel (duplex below, figure at beginning of post). It was not the inferior mesenteric artery which can be treated endovascularly (link) or laparoscopically (link). CT scan suggested that it was one of those anterior branch vessels that one would encounter in exposing the aorta. Usually these were higher up as accessory phrenic arteries, but these fragile vessels, larger than vasovasorum, but smaller than named aortic branches, are seen feeding the tissues of the retroperitoneum.
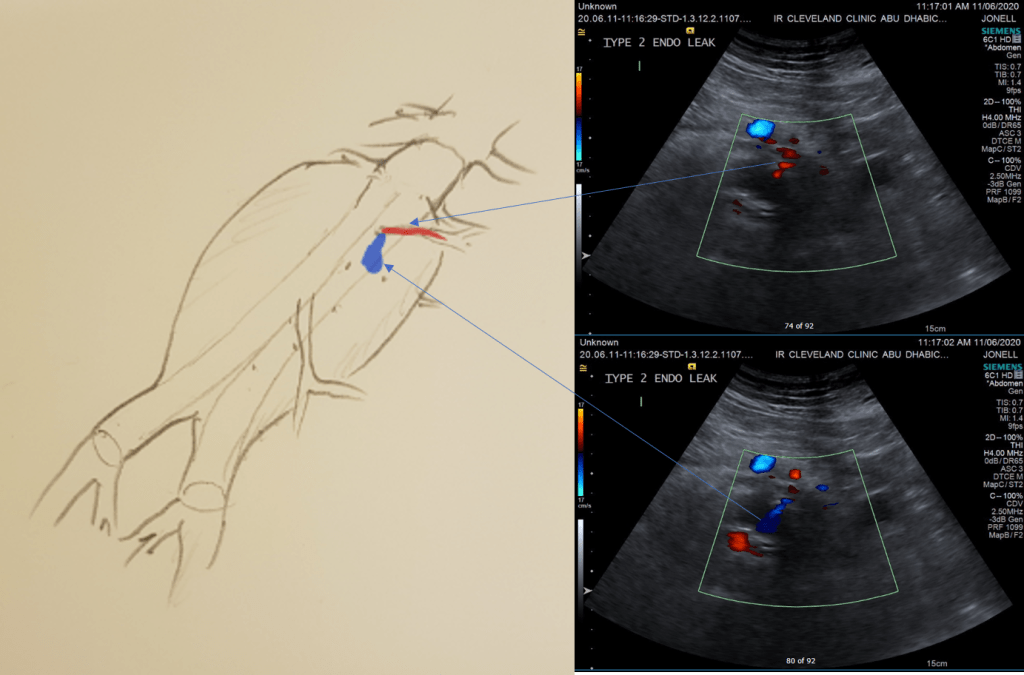
Type II endoleaks are not benign. The flow of blood into the aneurysm sac after stent graft repair is almost never benign. It is a contained hemorrhage. There are three components to the pressure signal seen by the aortic aneurysmal wall that could trigger breakdown, remodeling, and aneurysm growth. They include pressure, heart rate, and the rate of change of pressure. The presence of fresh thrombus may play an inflammatory role. Some endoleaks clearly have a circuit and others are sacs at the terminus of their feeding vessels, never shutting down because the AAA sac can both accept and eject the blood flow. Changes in AAA sac morphology due to sac growth can cause problems with marginal seals, component separation, and component wear. Sac growth can cause pain. Ruptures, while rare, can cause death. Mostly, type II endoleaks generate more procedures because it is hard to ignore continued growth.

Three years of followup showed growth of the AAA sac to 5.5cm, which ironically threshold for repair. Again, no type I or III endoleak could be seen. He reached his calculated repair date, and I discussed our options in detail.
1. Do nothing, keep following
2. Endovascular attempt
3. Open surgery, marsupialization
4. Laparoscopic ligation of target vessel
Doing nothing hasn’t worked for 3 years. What would more time buy? Endovascular -to where. The IMA is the usual target for an endovascular attempt, although iliolumbar access is possible (link), we really needed to fix this with one attempt. Open surgery is a great option -a short supraumbilical incision is all that would be needed to open the AAA sac and oversew the collaterals. The patient did not want a laparotomy. There are reports of laparoscopic guided endovascular access with endovascular coiling of the remnant sac with fluoroscopy. This adheres to the letter of the claim of minimal access, but really?
I compromised with the patient and offered laparoscopy. I have ligated the IMA a handful of times laparoscopically -these are relatively fast and straightforward cases. As I had the location of the endoleak, I felt it should be straighforward to dissect out the anterior sac much as in open repair and clip this vessel.

Of course, what should have been a 30 minute procedure through a minilaparotomy became a two hour enterprise getting through scar tissue (not the first time encountering this after EVAR) while pushing away retroperitoneum. I recruited the help of general surgery to get extra hands, but the patient was well aware that there was a good chance of conversion. Patience won out as the artery was ultimately clipped and endoleak no longer seen on ultrasound.
I waited a year before putting this together as I wanted CT followup. The sac stopped growing and has shrunk a bit back to 5cm or so. There will be those who argue that nothing needed to have been done about this leak as it would have stopped growing eventually, but I would counter that an aneurysm sac that kept growing like the stent graft never went in is one demanding attention. The key role of duplex ultrasound cannot be minimized. We have an excellent team of vascular scientists (their title in Europe), and postop duplex confirmed closure of the leak.

The patient is quite satisfied having avoided laparotomy. His hospital stay was but a few days. During my conversations with our general surgeons who are amazing laparoscopists, that this would have been a nice case with the robot. That’s a post for another day.
The definition of success in this case and many EVAR’s plagued by type II leaks leaves me wondering. Excellent marketing of the word “minimally invasive” has subtly defined laparotomy as failure, and not just in vascular surgery. When costs and efficacy are reviewed as we come out of this pandemic, I suspect that open surgery will selectively have its day in the sun. A ten blade, a retractor, a 3-0 silk is so much more cost effective than five ports and disposable instruments. And a stent graft system?
Maybe I am just a dinosaur.

2 replies on “Off the guidelines: type II endoleak can derail the perfect EVAR”
Perfect is enemy of good.”minimally invasive”but some endoleaks will happen in future,so life long follow up is mandatory.
Vascular Surgery is a quest for excellence.-Edwin J Wylie
It Is Not the Strongest of the Species that Survives But the Most Adaptable-sombody said.
There is no perfect answer,but,,,.I believe that there is excellent operation.
LikeLike
Life long surveillance and rarional action on the findings detected will be the cost to end aortic work.
Compound this with it being done buna cohort of IR Cardiology and Vascular Surgeons all with different training levels and exposure.
Rhennrhenicinhbos marketing of endo as the newer new.
Such experiences will be more common and all I can say is ” Expand our explant skills”
LikeLike