From my archives, the CTA illustrates two points. First, tunneling can be done without taking down much of the retroperitoneum. This lesson came to me after taking a course in laparoscopic aortic surgery with Dr. Dion in Quebec City. The old BARD-IMPRA tunnelers with their bullet tips -the short gently curved one, is particularly well suited for tunneling from the groin to the aorta -if you have a hand on the retroperitoneal pelvis, it is very straightforward to guide the tunneler to the proper location. The other point is that the graft is applied proximally end to side with a leftward orientation. This combined with dissection of the retroperitoneum with a large Ligasure or harmonic scalpel lets you avoid the problem of having no tissues to close over the graft. You take down the retroperitoneum with a cuff of tissue of about 5cm from the duodeum. Normally, this can be bloody but with the energy devices, it is not. This provides excellent graft coverage. You just have to mind the IMV which may or may not have to be taken down. End to side is preferred because you preserve endovascular options, but in this case, the anastomosis was done end to end.
April 30, 2009 11:33 PM
Aortic Bypass for occlusive disease
The patient arrived with the history of severe claudication. He was a middle
age smoker whose job required walking several miles a day. This became
increasingly difficult until he was clearly limping at short distances. He was
also developing cramps in his legs at night, worse in his left leg.
On examining him, he had no pulses in his left leg from the groin down.
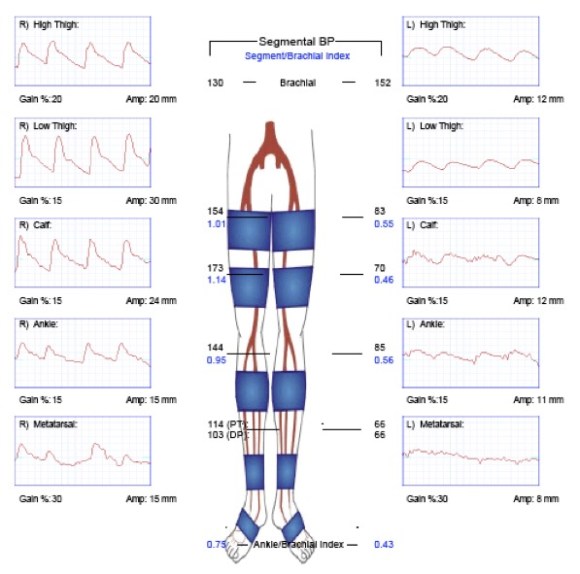
The pulse volume recordings (PVR’s, red lines) on the left clearly
demonstrate normal flows in his right leg with sharp upstrokes, dicrotic
notch, and shallow diastolic relaxation. The left leg had attenuated flows on
the pulse volume recordings with dampened, gradual series of mounds.
The flow was flat at the metatarsal level (foot). His ankle brachial index
( BI) on the right leg was 0.75 which was mildly depressed. The ABI on his
left leg was 0.43 which was severely depressed.
CT was performed (above left) showing that his left iliac system was
occluded. This is due to atherosclerosis which is a systemic disease. This
kind of blockage can occur in any organ, but it was most severe in this
patient’s leg. His right common iliac artery also had moderate plaque.
He underwent an aorto-right iliac and left femoral artery bypass with a
bifurcated graft (above right). This greatly improved flows in his left leg, with
his BI improving to 1.05 from 0.43. The PVR’s also reflect this improved
flow. The right leg, surprisingly, also had an improvement despite not
having a severe stenosis in his common iliac artery. The fact is, the
common iliac artery, but being heavily diseased over the length of the
artery, offered a hemodynamically significant stenosis despite being patent.
His BI on the right improved from 0.75 to 1.03.
The operation was done with minimal invasiveness in mind. The CT
allowed for planning of the abdominal incision directly over the part of the
aorta requiring operation. The groin incision on the left was created
obliquely as to avoid crossing the groin crease -which I believe increases
the chances for tension on the wound and subsequent infection. The graft was tunneled without mobilizing the sigmoid colon directly up to the bifurcation using an IMPRA tunneler -by placing the hand in the pelvis, the
tunneler can be felt and guided in the correct trajectory. The graft was a
Gelsoft Plus graft soaked in Rifampin. This antibiotic bonds to the gelatin in
the graft giving protection against indolent bacterial infections for about 3-6
months after the procedure -this is helpful especially with groin incisions. The operation took 2.5 hours and the patient went home within a few days.
The patient is now walking without pain and will be returning to work. He
has also successfully quit smoking which has a significant impact on his
risks of future heart attack, stroke, or peripheral vascular complication. His
relative youth (in his fifties) required that we give him a repair that would
give him the best chance at maintaining patency for many years. The aortic
bypass graft for occlusive disease has a proven track record with patency
measured in decades.